Ikpesu, T.O
Email: tomohwofasa@yahoo.com
© 2019 Sift Desk Journals. All Rights Reserved
VOLUME: 2 ISSUE: 1
Page No: 41-46
Ikpesu, T.O
Email: tomohwofasa@yahoo.com
1*Ikpesu, T.O and 2Opara C.N
1Department of Biology, Federal University Otuoke, Nigeria
2Department of Microbiology, Federal University Otuoke, Nigeria
Apinun Suvarnaraksha(apinun@mju.ac.th)
Ikpesu, Thomas Ohwofasa, Ecological and Biological Effects of Pollution and Dredging Activities on Fisheries and Fisheries Products in Niger Delta Ecological Zone(2017)SDRP Journal of Aquaculture, Fisheries & Fish Science 2(1)
The effects of anthropogenic activities on fisheries and fisheries products in Niger Delta water were investigated. Two major Rivers in the region were investigated and three stations were chosen per river depicting downstream and upstream stations and were sampled on monthly basis. The down streams stations are the polluted and heavily dredged sites, where the upstream station has no evidence of pollution. During these periods, the fishes of the same species were collected and analyzed for morphological and physiological changes, after which they were returned back to the rivers. Morphological changes such as skin ulcerations and other lesions, as well as fungi infections were observed in the down streams fishes. The fish in up streams look healthier than the downstream fishes. The physico-chemical parameters between the up streams and down streams stations vary significantly (p < 0.01). The release of pollutants into the water in the Niger Delta areas may triggers off naturally occurring bio toxicity cycles and other fish poisoning.
Key words: Fisheries; rivers, Niger Delta; anthropogenic; dredging; pollution
Growing human population densities in coastal communities have manifested a demonstrably adverse effect on aquatic resources. The scientific literature is replete with evidence of inorganic and organic pollutant accumulation in coastal waters from anthropogenic effluents [1,2,3,4,5]. These anthropogenic may alter natural processes and natural resource communities; unabated degradation of the aquatic environment caused by a wide spectrum of human activities poses consequences for fishery resources and their habitats.
Pollutant exposure can alter basic detoxification mechanisms, susceptibility to diseases, affect the ability of individuals or populations to counteract pollutant-induced metabolic stress; altering reproductive processes including gamete development and embryonic viability; deter normal processes including feeding rate, respiration, osmoregulation; and overall Darwinian fitness [6].
Sewage such as house hold sewage, human sewage and industrial sewage that is untreated causes water pollution in the same way as fertilizers do. Human sewage contains germs that cause diseases such as hepatitis and cholera. Soaps and washing detergents contain both natural and manmade chemicals. The natural chemicals can cause a pollution problem similar to that caused by fertilizers.
Like sewage outfalls, dredging activities are point sources for a variety of aquatic contaminants, particularly metals and other trace elements. These substances tend to adhere to solid particles within the water, become adsorbed onto finer sediment fractions once dispersed into coastal waters, and subsequently accumulate in depositional areas. Together with microbial action, local salinity and other properties of the riverine, estuarine, or marine receiving waters may alter the chemistry of these contaminant-particle complexes in ways that render them more toxic than their parent compounds. Upon entering the food web, such contaminants tend to accumulate in benthic organisms at higher concentrations than in surrounding waters [7] and may result in various physiological, biochemical, or behavioral effects [6,8]. Suction dredging most certainly can impact the reproductive success of fish. Fish, including salmon, steelhead, sculpin, minnows, suckers, lamprey and trout utilize small gravel substrates for spawning. Dredging can reduce substrate embeddedness (the degree to which gravel, cobble, boulders and snags are covered or sunken into the silt, sand or mud of the stream bottom) and therefore impact spawning habitat.
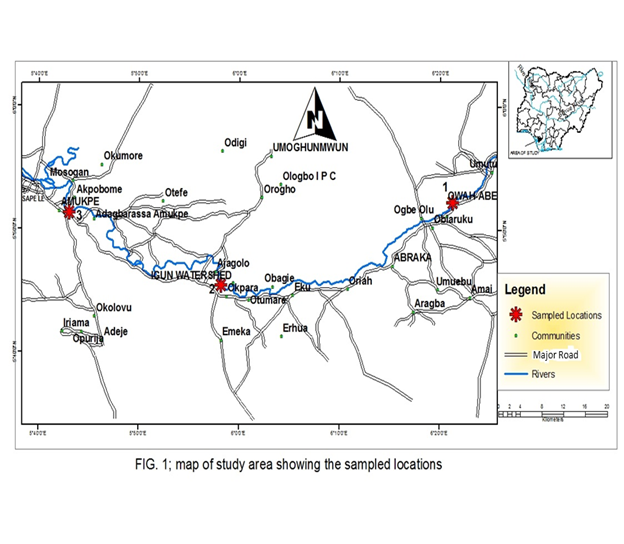
The Ethiope River takes its origin from around Umuaja and flows through zones of freshwater swamps, mangrove swamps, and coastal sand ridges. It is a relatively large water body which stretches within latitudes 6°30’-6°00’ N and longitude 8°24’-5°21’E, covering a surface area of 204 sq km, with a length of about 142km .It is one of the major River in the Niger Delta, Nigeria. It drains various tributaries and empties its content to Benin River that in turn empties into the Atlantic Ocean (Figure 1).
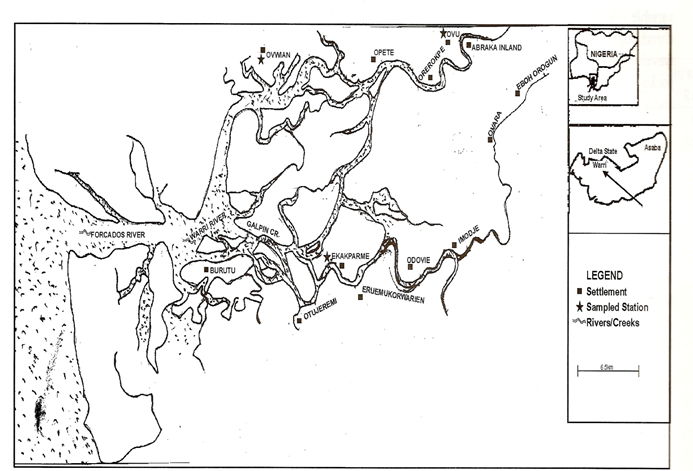
Warri River is a major navigable channel of the Niger Delta, southern Nigeria (Figure 2). It takes its origin from around Utagba Uno and flows through zones of freshwater swamps, mangrove swamps, and coastal sand ridges. The river is a relatively large water body which stretches within latitudes 5o 211 - 6o 001N and longitude 5 o 241 – 6o 211 E, covering a surface area of about 255 sq. km with a length of about 150km [9] .It drains various tributaries and empties into the brackish Forcados River that in turn empties into the Atlantic Ocean
The two Rivers are stress by anthropogenic activities and their qualities are influenced majorly by urban centers, commercial activities such as transportation and dredging. Biodiversity of these Rivers is remarkably high, with a large diversity of fish, mollusks, shellfishes, insects, amphibians, reptiles and birds.
The studied rivers; Warri River and Ethiope River with stations established within the rivers. Three stations were chosen in each rivers depicting downstream and upstream stations .The downstream stations were subjected to human activities (dredging and dumping sites), while the upstream stations were far from human activities and close to the source of the rivers. Other factors considered is the availability of aquatic organisms to be investigated
Ethiope River
Stations were established at Amukpe, Igun water shed and Owah abbey. Amukpe, Igun water shed stations were the downstream stations and Owah abbey served as control
Warri River
Stations were established at Ovwian, Ekakpamre and Ovu. Ovwian and Ekapamre stations were the downstream stations while Ovu served as control.
Sampling Schedule and Analysis
Only dominant aquatic fauna and limnological parameters were sampled. The same fauna were sampled in both rivers. The sampled organisms; Aporeca longa, crabs, Lymnea auricularia Chrysichthys furcatus, Clarias gariepinus, Tilapia zilli, and Clarotes laticeps. Sampling was on monthly basis from January, 2013 to December, 2013. Samples were replicated and subjected to appropriate statistical analysis. Total hardness was measured by the titration method [10]. Dissolved oxygen concentration was measured by the Winkler method. Water temperature and pH were determined with a glass electrode (Thermo Orion, Beverly, Massachusetts, USA). Faecal coliforms were determined by 5-tube MPN technique using selective media.
Other such as turbidity, total dissolve solid, biochemical oxygen demand, nitrate, and phosphate of each of the sampling stations were measured using appropriate technique.
The findings showed that sewage disposal dredging activities had a significant effect on the water quality in the studied rivers. In addition to the direct impacts, there are deleterious impacts that cause temporary or permanent flow diversions and impoundments and the transport of aquatic invasive species from one point to another [11, 12]. There were normalization in physicochemical parameters in away from the sewer points and dredging sites. Similar observation was reported by [13] in seawater close to the sewage outfall with rapid normalization in levels with increasing distance from the outfall in Antarctica Kerguelen Island.
Water quality parameters: The range of the concentrations of the limnological parameters at three stations each along the Ethiope Rivers and Warri River , sampled monthly from January to August 2014 is shown in Table 1 and 2. The samples varied significantly between various stations and between seasons (p < 0.05).
The temperature ranges observed in the polluted and dredged stations was higher than the temperature trend in water bodies in the tropical forest areas [14] .The recommended limit of dissolved oxygen (DO) for fish is 5mg/l [15], but conversely,[16] recommended a range of 2mg/l - 7mg/l, which fall within the range observed in all the stations including the reference stations. The pH obtained from this study was higher than the acceptable pH range for drinking water which is between 7.0 - 8.3 [17], 6.5 - 8.5 [14] .6.0 - 7.0 [17]. [18] stipulated a DO range of 5.5 - 10 for tropical fishes. Total alkalinity showed a positive index of potential productivity for the waters at 31.93 - 35.90 for fresh water species [14] .
Total coliforms have been used as indicators for many years in evaluating water quality for several water uses with respect to domestic waste [19]. The microbiological criterion for shellfish for human consumption has been accepted internationally to be 70 total coliforms per 100 ml using MPN, with no more than 10% of the values exceeding 230 MPN/100 ml. The UNEP/WHO standards for shellfish harvesting waters are at the maximum of 10 feacal coliforms /100 ml for 80% of samples taken and no feacal coliforms detected for drinking water [20]. Other parameters and the recommendation range by the appropriate authorities are summarized in the tables 1 and 2.
Morphological Changes
Biodiversity was seriously altered in the downstream stations, less fishes and other invertebrates were caught. The investigated fishes C. furcatus, Clarias gariepinus, T. zilli, and Clarotes laticeps were very scanty in downstream stations. Though, there were other species in reference stations, not present in down streams. These were chosen because they were found in all the stations. The few fishes caught in the downstream had as skin ulcerations and different lesions, as well as fungi infections. The magnitudes of alterations depend on the close proximity to pollution sites. Ovwian and Amukpe stations were the worst affected, which could be attributed to combining effects of dredging, sewage and anchor location for canoes and boats. Beside these stations were within the cities metropolis, where other human effects which could not be accounted, can detriment the life aquatic inhabitants.
The earthworms found in these stations also showed disparity. The downstream stations were darker, slimmer and found on soil surface instead in the soil as observed in the upstream stations. This could be as a result of the penetration of toxic that emanated from the toxic from the sewages and the dredgers (diesel, lubricant and metals), leading to the poor soil aeration and discomfort of the annelids. The upstream annelids were similar, healthy and no conspicuous colour alteration. Crabs and the periwinkles showed multiple colours in the down streams stations and were not as large as the up streams.
Sewage discharge had significant effect on the water quality in terms of feacal coliform indicator bacteria levels. This effect was well spread throughout the study area. People who bathe or surf in the water can fall ill if they swallow polluted water. Similarly, sewage can poison aquatic organisms that grow near the shore. People who eat poisoned fish, snail and crabs have risk suffering from an acute and sometimes fatal illness. Similarly, dredging without necessary precaution is devastating. Destruction of the shoreline and alter biodiversity. Many people that involve in this act only do so to enrich themselves without considering the environmental impacts.
In order to have a balance environment, all hands must be on deck, though not handy, hence to contain these anomalies, there should be stringent environmental laws that will make it tougher for people to pollute. Similarly, there should be introduction of polluter pays principle. This means that whoever causes pollution should have to pay to clean it up, one way or another. Life is ultimately about choices and so is pollution. We can work together to keep the environment clean so the plants, animals, and people who depend on it remain healthy. We can take individual action to help reduce water pollution, for example, by disposing sewage appropriately, using environmentally friendly detergents not pouring oil down drains, reducing pesticides etc. We can take community action too, by picking litter to keep our rivers and seas little bit cleaner. And we can take action as countries and continents to pass laws that will make pollution harder and the world less polluted. Working together, we can make pollution less of a problem and the world a better place.
Because virtually nothing is known about the biological effects of pollutants in the Niger Delta ecological zone, and because of the elevated current public attention for the water quality effects in the river, it is urgent that more scientific data are collected on the health of the aquatic ecosystem in the rivers in this region. We propose to initiate a study in which aquatic animals will be collected from these rivers and then analyze for health status and pollution effects. Because effects in animals that are in the top tiers of the food web are often good indicators for ecosystem health. Thus, the overall goal for this project is to investigate exposure and effects of anthropogenic pollutants on fish in the Niger Delta Rivers. We will achieve this goal by collecting different fish’s species from a variety of sites along the rivers, and by measuring a suite of health parameters, among which: that include somatic indices, tissue histology, blood and bile analysis and expression of detoxifying enzymes and proteins in liver. An important part of the project is to get students from high school to graduate school levels involved in the project and also to actively relay the obtained information to local conservation interest groups.
|
Parameters |
Amukpe |
Igun Water shed |
Owah abbey |
DPR/FMEnv Recommendation |
|
Water Temperature (oC) |
34.40 -36.70 |
25.30 – 39.40 |
28.40-30.00 |
30 |
|
pH |
3.60 -4.80 |
5.20 - 6.00 |
6.20-8.20 |
6.5-9.2 |
|
Turbidity |
1.20 - 6.30 |
2.40 - 6.10 |
5.20-8.00 |
10 |
|
Total dissolve solid (mg/L) |
1790 -2116 |
1340 - 2009 |
1290- 2080 |
2000 (USEPA; 100- 1000) |
|
Dissolve Oxygen(mg/L) |
3 -5 |
2- 4 |
5 – 7 |
5 |
|
Chemical Oxygen Demand (COD) |
40 -47 |
40- 53 |
26-45 |
40 |
|
Nitrate |
28- 50 |
33 - 44 |
20 – 22 |
20 |
|
Phosphate (mg/L) |
18-23 |
24- 34 |
3 – 5 |
5 |
|
Total Hardness |
2150 -2206 |
437 -950 |
198- 410 |
100-500 |
|
Faecal Coliform CFU/Ml) |
44-58 |
16-33 |
18-24 |
NR |
DPR: Department of Petroleum Resources, FMEnv: Federal Ministry of Environment: USEPA; United state Environmental Protection Agency: NR: No Record.
|
Parameters |
Ovwian |
Ekakpamre |
Ovu |
DPR/FMEnv Recommendation |
|
Water Temperature (oC) |
36.20 -38.50 |
26.36 – 37.80 |
29.60-31.00 |
30 |
|
pH |
4.90 -5.83 |
5.22 - 5.75 |
6.75-7.80 |
6.5-9.2 |
|
Turbidity |
3.25 - 8.38 |
2.46 - 7.10 |
5.00-9.40 |
10 |
|
Total dissolve solid (mg/L) |
2235 -2440 |
2540 - 2820 |
813- 980 |
2000 (USEPA; 100- 1000) |
|
Dissolve Oxygen(mg/L) |
2 -3 |
2- 5 |
5 – 7 |
5 |
|
Chemical Oxygen Demand (COD) |
35 -40 |
39- 41 |
28-39 |
40 |
|
Nitrate |
32- 43 |
28 - 54 |
15 – 18 |
20 |
|
Phosphate (mg/L) |
13-3 6 |
17- 46 |
4 – 5 |
5 |
|
Total Hardness |
1112 -1206 |
940 -1150 |
180- 210 |
100-500 |
|
Faecal Coliform CFU/Ml) |
10-64 |
6-43 |
8-32 |
NR |
DPR: Department of Petroleum Resources, FMEnv: Federal Ministry of Environment: USEPA; United state Environmental Protection Agency: NR: No Record.
Chapman, P. and Wang, F., Assessing sediment contamination in estuaries. Bulletin of Environmental Contamination and Toxicology 20:3-22, 2001
View ArticleHare, L., Tessier, A. and Warren, L., Cadmium accumulation by invertebrates living at the sediment-water interface. Environmental Toxicology and Chemistry 20(4):880-9, 2001 PMid:11345465
PubMed/NCBIO'Connor, T., National distribution of chemical concentrations in mussels and oysters in the USA. Marine Environmental Research 53:117-43, 2002 00116-7
View ArticleRobinet, T., Feunteun, E., Sublethal effects of exposure to chemical compounds: a cause for decline in Atlantic eels? Ecotoxicology 11:265-77, 2002 PMid:12211699
View Article PubMed/NCBIWurl, O. and Obbard J., A review of pollutants in the sea-surface microlayer (SML): a unique habitat for marine organisms. Marine Pollution Bulletin 48(11-12):1016-30, 2004 PMid:15172807
View Article PubMed/NCBIThurberg, F.P. and Gould, E., Pollutant effects upon cod, haddock, pollock, and flounder of the inshore fisheries of Massachusetts and Cape Cod Bays. In: Buchsbaum R, Pederson J, Robinson WE, editors. The decline of fisheries resources in New England: evaluating the impact of overfishing, contamination, and habitat degradation. Cambridge (MA): MIT Sea Grant College Program; Publication No. MITSG 05-5. 2005, p 43-66. PMCid:PMC1180711
McDonald, D. G., Goldstein, M. D., and Mitton, C., Responses of hatchery-reared brook trout, lake trout, and splake to transport stress. Trans. Am. Fish. Soc. 122:1127?1138,1993 122<1127:ROHRBT>2.3.CO;2
View ArticleUSEPA US Environmental Protection Agency., The Long Island Sound Study: the comprehensive conservation and management plan. Stony Brook (NY): University of New York; Long Island Sound Office of the US EPA. 1994, 168 p.
NEDECO (Netherlands Engineering Company)., The waters of the Western Niger Delta. NEDECO, The Hague, 1954.
Boyd, C.E.and Tucker,C. S., Water Quality and Pond Soil Analysis for Aquaculture, vol. 1014, Alabama Agricultural Experiment Station, Auburn University, Auburn, Ala, USA, 1992
Roy, H., Pollution: Causes, Effects, and Control by (editor). Royal Society of Chemistry, Comprehensive, 2001, (579-page) introduction to all forms of pollution
Christopher, M., Biology of Freshwater Pollution . Prentice Hall, 2001. This book is aimed at advanced undergraduates and water pollution professionals, 2001.
Delille, D and Gleizon, F., Distribution of enteric bacteria in Antarctic seawater surrounding the Port-aux-Francais permanent station (Kenguelen Island). Marine Pollution Bulletin [Mar. Pollut. Bull.]. Vol. 46, no.9, pp.1179-1183. Sep 2003. 00164-4
View ArticleFEPA., Guidelines and standards for environmental pollution control in Nigeria.Federal Environmental Protection Agency, Nigeria. 1991, p. 20.
Campbell, D., A review of the culture of Sarotherodon melanotheron in West Africa. FAO corporate Document Repository. Project RAF/82/009, 1978.
Alabaster, J.S. and Lloyd, R., Water quality criteria for fresh water fish. Butter- worth and Co.Ltd, London 1980,:pp.12-260. PMCid:PMC2044270
WHO., Guidelines for Drinking Water Quality Vol. 1 Geneva, 2006
Bennet, GN., Winter kill of fishes in an Illinois lake. Lake management reports Illinois biology notes. 1973, 19:1-9.
Hughes, K.A. and Thompson, A., Distribution of sewage pollution around a maritime Antarctic research station indicated by faecal coliforms, Clostridium perfringens and faecal sterol markers. Environmental Pollution. 127 315- 321, 2003. Available online at www.sciencedirect.com
View ArticleUNESCO/WHO/UNEP.,Water quality Assessments-Aguide to use Biota,Sediments and Water in environmental Monitoring-Second Edition, 1992.
