Nasibova E.M
Email: doc.nasibova.esmira@gmail.com
© 2019 Sift Desk Journals. All Rights Reserved
VOLUME: 3 ISSUE: 2
Page No: 147-152
Nasibova E.M
Email: doc.nasibova.esmira@gmail.com
Nasibova E.M., Pashayev C.N.
Azerbaijan Medical University
Nasibova E.M., Pashayev C.N., The use of non-invasive ventilation (NIV) in the treatment of patients with COVID-19(2020) Journal of Anesthesia & Surgery 3(2)
Non-invasive ventilation (NIV) is a method of respiratory support, in which a mask is used as the main interface, which can be easily applied and also easily disconnected from the patient's respiratory tract. The study included patients admitted to the intensive care unit of the surgical clinic of the AMU from April 1 to May 1, 2020. NIV has significant advantages over traditional mechanical ventilation. But it must be remembered that even in experienced hands, NIV is successful only in 75–90% of all cases, which depends on many factors, such as the severity of acute respiratory failure, training and experience of medical personnel, and the place of respiratory support. As with many types of therapy, operations, and technologies, improvement in the results of this method can be expected as experience is gained.
Key words: COVID-19, non-invasive ventilation, mechanical ventilation.
Non-invasive ventilation (NIV) is a method of respiratory support, in which a mask is used as the main interface, which can be easily applied and also easily disconnected from the patient's respiratory tract.
The harmful effects of the treatment of respiratory distress syndrome (RDS) with invasive ventilation have led to a deeper study of non-invasive ventilation methods (NIV). The key to success with NIV is proper patient selection. It is important to emphasize that a strict selection of patients is required for NIV, the main criteria are the preservation of consciousness and patient consent, as well as stable hemodynamics. Based on extensive experience using NIV in 147 patients with ARDS, Antonelli and count. recommend avoiding NIV in patients with SAPS greater than 34 points. According to Rana and colleagues, metabolic acidosis and severe hypoxemia are predictors of a poor response to NIV patients with ARDS. The possibility of using NIV in well-selected patients with ARDS has been shown in several studies (4, 7, 8). The authors of all studies devoted to NIV are unanimous in that there are significantly fewer complications when used than NIV with traditional mechanical ventilation. Mask ventilation allows you to minimize the number of infectious and "mechanical" complications. Nosocomial pneumonia is a common complication of lung ventilation and is a critical factor in determining patient outcome. With NIV, there is no direct contact with the trachea (endotracheal tube, aspiration catheter), the patient can release sputum after removing the mask. In the largest study by Meduri et al., Which included 158 patients who received NIV, nosocomial pneumonia was detected in only one patient. Studies show that complications of NIV, as a rule, do not require cessation of respiratory support (12, 14, 17, 19). The most common of these are facial skin necrosis, conjunctivitis, nasal irritation, transient hypoxemia, general discomfort, aerophagia, leakage. Erosions and necrosis of the skin are formed most often in the place of the greatest pressure of the mask on the skin of the face (usually the nose bridge). According to various studies, they occur in 6–18% of cases. Erosions and necrosis of the skin are not a serious complication, because they usually heal very quickly (after 2-7 days). The disadvantage of NIV is the need for patient motivation. According to a meta-analysis conducted by Muir, the NIV procedure was discontinued due to intolerance to patients with the presence of a mask in 37 of 747 cases (5%) of using NIV in ONE (5, 15, 20). This method is practically not used in patients with severe impaired consciousness (the number of points on the Glasgow scale is less than 9), because these patients need protection of the respiratory tract and require frequent sanitation of the tracheobronchial tree, which is difficult to do when using NIV. Unfortunately, in our clinical practice, NIV is not widely used, while the experience of many countries has demonstrated the benefits of a wider implementation of NIV. For example, in the USA, the use of NIV over one decade increased by 462%, which led to a decrease in cases of invasive ventilation by 42% and was generally associated with a decrease in hospital mortality (5). For a wider implementation of NIV, an adequate level of providing high-quality equipment is important (6). The use of high-quality respirators is more comfortable for patients, and the ease and simplicity of setting respiratory support parameters are important for a doctor (6). A wide range of types and sizes of masks is also important. In recent years, manufacturers of respiratory equipment have presented a wide selection of different masks and helmets that are comfortable for patients, which reduces the risk of mechanical complications.
The aim of the study: to determine the benefits of NIV in ODN patients with COVID-19.
Study material: The study included patients admitted to the intensive care unit of the surgical clinic of the AMU from April 1 to May 1, 2020.
The results of the study: Our experience with NIV has shown that most patients treated with NIV tolerate this procedure relatively well already at the initial stage. However, in a number of patients, during the first minutes or hours of NIV, no improvement (clinical indicators and gas exchange) is observed or the procedure is poorly tolerated, the proportion of such patients is usually about 15–35%. Usually, a respiratory support session of 2–3 hours is sufficient to predict the success of the NIV or response to the NIV. In normal practice, the effectiveness of NIV therapy is obvious with a simple examination - there is a decrease in the frequency of respiratory movements and the work of auxiliary respiratory muscles. Objective markers of the effectiveness of mask ventilation are changes in arterial blood gas parameters: an increase in pH and a decrease in PaCO2. A short NIV session allows you to identify not only patients who can be effectively managed with NIV in the future, but also patients with a poor response who subsequently need urgent tracheal intubation and connection to a ventilator. Experience shows that longer attempts to use NIV without achieving a noticeable improvement only delay the time of the use of mechanical ventilation, which significantly increases the risk of increased respiratory failure, an adverse outcome, up to a lethal one. Using NIV, we came to the conclusion that, in most cases, NIV therapy failures are detected quite early - in the first hours from the initiation of respiratory support, however, in some patients, NIV therapy failure manifests itself later - 24–48-72 hours after the initial improvement. Lack of improvement in consciousness or respiratory acidosis 24 hours after onset is NIV another predictor of NIV failure. Indications for the implementation of NIV are as follows:
The criteria for the exclusion of NIV in acute respiratory failure are as follows:
The criteria for termination of NIV and the transition to traditional mechanical ventilation include the following:
The physiological effects of NIV are as follows:
NIV also has the following economic significance:
The study identified the following benefits of non-invasive ventilation:
In our clinic, NIV was performed using Salvia Elisa ventilator respirators in CPAP + PSV mode through a face mask. Used standard masks from Drager (Germany) or Respironics (USA). To determine the parameters of the gas and acid-base composition of the blood, an ABL500 gas analyzer with an OSM3 oximeter (Radiometer, Denmark) was used. Indicators of the function of external respiration were recorded from the display of the respirator. All data were recorded immediately before the start of ventilation. The level of PEEP and pressure support was set individually, based on the specific clinical situation. The ventilation parameters required by patients were as follows: PEEP - from 5 to 12 cm of water, PSV - from 0 to 14 cm of water. Art., FiO2 - from 0.3 to 0.6. At the initial stage, auxiliary ventilation was carried out in a continuous mode. Further, a gradual decrease in respiratory support was carried out in accordance with the degree of clinical improvement, after which they switched to NIV sessions for several hours a day until it was completely canceled. The criterion for successful NIV was the improvement of the arterial blood gas composition and the ability to avoid endotracheal intubation.
Clinical case:
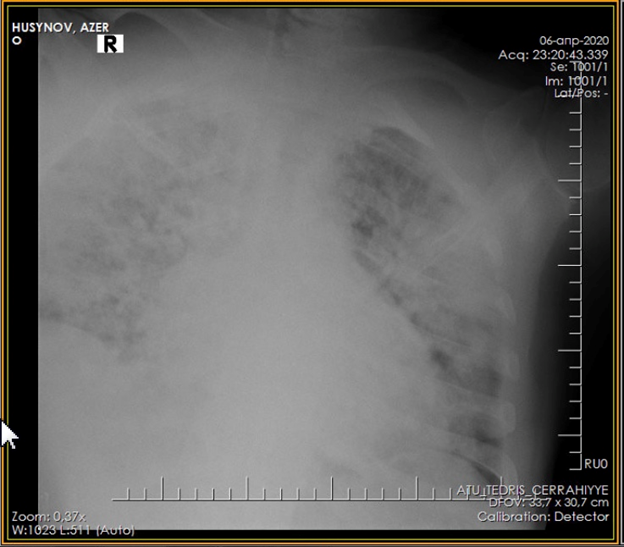
Figure 1. X-ray upon admission
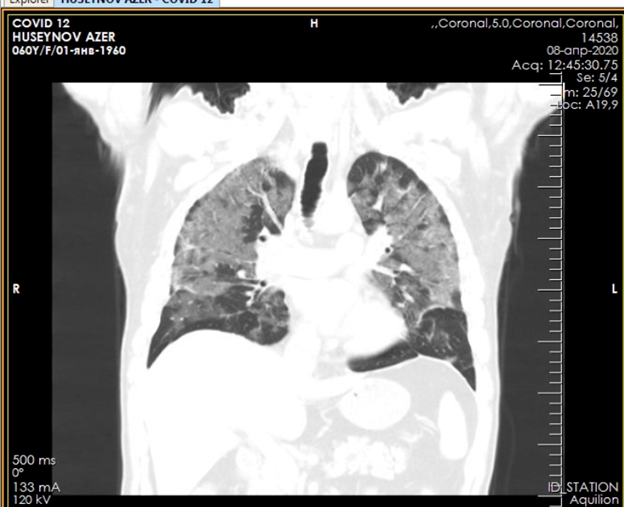
Figure 2. CT scan of the lungs upon admission
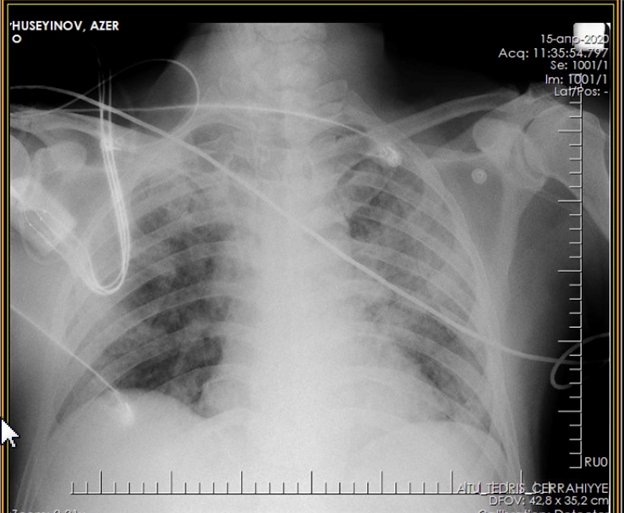
Figure 3. Radiograph after improvement of the patient
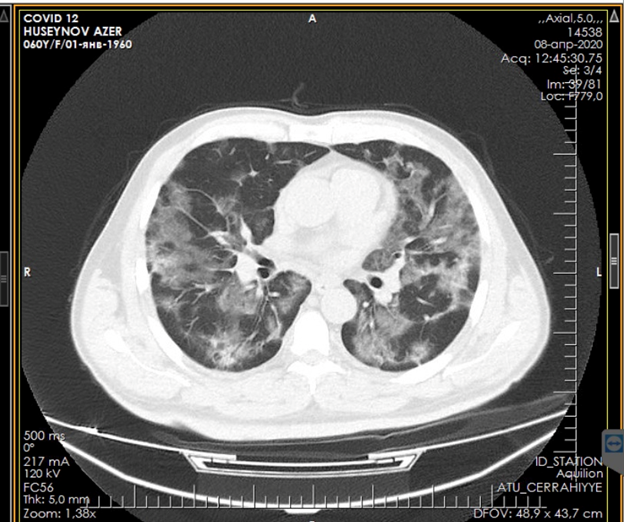
Figure 4. CT scan after patient improvement
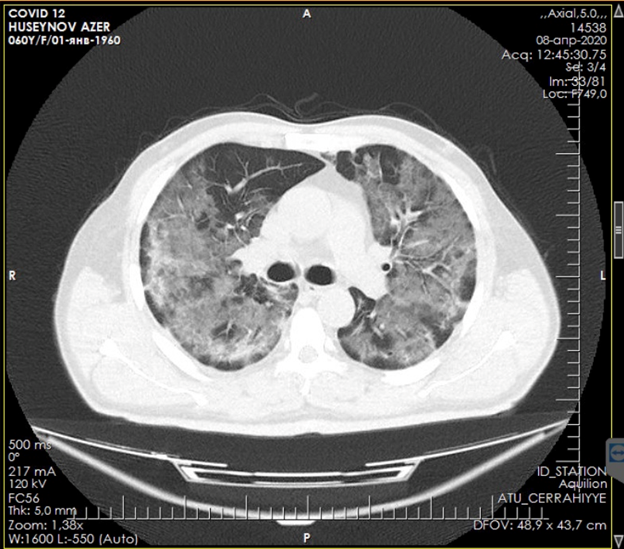
Figure 5. CT scan of the lungs after improvement
Non-invasive ventilation was carried out by an oral-nasal mask with a ventilator ELISA. Installation and adjustment of the parameters was carried out according to the general condition and according to the data of blood gases: respiratory rate <35, pH> 7.30, neurological dysfunction according to the Kelly scale> 3-5, a modified scale for determining the participation of auxiliary respiratory muscles <3 points. For hypercapnia, the following parameters were set: Ps - 12, PEEP - 5 cm water column, FiO2 -30-40%, and with hypoxemia - Ps - 12, PEEP - 5 cm water column, FiO2 -50-60 %. The median treatment period with NIV was 6 days. The average daily treatment time with NIV on the first day was 17.3 hours, on the second day - 18.2 hours and on the third day 16.7 hours. The patient was discharged on the 12th day with improvement.
Alexiou S, Panitch HB. Physiology of non-invasive respiratory support. Semin Fetal Neonatal Med. 2016;21(3):174-80. PMid:26923501
View Article PubMed/NCBIAlraddadi, B. M., Qushmaq, I., Al-Hameed, F. M., Mandourah, Y., Almekhlafi, G. A., Jose, J., et al. Saudi Critical Care Trials Group. (2019). Noninvasive ventilation in critically ill patients with the middle east respiratory syndrome. Influenza & Other Respiratory Viruses, 13(4), 382-390. PMid:30884185 PMCid:PMC6586182
View Article PubMed/NCBIArabi, Y. M., Fowler, R., & Hayden, F. G. (2020). Critical care management of adults with community-acquired severe respiratory viral infection. Intensive Care Medicine, 46(2), 315- 328 3. Guan, L, Zhou, L, Zhang, J, et al. (2020). PMid:32040667
View Article PubMed/NCBIArabi Y, Al‐Omari A, Mandourah Y, et al. Critically ill patients with the middle east respiratory syndrome (MERS): a multicenter retrospective cohort study. Crit Care Med. 2017;45(10):1683‐1695. PMid:28787295
View Article PubMed/NCBIBellani G, Laffey JG, Pham T, et al. Noninvasive ventilation of patients with acute respiratory distress syndrome. Insights from the LUNG SAFE study. Am J Respir Crit Care Med. 2017;195(1):67‐77. PMid:27753501
View Article PubMed/NCBIBrewster D., Chrimes, N., Do, T., Fraser, K., Groombridge, C., et al (2020). Consensus statement: Safe Airway Society principles of airway management and tracheal intubation specific to the COVID-19 adult patient group. Medical Journal of Australia. PMid:32356900
View Article PubMed/NCBICarteaux G, Millan-Guilarte T, De Prost N, Razazi K, Abid S, Thille AW, Schortgen F, Brochard L, Brun-Buisson C, Mekontso Dessap A. Failure of noninvasive ventilation for de novo acute hypoxemic respiratory failure: role of tidal volume. Crit Care Med. 2016;44(2):282-90. PMid:26584191
View Article PubMed/NCBIDu, K. Noninvasive ventilation in patients with acute respiratory distress syndrome. Crit Care 23, 358 (2019). PMid:31730002
View Article PubMed/NCBIGuan, L, Zhou, L, Zhang, J, et al. (2020). More awareness is needed for severe acute respiratory syndrome coronavirus 2019 transmission through exhaled air during noninvasive respiratory support: experience from China. Eur Respir J 55: 2000352 PMid:32198275
View Article PubMed/NCBIHe H, Sun B, Liang L, Li Y, Wang H, Wei L, Li G, Guo S, Duan J, Li Y, et al. A multicenter RCT of noninvasive ventilation in pneumonia-induced early mild acute respiratory distress syndrome. Crit Care. 2019;23(1):300. PMid:31484582
View Article PubMed/NCBIHui DS, Chow BK, Lo T, et al. Exhaled air dispersion during noninvasive ventilation via helmets and a total facemask. Chest. 2015;147(5):1336‐1343. PMid:25392954
View Article PubMed/NCBILiu, Q., Gao, Y., Chen, R., & Cheng, Z. (2016). Noninvasive ventilation with helmet versus control strategy in patients with acute respiratory failure: A systematic review and meta-analysis of controlled studies. Critical Care (London, England), 20(1), 265. PMid:27549178
View Article PubMed/NCBILuo J, Wang MY, Zhu H, Liang BM, Liu D, Peng XY, et al. Can non-invasive positive pressure ventilation prevent endotracheal intubation in acute lung injury/acute respiratory distress syndrome? A meta-analysis. Respirology. 2014;19:1149-57. PMid:25208731
View Article PubMed/NCBIPatel BK, Wolfe KS, Pohlman AS, Hall JB, Kress JP. Effect of noninvasive ventilation delivered by helmet vs face mask on the rate of endotracheal intubation in patients with acute respiratory distress syndrome: a randomized clinical trial. JAMA. 2016;315(22):2435-41. PMid:27179847
View Article PubMed/NCBIShen Y, Cai G, Gong S, Dong L, Yan J, Cai W. Interaction between low tidal volume ventilation strategy and severity of acute respiratory distress syndrome: a retrospective cohort study. Crit Care. 2019;23(1):254. PMid:31300012
View Article PubMed/NCBITeam NCPERE. Vital surveillances: the epidemiological characteristics of an outbreak of 2019 novel coronavirus diseases (COVID-19) - China. China CDC Weekly. 2020;2(8):113-22. 2. Yang X, Yu Y, Xu J, Shu H, Xia J, Liu H et al.
Vital FM, Ladeira MT, Atallah AN. Non‐invasive positive pressure ventilation (CPAP or bilevel NPPV) for cardiogenic pulmonary oedema. Cochrane Database Syst Rev. 2013;5:CD005351. PMid:23728654
View Article PubMed/NCBIXia, J., Zhao, J., Cheng, Z., Hu, Y., Duan, J., & Zhan, Q. (2020). Non-invasive respiratory support for patients with novel coronavirus pneumonia: Clinical efficacy and reduction in risk of infection transmission. Chinese Medical Journal1. doi:10.1097/CM9.0000000000000761 PMid:32097201
View Article PubMed/NCBIXu X, Yuan B, Liang Q, et al. Noninvasive ventilation for acute lung injury a meta‐analysis of randomized controlled trials. Heart Lung. 2016;45(3):249‐257. PMid:27154849
View Article PubMed/NCBIZhou F, Yu T, Du R, Fan G, Liu Y, Liu Z et al. Clinical course and risk factors for mortality of adult inpatients with COVID-19 in Wuhan, China: a retrospective study. Lancet, 2020. doi: 1016/S0140-6736(20)30566-3.
