L. Wang
E-mail: wanglimin@im.ac.cn
Phone/Fax: +86-10-64806132
© 2019 Sift Desk Journals. All Rights Reserved
VOLUME: 5 ISSUE: 6
Page No: 252-262
L. Wang
E-mail: wanglimin@im.ac.cn
Phone/Fax: +86-10-64806132
Lihong Fu1,2, 3, Jiansong Ju 3, Hamed M. El-Shora4, Shuwen Zhang1, Bo Yu2, Limin Wang1, 2*, Jiaping Lv1
1 Key Laboratory of Agro-Products Processing,Ministry of Agriculture and Rural Affairs/ Institute of Food Science and Technology, Chinese Academy of Agricultural Sciences, Beijing 100193, People’s Republic of China
2 CAS Key Laboratory of Microbial Physiological and Metabolic Engineering, Institute of Microbiology, Chinese Academy of Sciences, Beijing 100101, PR China and State Key Laboratory of Mycology, Institute of Microbiology, Chinese Academy of Sciences, Beijing 100190, People’s Republic of China
3 College of Life Science, Hebei Normal University, Shijiazhuang 050024, PR China
4 Department of Botany, Faculty of Science, Mansoura University, Dakahlia 35516, Egypt
Lihong Fu, Jiansong Ju, H. M. El-Shora, Shuwen Zhang, Bo Yu, L. Wang, Jiaping Lv. Extracellular production of Strepto-myces ladakanum transglutaminase in a food-grade strain, Bacillus subtilis. Journal of Food Science & Technology 2020; 5(6): 252-262
Background: Transglutaminase (TG) is an enzyme of the transferase family with cross-linking properties, which has been widely used in the food industry. Traditionally, TG is isolated from strains of Streptomyces sp. However, the development of a facile and efficient production of commercial TG is always desirable.
Purpose: In the current study, we described an efficient route for TG production in a food-grade strain of bacteria, Bacillus subtilis.
Method: Two strategies were employed for the extracellular production of S. ladakanum TG in B. subtilis. Sixteen signal peptides were optimized to secret TG into the extracelluar medium. Site-directed mutageneses in pro-peptide were further utilized to improve the enzymatic activity. The enzymatic characteristics of S. ladakanum TG expressed in B. subtilis were analyzed.
Results: The N-terminal amino acids played important roles in enzymatic activity. Signal peptides of SacB (SPSacB) and AbnA (SPAbnA) showed good abilities to direct the secretion of TG into the medium. The enzyme was secreted into the medium and exhibited good Ca2+ stability and temperature stability, which were comparable to those produced by commercial strains. The enzymatic activity in the supernatant of culture reached 7.6 U/mg.
Conclusion: Our study demonstrated that B. subtilis may be a good candidate for the efficient and stable production of TG and has a much easier purification process.
Keywords: Transglutaminase (TG), Streptomyces TG, Bacillus subtilis, Food-grade strain.
Transglutaminase (TG, EC 2.3.2.13) is an enzyme that catalyze the cross-linking between proteins, peptides and various primary amines [1]. It has a great commercial use in food industry. In recent years, it is also widely used in tissue engineering, textile and leather processing, biotechnology tools and other non-food fields, which increases the demand for TG [2]. TGs are distributed in various sources, including mammals, plants, microorganisms. It was firstly found in animal’s tissues in 1973. But animal TG is rarely used in food industry, because of the red pigmentation and the property of calcium (Ca2+)-dependence [3, 4]. Microbial TG was discovered in bacteria belonging to the actinomycetes [5]. Microbial TG has low molecular weight (approximately 40 KDa) and is stable at a pH range of 4.5 to 8.0 [6]. It is Ca2+-independent and could be obtained by conventional fermentation. Because some ingredients in food are easily precipitated by Ca2+, Ca2+-independent TG has a great potential in food industries [7]. Until now, many TG producing microorganisms have been identified, and some TGs were heterologously expressed in Escherichia coli, Corynebacterium glutamicum and Streptomyces species [6]. Although microbial sources of TGs allow for simplified extraction processes compared with those from animal sources, the development of an efficient and easy-to-use method for TG production, which requires less energy and provides economical savings, is highly desirable [8].
In Streptomyces, TG is synthesized as inactive zymogen containing the central body and an N-terminal pro-peptide and is then activated by the removal of pro-peptide. Apart from pro-peptide and central body, the N-terminal signal peptide plays an important role in targeting TG to the periplasm [9]. The obtain of soluble TG is one of the key problems for heterologous TG. It has been reported that expression of pro-peptide prior to that of TG was essential for the active TG production in E. coli [8]. E. coli is a popular host for heterologous protein expression. However, considering TG’s application in food processing, E. coli is not a suitable host for TG production because of the formation of endotoxin [10]. Compared with E. coli, Bacillus subtilis is a commonly used host for secreted expression of heterologous proteins. It is recognized as GRAS (Generally Recognized as Safe) strain by FAD in the United States with the known genetic background [11]. Some wild-type B. subtilis strains can produce TG, but the yield is too low to be industrial used [12]. It has been reported that S. mobaraensis TG expressed in B. subtilis showed similar enzymatic activities to that produced from E. coli and catalyzed the cross-linking reactions [13]. In a previous report, we demonstrated that active-form S. mobaraensis TG was efficiently secreted by B. subtilis when a modified Saccharomyces cerevisiae vacuolar ATPase subunit (VMA) intein was introduced into TG zymogen [14]. In this study, two strategies were employed to express S. ladakanum TG in B. subtilis. Sixteen signal peptides were optimized to secret TG into the extracellular medium. Site-directed mutageneses in pro-peptide were further utilized to improve the enzymatic activity. Our study demonstrated that B. subtilis may be a good candidate for the efficient and facile production of TG.
2.1. Strains and chemicals
Bacterial strains and plasmids used in the study are listed in Table 1. S. ladakanum (China Center of Industrial Culture Collection, CICC 11018) was a gift from Prof. Chunbo Lou, Institute of Microbiology CAS, China. The B. subtilis strain WB600 (Novagen Company, Shanghai, China) and Bacillus subtilis 168 were stock preserved and maintained in our laboratory. Competent E. coli TOP10 was purchased from Tiangen Co., China. Plasmid pWB980 was purchased from Novagen Company, Shanghai, China. Plasmid pMD19-T was purchased from TaKaRa Company, Beijing, China. Kanamycin (Km) and ampicillin (Amp) were purchased from Tiangen Co., China, and added to the medium at a final concentration of 20 μg/mL and 100 μg/mL, respectively, whenever necessary. The restriction enzymes and DNA modifying enzymes were all purchased from TaKaRa Co., Ltd, China. The Clone Express® MultiS One Step Cloning Kit was purchased from Vazyme Biotech Co., Ltd, China. Other chemicals and reagents were purchased from Sigma-Aldrich (Shanghai, China).
Table 1. Bacterial strains and plasmids used in this study
|
Strains |
Genotype/properties |
Source |
|
Escherichia coli top10 |
Cloned strain; mcrA Δ (mrr-hsd RMS-mcrBC) φ80 lacZ ΔM15 △lacⅩ74 recA1 ara Δ139 Δ(ara-leu)7697 galU galK rps (Strr) endA1 nupG |
Tiangen |
|
Bacillus subtilis 168 |
Cloning of promoters (PabrB, PspoVG, PyabR, PrsbW, PgsiB, PyqfD, PsigW, PftsZ, PyvyD) encoding genes; wild-type |
Our laboratory |
|
Bacillus subtilis WB600 |
Bacillus subtilis 168 derivate,ΔnprE ΔaprE Δepr Δbpr Δmpr ΔnprB; Protease deficiency type, expression host strain |
Our laboratory
|
|
Streptomyces ladakanum |
Cloning of Lpro gene;wild-type |
A gift from Prof. Chunbo Lou |
|
Plasmids |
|
|
|
pMD19-T |
Efficient TA cloning vector constructed based on pUC19; Apr |
TaKaRa |
|
pMD19-T-LTG |
pMD19-T carrying TG gene from S. ladakanum; Apr |
This work |
|
pMD19-T-LTGDSD80-82AAA |
pMD19-T carrying TG gene from S. ladakanum with DSD80-82AAA mutation; Apr |
This work |
|
pMD19-T-LTG△PS |
pMD19-T carrying TG gene with PS deletion from S. ladakanum; Apr |
This work |
|
pMD19-T-LTGSGS34-36AAA |
pMD19-T carrying TG gene with SGS34-36AAA mutation from S. ladakanum; Apr |
This work |
|
pMD19-T-LTGPS74-75AA/DSD80-82AAA |
pMD19-T carrying TG gene with PS74-75AA/DSD80-82AAA mutation from S. ladakanum; Apr |
This work |
|
pWB980 |
Expression plasmid with signal peptide SacB in B. Subtilis; Kmr |
Novagen |
|
pWBsacB-LTG |
pWB980 carrying TG gene from S. ladakanum; Kmr |
This work |
|
pWBsacB-pro-sacB-LTG |
pWB980 carrying the genes encoding SPsacB, pro-peptide, SPsacB and TG from S. ladakanum; Kmr |
This work |
|
pWBsacB-LTGDSD80-82AAA |
pWB980 carrying TG gene from S. ladakanum with DSD80-82AAA mutation; Kmr |
This work |
|
pWBsacB-LTG△PS |
pWB980 carrying TG gene from S. ladakanum with PS deletion; Kmr |
This work |
|
pWBsacB-LTGPS74-75AA |
pWB980 carrying TG gene from S. ladakanum with PS74-75AA mutation; Kmr |
This work |
|
pWBsacB-LTGSGS34-36AAA |
pWB980 carrying TG gene from S. ladakanum with SGS34-36AAA mutation; Kmr |
This work |
|
pWBsacB-LTGPS74-75AA/ DSD80-82AAA |
pWB980 carrying TG gene from S. ladakanum with PS74-75AA/ DSD80-82AAA mutation; Kmr |
This work |
|
pWBn-LTGPS74-75AA/ DSD80-82AAA |
pWBsacB-LTGPS74-75AA/DSD80-82AAA carrying SPn (15 signal peptides) instead of SPsacB; Kmr |
This work |
Apr, ampicillin resistance; Kmr, kanamycin resistance.
2.2. Construction of TG expression plasmids
The primers used in the study are listed in Table S1. The sequence of TG gene from S. ladakanum was deposited in the GenBank database under accession number AAO48277.1. Two separate TG secretion systems were constructed in B. subtilis. The first one involved constitutive expression of the TG gene encoding the pro-peptide domain and central body of S. ladakanum TG with signal peptide SacB (SPsacB) (Fig. 1A). TG gene was amplified from S. ladakanum genomic DNA by polymerase chain reaction (PCR) using primers TG-F/TG-R. The amplified DNA fragment and the pWB980 expression vector containing SPsacB were each double-digested with XmaI and XbaI and ligated to generate plasmid pWBsacB-LTG .
For the second system, gene encoding the pro-peptide domain of TG was PCR amplified using primers ProTG-F/ProTG-R and the gene encoding the central body was PCR amplified using primers CD-F/CD-R. Each PCR product was paired with a preceding sacB secretory sequence. The sacB gene was PCR amplified from plasmid pWB980 using primers SacB-F/SacB-R. The PCR products of the three genes were ligated by overlap extension PCR using primers ProTG-F/CD-R. The DNA fragment was double-digested with XmaI and NheI and ligated into the sacB signal peptide-containing pWB980 expression vector that had been treated with the same enzymes, generating plasmid pWBsacB-pro-sacB-LTG (Fig. 1B).
Plasmids pWBsacB-LTG and pWBsacB-pro-sacB-LTG were transformed into B. subtilis WB600, respectively. Competent cells of B. subtilis were prepared and transformed as described [14]. The recombinant B. subtilis strains were grown at 37 °C for 12 h in Luria-Bertani (LB) medium with 20 μg/mL Km. The positive clones were selected for colony PCR amplification and DNA sequencing. The molecular weight of the TG was estimated by sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE).
2.3. Site-directed mutagenesis construction
The pro-peptide region folded into an L-shape to cover the active site. So, N-terminal residues played important roles in enzymatic activity. The candidate mutation sites of Ser34_Ser36 and Pro74_Ser75 in the pro-peptide region and mutation sites of Asp80_Glu83 in the junction between pro-peptide and corn domain were selected in this study (Fig. 2). Site-directed mutations were carried out by PCR-based overlap extension mutagenesis [15]. Briefly, the Asp80Ser81Asp82→Ala80Ala81Ala82 substitution (DSD80–82AAA) was completed using primers DSD-F/DSD-R. The deletion of Pro74–Ser75 (△PS) was completed using primers △PS-F/△PS-R. The Pro74Ser75→Ala74Ala75 substitution (PS74–75AA) was completed using primers PS-F/PS-R. The Ser34Gly35Ser36→Ala34Ala35Ala36 substitution (SGS34–36AAA) was completed using primers SGS-F/SGS-R. Plasmid pMD19-T-LTG was used as a template. The fragments obtained by overlap PCR were ligated into vector pMD19-T, yielding plasmid pMD19-T-LTGDSD80-82AAA, pMD19-T-LTG△PS, pMD19-T-LTGSGS34-36AAA, and pMD19-T-LTGPS74-75AA, respectively. The mutations were verified by DNA sequencing. The correct fragments were digested by XmaI/XbaI and inserted into XmaI/XbaI-digested pWB980, yielding pWBsacB-LTGDSD80-82AAA, pWBsacB-LTG△PS, pWBsacB-LTGPS74-75AA, and pWBsacB-LTGSGS34-36AAA, respectively.
Multi-mutation at positions Pro74Ser75 and Asp80Ser81Asp82 (PS74–75AA/DSD80–82AAA) was completed using primers PD-F/PD-R with plasmid pMD19-T-LTGPS74-75AA as template. The mutations were verified by DNA sequencing, and the correct DNA product was transformed into E. coli TOP10 to obtain plasmid pMD19-T-LTGPS74-75AA/DSD80-82AAA. Plasmid pMD19-T-LTGPS74-75AA/DSD80-82AAA were digested by XmaI/XbaI and inserted into XmaI/XbaI-digested pWB980, yielding pWBsacB-LTGPS74-75AA/DSD80-82AAA. The recombinant plasmid was transformed into B. subtilis WB600 as described [14]. The positive clones were selected for colony PCR amplification and DNA sequencing.
2.4. Signal peptide optimization
The construction of plasmids containing different signal peptides (SP) were shown in Fig. S1. Signal peptides used in the study are listed in Table S2. The encoding genes of SPwapA, SPepR, SPnucB, SPyncM, SPyhcR, SPwprA, SPamyE, SPlytD, SPpenP, SPabnA, SPnprB, SPbglS, SPmotB, SPlipB and SPlipA were PCR-amplified from the genome of B. subtilis W168 using primers WapA-F/WapA-R, EpR-F/EpR-R, NucB-F/NucB-R, YncM-F/YncM-R, YhcR-F/YhcR-R, WprA-F/WprA-R, AmyE-F/AmyE-R, LytD-F/LytD-R, PenP-F/PenP-R, AbnA-F/AbnA-R, NprB-F/NprB-R, BglS-F/BglS-R, MotB-F/MotB-R, LipB-F/LipB-R, and LipA-F/LipA-R, respectively. Then, the PCR products were digested and inserted into BsaBI-XmaI-cleaved pWBsacB-LTGPS74-75AA/DSD80-82AAA, yielding pWBn-LTGPS74-75AA/ DSD80-82AAA (n denotes 15 signal peptides).
2.5. Expression of TG in B. subtilis
B. subtilis strains carrying the recombinant plasmids were inoculated into 5 mL LB medium containing 20 μg/mL Km and cultured overnight at 37 °C with shaking at 200 rpm. The seed cultures were inoculated at 2% (v/v) into the fresh LB medium containing 20 μg/mL Km and cultured at 37 °C, 200 rpm. Samples were taken at 48 h, and supernatants were collected following centrifugation at 4000 × g for 10 min. The clarified supernatants were evaluated for enzymatic activity.
2.6. Activity assay of TG
TG activity was measured by a modification method of calorimetric hydroxamate [16]. Enzyme solution (50 μL) was mixed with 50 μg/mL dispase, and incubated at 37 °C for 20 min. Then, the mixture was added with 90 μL of reagent A (0.2 M Tris-HCl (pH 6.0), 30 mM CBZ-Gln-Gly, 100 mM hydroxylamine, and 10 mM glutathione), and incubated at 37°C for 10 min. The reaction was stopped by adding 160 μL of reagent B (a, 3 M HCl; b, 12% TCA; c, 5% ferric chloride–trichloroacetic acid; a:b:c=1:1:1). The supernatant was collected by centrifugation at 4000 × g for 10 min, and the absorbance at 525 nm was measured to determine the TG activity. One unit of microbial TG activity was defined as the amount of enzyme needed to produce 1 μmol of hydroxamic acid per min. Protein concentration was determined using a Bradford protein assay kit (Bio-Rad, 17).
2.7. Optimal pH and temperature
The optimal pH was assayed using the following various buffers (0.2 M): sodium phosphate-citrate buffer (pH 3, pH 4, and pH 5) and Tris-HCl (pH 6, pH 7, pH 8, and pH 9) at 37 °C. The optimal temperature was assayed in Tris-HCl, pH 6 at 20 °C, 30 °C, 40 °C, 50 °C, 60 °C, and 70 °C. The relative activities were expressed as percentages of the activity at the optimal temperature or pH [18]. All tests were repeated at least three times and the data were expressed as mean ± standard deviation (SD). The highest activity was defined as 100%.
2.8. The effects of metal ions on TG activity
Various metal ions (Na+, K+, Ca2+, Mg2+, Cu2+, Zn2+, Mn2+) with a final concentration of 5 mM were added to the enzyme solution. The mixtures were incubated for 30 min on ice prior to performing the enzyme assay. The enzymatic activity with no metal ion was set to 100%. To determine the effect of Ca2+ concentration on the activity of TG, different concentrations of calcium chloride (CaCl2) was added to the enzyme solution (0 mM, 5 mM, 10 mM, 15 mM, 20 mM, and 25 mM). The mixtures were incubated for 30 min on ice prior to performing the enzyme assay [18]. All tests were repeated at least three times and the data were expressed as mean ± SD.
2.9. TG production in a fermentor
High-level production of TG was carried out in a 5-L bioreactor (NBS BioFlo 310, Eppendorf, Shanghai, China) with a working volume of 2 L. The pH was adjusted to 7.0. The dissolved oxygen was related to speed, and it was higher than 15% during fermentation. Seed culture (1%, v/v) was inoculated into fresh LB medium containing 20 μg/mL Km. Samples were collected periodically to determine cell density (OD600). The clarified supernatants were measured for enzymatic activity.
3.1.Synthesis of TG in B. subtilis without its native signal peptide
S. ladakanum is a TG producer . S. ladakanum TG is Ca2+-independent and is stable at pH 5.0~7.0 [19]. Crystal structure showed that Streptomyces TG contained signal peptide, pro-peptide, and central body (Fig. 2A). To produce secreted TG in B. subtilis, B. subtilis WB600, a strain deficient in six extracellular proteases [20], was used as host, and two separate secretion systems were constructed. As shown in Fig. 1A, the original signal peptide in S. ladakanum TG was replaced with the sacB signal peptide in the first strategy. The gene (1172 bp) encoding the pro-peptide domain and central body of S. ladakanum TG was fused with sacB, and the recombinant plasmid was transformed into B. subtilis WB600 (Fig. S1A). A protein band was detected in the supernatants of B. subtilis WB600 carrying plasmid pWBsacB-LTG in SDS-PAGE (Fig. S1B), which suggested that TG was successfully expressed extracellularly. Extracellular enzyme activity was determined to be as high as 1.62 U/mg (Table 2).
Table 2. Relative amounts of TG accumulation in the culture supernatant of B. subtilisa
|
Type |
Feature |
Enzyme activity (U/mg) |
Extracellular protein content (mg/mL) |
Relative amountb (%) |
Region in TG |
|
WTb |
Wild type |
1.62 ± 0.08 |
0.23 ± 0.02 |
100 |
- |
|
DSD80-82AAA |
Asp80Ser81Asp82→Ala substitution |
3.21 ± 0.03 |
0.26 ± 0.05 |
198 |
Conjunction |
|
△PS |
Pro74 to Ser75 deletion |
3.65 ± 0.02 |
0.27 ± 0.03 |
225 |
Pro-peptide |
|
PS74-75AA |
Pro74 Ser75→Ala substitution |
4.32 ± 0.04 |
0.25 ± 0.01 |
267 |
Pro-peptide |
|
SGS34-36AAA |
Ser34 Gly35Ser36→Ala substitution |
1.57 ± 0.04 |
0.24 ± 0.03 |
97 |
Pro-peptide |
|
PS74-75AA/ DSD80-82AAA |
Substitutions of Pro74 Ser75→Ala and Asp80Ser81Asp82→Ala |
5.84 ± 0.06 |
0.25 ± 0.04 |
361 |
Multi-mutations |
a Results are the average of duplicates.
bThe enzyme without mutation sites was named as WT.
cThe enzyme activity of mutation (U/mg) /that of mild type. The enzymatic activity produced by wild type was set as 100%.
Because the order of expression of the pro-peptide and central body plays an important role in the reconstitution of TG and a successful example has been reported in E. coli [8], in the second strategy we paired each part of the pro-peptide region and central body region with a preceding sacB signal peptide (Fig. 1B). However, this expression system failed to produce active TG. No protein band was detectable by SDS-PAGE nor was any enzymatic activity detected in the cell supernatants (data not shown). The reason maybe that pro-peptide functions in an intermolecular manner to mediate TG folding. For the first strategy, pro-peptide and core domain secreted simultaneously. However, for the second strategy, pro-peptide and core domain were secreted outside cells separately. Wrong folding may occur, which resulted in no enzymatic activity detection. Based on our findings, the first expression system encoding the sacB signal peptide, pro-peptide, and central body was used in the subsequent studies. These findings also indicated that the sacB signal peptide could direct TG to be secreted into the periplasm.
Currently, TG has been identified from various microorganisms, and has been heterologously expressed in different strains. However, only Streptomyces TG has commercial value [10]. Microbial TG has received increasing interest for food treatment and has been shown to improve food flavor, appearance and texture [21]. The complexity of the current procedures prompted scientists to develop an efficient and easy-to-use system for the “green” production of TG. It has been reported that in Streptomyces sp., TG is secreted as an inactive zymogen in cultures and is activated during culture [22]. Obtaining soluble TG is a key problem for the recombinant expression. Although Streptomyces TG has been successfully expressed in E. coli by sequence expressing pro-peptide and central body [8], the pre-expression of pro-peptide does not result in soluble TG expression in this study. The reason may be the wrong folding of soluble TG [22]. B. subtilis is classified as a GRAS organism and is used as a secretion host for large amounts of commercial enzymes. The secretion of recombinant proteins into the culture medium greatly simplified downstream processing [23, 24]. In our previous study, active S. mobaraense TG was secreted by B. subtilis through the introduction of VMM into zymogen [14]. S. mobaraense TG was also secreted by B. subtilis through the Tat pathway [13]. These studies indicated that B. subtilis was a promising host for TG production.
The signal peptide was responsible for TG transport across the cytoplasmic membrane. However, it has been reported that heterologous Streptomyces TG using E. coli as a host was expressed as inclusion bodies with its native signal peptide [25]. In B. subtilis, the Sec pathway is the major route of protein transport [23]. To produce extracellular S. ladakanum TG in B. subtilis, the sacB signal peptide (SPsacB) was used to replace its native signal peptide (Fig. 1). With the direction of SPsacB, TG was released into extracellular medium of B. subtilis. A single band was detected in SDS-PAGE (Fig. S1).
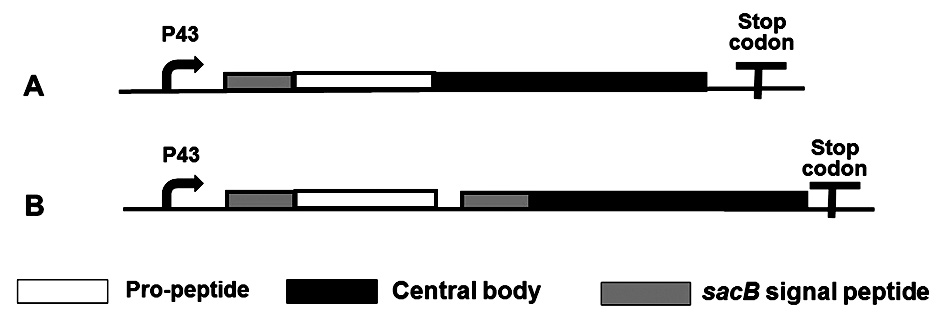
Fig. 1. Genetic organization for the construction of two separate TG secretion systems. Construction of plasmid pWBsacB-LTG (A). Construction of plasmid pWBsacB-pro-sacB-LTG (B).
3.2. Involvement of the N-terminal region of TG in increasing enzyme activity
To further increase the enzyme activity in this study, site-directed mutagenesis was used. S. mobarensis TG was used as model since it is currently the type of TG used commercially and its structure has been extensively investigated [26]. Candidate mutation sites were selected based on sequence homology and structural analysis of S. mobaraensis TG (PDB: 3IU0), an enzyme highly homologous to S. ladakanum TG sharing 88% identity at the amino acid level. Studies have shown that the pro-peptide in the N-terminal region of S. mobarensis TG are the most important for achieving active and soluble enzyme [27]. It folded into an L-shape and covered the active-site cleft in a complementary manner [26]. It is assumed to act as an intermolecular chaperone to stimulate TG folding, enzymatic activity, or secretion [16]. Therefore, the site-mutations in the current study were all within the pro-peptide region (Ser34–Ser36 and Pro74Ser75) and at the junction between the pro-peptide and central body (Asp80–Asp82 and Glu83) (Fig. 2B). Ala has propensity to form alpha helices but can also occur in beta sheets. It is generally an accepted single residue first choice for mutations because it may not cause changes in polar and differently charged. So, it was chosen for site-directed mutations in this study. The results in Table 2 demonstrated that the changes in the N-terminal residues indeed affect the activity of TG. Although the contents of extracellular proteins of different mutations maintained at 0.24~0.27 mg/mL, there was a difference in enzymatic activities of mutations. The mutation SGS34–36AAA in the N-terminal of the pro-peptide showed little decrease in enzymatic activity in the supernatant with 97% of the activity remaining. However, the mutation DSD80–82AAA at the junction of the pro-peptide and central body resulted increase in enzymatic activity with 98% higher than that of the wild type enzyme. Similarly, the Pro74–Ser75 deletion (△PS) showed improved activity with a 125% increase compared to that of the wild type. In the same way, the pro-peptide mutant PS74–75AA also displayed improved TG activity. Multi-mutations at positions Pro74, Ser75, and Asp80Ser81Asp82 (PS74–75AA/DSD80–82AAA) resulted in the highest enzymatic activity of 5.84 U/mg. This was 261% higher than that of the wild type enzyme. The B. subtilis strain transformed with the gene encoding the mutated TGasePS74–75AA/DSD80–82AAA was used in further study, and its enzymatic characteristics were systematically evaluated to explore its potential utilization.
The deletion or substitution at positions Asp80–Asp82 and Pro74Ser75 resulted in good performance in our study. Enzymatic activity was efficiently improved, especially in the case of multi-mutations, which exhibited activity levels 361% compared to that of the wild type enzyme (Table 2). The results obtained in this study provide clear evidence that mutations in the pro-domain affect TG activity. Several proteases, including subtilisins and TG, require the N-terminal pro-peptide to produce mature enzymes [28]. Although pro-peptide is essential only during the late stages of the folding process, a mutated pro-peptide may result in an altered conformation of protease and affect its bioactivity. This phenomenon is known as “protein memory” [29]. Therefore, mutations in the pro-peptide region may make changes in the structure of the mature TG resulting in high enzymatic activities being obtained in these mutants. In the current study, substitutions in Asp80–Asp82 (DSD80–82AAA), which were located at the junction between the pro-peptide and central body, may have affected the association of the central TG region with the pro-peptide resulting in the enhancement of the enzyme activity. Our results clearly demonstrated that engineering of the pro-peptide region is an efficient method for modifying enzymatic characteristics.
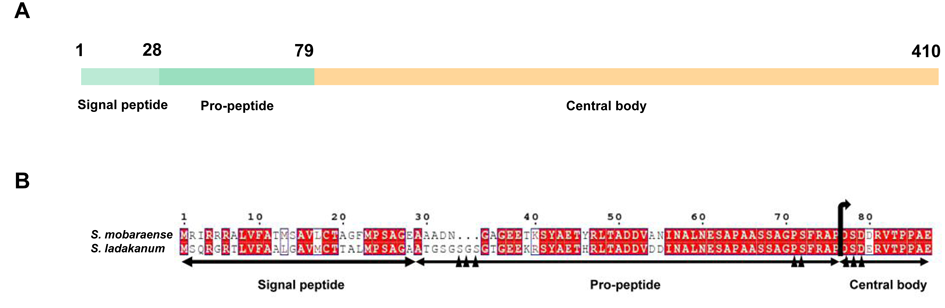
Fig. 2. Schematic representation of S. ladakanum TG (A). Homology of the N-terminal region of TG from S. ladakanum and S. mobaraense (B). Triangle indicates the mutation sites.
3.3. Effects of signal peptide on TG activity in B. subtilis
In order to improve the production of extracellular TG in B. subtilis, 16 signal peptide from B. subtilis 168, including SPsacB, SPwapA, SPepR, SPnucB, SPyncM, SPyhcR, SPwprA, SPamyE, SPlytD, SPpenP, SPabnA, SPnprB, SPbglS, SPmotB, SPlipB and SPlipA, were used to replace the original signal peptide of S. ladakanum TG. The genes encoding the pro-peptide and core domain of S. mobaraens TG were fused with SPn (n denotes 16 signal peptides) into plasmid pWB980, under the control of the constitutive promoter P43 (Fig. S1). As shown in Fig. 3, different signal peptides showed different secretion efficiency. SPSacB and SPAbnA showed better abilities to direct the secretion of TG into the medium than other SPs. The enzymatic activities in the supernatants of strains with SPwapA, SPepR, SPnucB, SPwprA, SPamyE, SPlytD , SPpenP, SPmotB, SPlipB and SPlipA were only 0.8% to 2% compared to those with SPSacB and SPAbnA. Our results indicated that SPwapA, SPepR, SPnucB, SPwprA, SPamyE, SPlytD , SPpenP, SPmotB, SPlipB and SPlipA could not complete the secretion of TG in B. subtilis. Although SPyncM, SPyhcR, SPnprB, and SPbglS could secrete TG outside the cell, the TG activity of the culture supernatant with SPyncM, SPyhcR, SPnprB, and SPbglS were only approximately 30% compared to those with SPSacB. Although SPSacB and SPAbnA showed better abilities than other SPs. Considering SPsacB is a commonly used levansucrase SP in B. subtilis [14], it was selected for further study. After shake-flask fermentation for 48 h, the TG activity of the culture supernatant was 5.98 U/mg .
In general, there is a hydrophobic signal peptide (SP) at the N-terminal of the secreted protein, which leads the protein to be secreted into the cell periplasm. SP is composed of a positively charged N-terminal domain, a long hydrophobic H-domain and a C-terminal domain, which is the signal peptidase recognition site [30]. The optimal match between SP and the target protein is very important. It is critical to the optimization of SP for the heterologous protein. S. mobaraensis TG was expressed in B. subtilis using SPwapA and SPamyQ [13]. Our study demonstrated that for S. ladakanum TG, the SPsacB and SPabnA were more suitable than SPwapA (Fig. 3). Furthermore, SPamyQ was also been detected for TG secretion and our results showed that SPsacB and SPabnA were better than SPamyQ (data not shown).
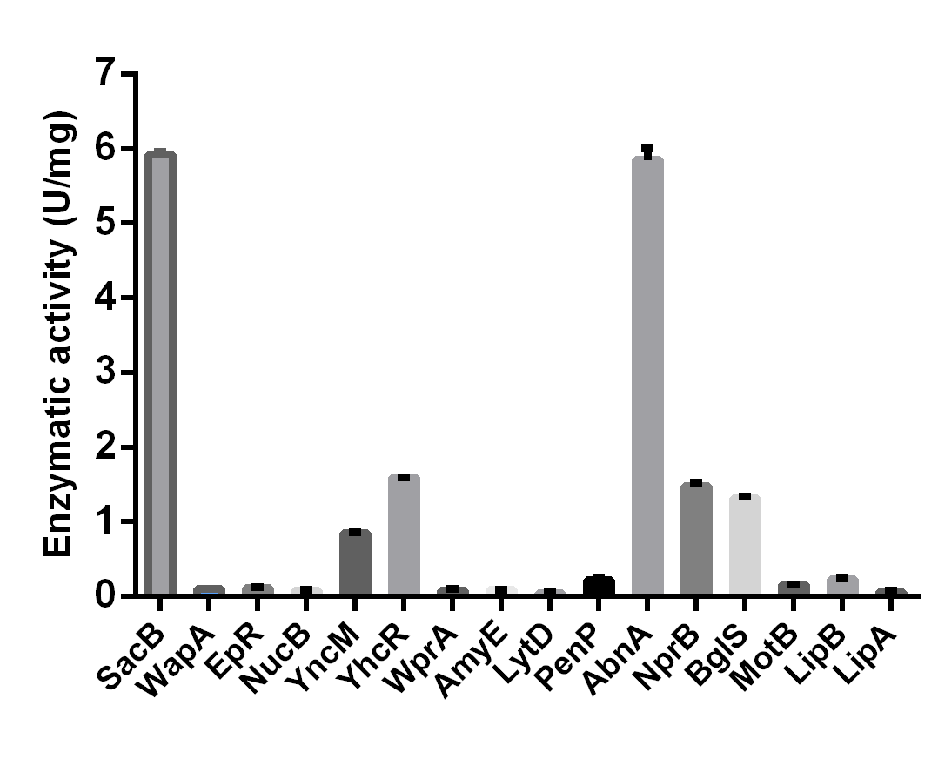
Fig. 3. Effects of signal peptides on TG secretion.Error bars represent SD calculated from three independent determinations.
3.4. Enzymatic characterization of TG expressed in B. subtilis
The extracellular TG expressed in B. subtilis exhibited optimum activity at pH 6.0. The activity gradually decreased at pH 9.0 and rapidly decreased at pH 4.0, indicating that this enzyme was not tolerant under acidic conditions. The extracellular TG expressed in B. subtilis was relatively stable at pH 6.0–8.0 (Fig. 4A). It exhibited high activity at 50 °C. The enzyme activity decreased rapidly when temperature was higher than 50 °C. Only approximately 30% activity remained when temperature was increased to 60 °C (Fig. 4B). Ca2+, Na+ and Mg2+ had little influence on the activity of TG. The presence of K+ and Mn2+ could maintain the activity of TG at approximately 90%, but Cu2+ and Zn2+ significantly inhibited TG activity, especially Zn2+. The relative activity was only 9.8% (Fig. 4C). The TG expressed in B. subtilis required no Ca2+ for activity. It was highly stable in the presence of Ca2+ and exhibited 113% activity in 25 mM Ca2+ (Fig. 4D).
Similar to that of the wild-type Streptomyces TG, S. ladakanum TG expressed in B. subtilis is Ca2+-independent, and its activation requires no special cofactors, which is highly beneficial in industrial applications. For instance, TG could be used to modify some Ca2+ sensitive proteins, such as soybean globulins. Because these proteins are easily precipitated by Ca2+, Ca2+ cannot be used in this food [1, 31]. The enzyme obtained in our study exhibited stable activity in the absence of Ca2+ and therefore can be used to modify proteins that are sensitive to Ca2+. Furthermore, the presence of Ca2+ did not inhibit the activity of S. ladakanum TG expressed in B. subtilis (Fig. 4), suggesting S. ladakanum TG expressed in B. subtilis may also be used to modify proteins containing Ca2+. Taken together, it appears that S. ladakanum TG expressed in B. subtilis is likely to have a broad range of applications.
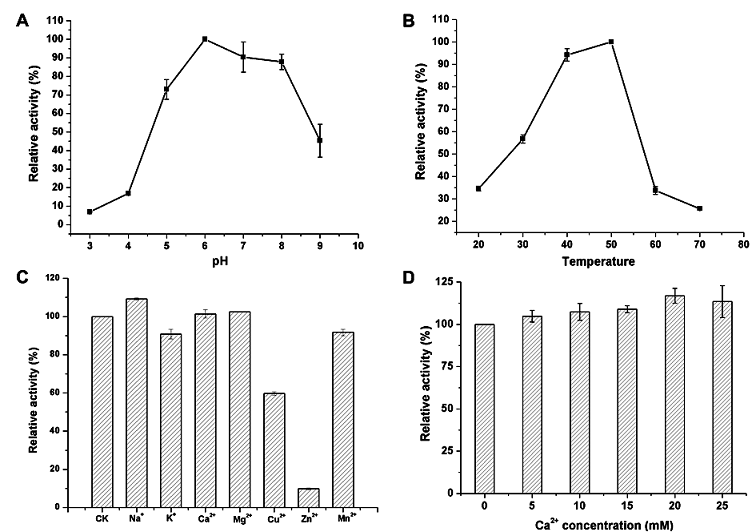
Fig. 4. Analysis of enzymatic characteristics. Optical pH of TG expressed in B. subtilis (A). Optical temperature of TG expressed in B. subtilis (B). Effects of different metal ions on enzyme activity (C). Effects of Ca2+ on enzyme activity (D). The highest activity was defined as 100%. Error bars represent SD calculated from three independent determinations.
3.5. Production of extracellular TG in B. subtilis
The B. subtilis transformant containing the optimal gene encoding TGPS74–75AA/DSD80–82AAA with SPsacB was evaluated for small-scale TG production in a 5-L fermentor. As shown in Fig. 5, during the first 16 h, B. subtilis was in logarithmic growth stage. The OD600 value increased significantly and reached 64 at 21 h. Extracellular activity of TG in the culture broth steadily increased in a linear fashion during the first 48 h and reached a maximum of 7.60 U/mg at 48 h. Interestingly, the TG expressed in B. subtilis demonstrated good stability during fermentation compared with that of the Streptomyces TG. The maximum enzymatic activity at 48 h was set as 100% and it retained a large percentage of its initial activity (60%) at 60 h. Importantly, the 60% activity remained with increased incubation time (data not shown).
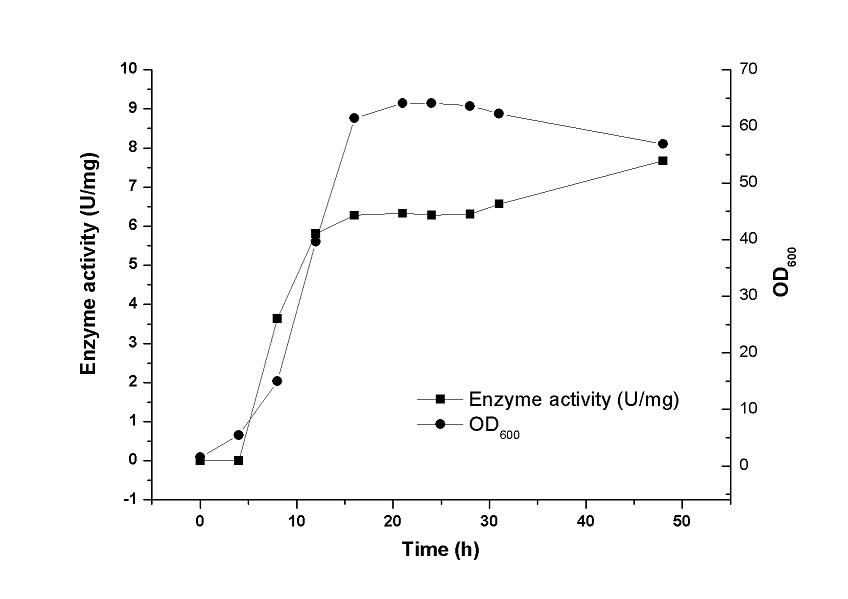
Fig. 5. TG production in B. subtilis. Error bars represent SD calculated from three independent determinations.
This study presents the secretion of S. ladakanum TG in B. subtilis. After replacing the native signal peptide with SPsacB and site-directed mutagenesis, an activity of 7.60 U/mg was obtained in the supernatant of culture at 48 h. The enzyme from our current study is Ca2+-independent and is stable at a wide range of pH, which are comparable to that of currently used commercial TG. This system could secret TG into the culture medium, which simplified downstream processing and may be useful for producing easily purified TG with widespread application.
Supplementary Files
https://www.siftdesk.org/articles/images/10678/supplementary-files.pdf
This work was supported by grants from the Key Laboratory of Agro-Products Processing,Ministry of Agriculture and Rural Affairs/ Institute of Food Science and Technology, Chinese Academy of Agricultural Sciences, and the National Natural Science Foundation of China (31670045).
Competing interests
The author hereby declares that there were no competing interests in this research.
Authors contributions
LF and LW performed the experiment. JJ, HE, SZ, BY and JL performed data analysis. LW wrote the paper. All authors read and approved the final manuscript.
Yokoyama K, Nio N, Kikuchi Y. Properties and applications of microbial transglutaminase. Appl Microbiol Biotechnol 2004; 64: 447-54. PMid:14740191
View Article PubMed/NCBIZhu Y, Tramper J. Novel applications for microbial transglutaminase beyond food processing. Trends in Biotech 2008; 26: 559-565. PMid:18706723
View Article PubMed/NCBIPasternack R, Dorsch S, Otterbach JT, Robenek IR, Wolf S, Fuchsbauer HL. Bacterial pro-transglutaminase from Streptoverticillium mobaraense: purification, characterisation and sequence of the zymogen. Febs Journal 1998; 257: 570-576. PMid:9839945
View Article PubMed/NCBIYasueda H, Nakanishi K, Kumazawa Y, Nagase K, Motoki M, Matsui H. Tissue-type transglutaminase from red sea bream (Pagrus major). sequence analysis of the cDNA and functional expression in Escherichia coli. Eur J Biochem 1995; 232: 411-419. PMid:7556189
View Article PubMed/NCBIDate M, Yokoyama K, Umezawa Y, Matsui H, Kikuchi Y. Production of native-type Streptoverticillium mobaraense transglutaminase in Corynebacterium glutamicum. Appl Environ Microbiol 2003; 69: 3011-3014. PMid:12732581
View Article PubMed/NCBIWang LM, Yu B, Wang RX, Xie JC. Biotechnological routes for transglutaminase production: recent achievements, perspectives and limits. Trends Food Sci Technol 2018; 81: 116-120.
View ArticleAna L, Camolezi G, Goes-Favoni de, Pedroso S. Action of microbial transglutaminase (MTGase) in the modification of food proteins: A review. Food Chem 2015; 171: 315-322. PMid:25308675
View Article PubMed/NCBILiu S, Zhang D, Wang M, Cui W, Chen K, Du G, Chen J, Zhou Z. The order of expression is a key factor in the production of active transglutaminase in Escherichia coli by co-expression with its pro-peptide. Microb Cell Fact 2011; 10: 112-119. PMid:22196373
View Article PubMed/NCBINoda S, Miyazaki T, Tanaka T, Chiaki O, Kondo A. High-level production of mature active-form Streptomyces mobaraensis transglutaminase via pro-transglutaminase processing using Streptomyces lividans as a host. Biochem Eng J 2013; 74: 76-80.
View ArticleChristian S, Norbert V, Markus P. Model based optimization of the fed-batch production of a highly active transglutaminase variant in Escherichia coli. Protein Expr Purif 2011; 77: 9-19. PMid:21168505
View Article PubMed/NCBILiu Y, Li J, Du G, Chen J, Liu L. Metabolic engineering of Bacillus subtilis fueled by systems biology: Recent advances and future directions. Biotechnol Adv 2017; 35: 20-30. PMid:27867004
View Article PubMed/NCBILiu Y, Lin S, Zhang X, Liu X, Wang J, Lu F. A novel approach for improving the yield of Bacillus subtilis transglutaminase in heterologous strains. J Ind Microbiol Biot 2014; 41: 1227-1235. PMid:24947581
View Article PubMed/NCBIMu D, Lu J, Qiao M, Kuipers O, Zhu J, Li X, Yang P, Zhao Y, Luo S, Wu X, Jiang S, Zheng Z. Heterologous signal peptides-directing secretion of Streptomyces mobaraensis transglutaminase by Bacillus subtilis. Appl Microbiol Biotechnol 2018; 102: 5533-5543. PMid:29691630
View Article PubMed/NCBIFu L, Wang Y, Ju J, Cheng L, Xu Y, Yu B, Wang L. Extracellular production of active-form Streptomyces mobaraensis transglutaminase in Bacillus subtilis. Appl Microbiol Biotechnol 2020; 104: 623-631. PMid:31797004
View Article PubMed/NCBIKobayashi H, Tateishi A, Tsuge H, Takahashi E, Okamoto K, Yamanaka H. The carboxy-terminal tail of Aeromonas sobria serine protease is associated with the chaperone. Microbiol Immunol 2009; 53: 647-657. PMid:19954452
View Article PubMed/NCBIYurimoto H, Yamane M, Kikuchi Y, Matsui H, Kato N, Sakai Y. The pro-peptide of Streptomyces mobaraensis transglutaminase functions in cis and in trans to mediate efficient secretion of active enzyme from methylotrophic yeasts. Biosci Biotechnol Biochem 2004; 68: 2058-2069. PMid:15502350
View Article PubMed/NCBIBradford MM. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 1976; 72: 248-254. 90527-3
View ArticleSuzuki S, Izawa Y, Kobayashi K, Eto Y, Yamanaka S, Kubota K, Yokozeki K. Purification and characterization of novel transglutaminase from Bacillus subtilis spores. Biosci Biotech Bioch 2000; 64: 2344-2351. PMid:11193401
View Article PubMed/NCBILin Y, Chao M, Liu C, Chu W. Cloning and expression of the transglutaminase gene from Streptoverticillium ladakanum in Streptomyces lividans. Process Biochem 2004; 39: 591-598. 00134-1
View ArticleWu XC, Lee W, Tran L, Wong SL. Engineering a Bacillus subtilis expression-secretion system with a strain deficient in six extracellular proteases. J Bacteriol 1991; 173: 4952-4958. PMid:1907264
View Article PubMed/NCBIMotoki M, Seguro K. Transglutaminase and its use for food processing. Trends Food Sci Tech 1998; 9: 204-210. 00038-7
View ArticleZotzel J. Transglutaminase from Streptomyces mobaraensis is activated by an endogenous metalloprotease. Eur J Biochem 2003; 270; 3214-3222. PMid:12869197
View Article PubMed/NCBIChen J, Fu G, Gai Y, Zheng P, Zhang D, Wen J. Combinatorial Sec pathway analysis for improved heterologous protein secretion in Bacillus subtilis: identification of bottlenecks by systematic gene overexpression. Microb Cell Fact 2015; 14: 92. PMid:26112883
View Article PubMed/NCBILiu X, Wang H, Wang B, Pan L. Efficient production of extracellular pullulanase in Bacillus subtilis ATCC6051 using the host strain construction and promoter optimization expression system. Microb Cell Fact 2018; 17: 163. PMid:30348150
View Article PubMed/NCBITakehana S, Washizu K, Ando K, Koikeda S, Takeuchi K, Matsui H, Motoki M, Takagi H. Chemical synthesis of the gene for microbial transglutaminase from Streptoverticillium and its expression in Escherichia coli. Biosci Biotechnol Biochem 1994; 58: 88-92. PMid:7765335
View Article PubMed/NCBIYang MT, Chang CH, Wang JM, Wu TK, Wang YK, Chang CY, Li TT. Crystal structure and inhibition studies of transglutaminase from Streptomyces mobaraense. J Biol Chem 2011; 286: 7301-7307. PMid:21193394
View Article PubMed/NCBIRickert M, Strop P, Lui V, Melton-Witt J, Farias SE, Foletti D, Shelton D, Pons J, Rajpal A. Production of soluble and active microbial transglutaminase in Escherichia coli for site-specific antibody drug conjugation. Protein Sci 2015; 25: 442-455. PMid:26481561
View Article PubMed/NCBITakagi H, Takahashi M. A new approach for alteration of protease functions: pro-sequence engineering. Appl Microbiol Biotech 2003; 63: 1-9. PMid:12879301
View Article PubMed/NCBIShinde U, Liu J, Inouye M. Protein memory through altered folding mediated by intramolecular chaperones. Nature, 1997; 389: 520-522. PMid:9333245
View Article PubMed/NCBIDegering C, Eggert T, Puls M, Bongaerts J, Evers S, Maurer KH, Jaeger KE. Optimization of protease secretion in Bacillus subtilis and Bacillus licheniformis by screening of homologous and heterologous signal peptides. Appl Environ Microbiol 2010; 76: 6370-6376. PMid:20709850
View Article PubMed/NCBILovaine D, Carla R, Cristiano V, Marco A. Review transglutaminases: part II-industrial applications in food, biotechnology, textiles and leather products. World J Microbiol Biotechnol 2019; 36: 11. PMid:31879822
View Article PubMed/NCBI