Moustafa Gamal Snousy
E-MAIL: moustafa_gamal93@yahoo.com
Prevalent Remediation Technologies for Organic Contaminants
Corresponding Author
Affiliation
Moustafa Gamal Snousy*1, Thanaa Abdel-Moghny2, El-Sayed, E.3
1Petrotread co., 1 Anwar Al Moftty st.-Abbas El Akkad st.-Naser City, Cairo, Egypt Tel:+201006517228
2Applications Department, Egyptian Petroleum Research Institute, Ahmed El-Zomer, Nasr City, P.O. Box 11727, Cairo, Egypt
3Geology Department, Faculty of Science, El-Minia University, P.O. Box 61519, El-Minia, Egypt
Article Reviewed By:
Esther Bloem(Esther.Bloem@nibio.no)
Citation
Moustafa Gamal Snousy, Prevalent Remediation Technologies for Organic Contaminants(2017)SDRP Journal of Earth Sciences & Environmental Studies 2(1)
Abstract
Any chemical spill that may affect the surrounding ecosystems must be remediated quickly and efficiently to minimize possible contamination. When the problem of groundwater pollution began to be discovered, the initial approach to remediation assumed that most of contamination was in the soil at the site of the spill; from where it was reasoned that the contaminated soil could be treated, and contaminated water. Various physical, chemical, biological, and their combined (i.e. Physico-chemical, thermal) technologies have been attempted to remediate or remove the pollutants from soils and groundwater or to reduce the contamination to a safe and acceptable level. There are several methods used for the clean-up of contaminated soils and groundwater, combination of processes (treatment trains) may offer the most effective remediation and easily to applied where soils and groundwater were contaminated by petroleum hydrocarbons.
Introduction
Pollution defined as the introduction of elements, compounds or energy into the environment at levels that present an unacceptable risk to the ecosystems. For example, polluted soils may affect the hydrosphere compromising the quality of drinking water resources and threatening the aquatic ecosystems [1]. Pollutants can build up in soils from several sources, such as spreading of wastes (e.g. sewage sludge or other biosolids) to land especially where these wastes have been applied repeatedly over several years. Some wastes (e.g. dredgings, pulverised fuel ash or mine spoils) constitute new “soil-forming” materials [2]. Soils may become polluted by atmospheric deposition from traffic, and incinerator or metal smelting emissions over a period [3]. Soils may also be polluted through the spillage of liquids such as oil or industrial solvents or through flooding or irrigation with polluted water
[4]. Whilst most of the early soil pollution related to metals or other inorganic pollutants, there has been an increasing concern over the last few decades regarding organic contaminants, a reflection of their widespread use in industry as solvents, feedstocks and their presence in industrial wastes [5]. Fuel hydrocarbons, for examples, are major pollutants of environment [6].
Materials & Methods
- Treatment Technologies
The pollutants leakage that may affect the surrounding groundwater must be remediated quickly and efficiently to minimize possible contamination. When the problem of groundwater pollution began to be discovered, the initial approach to remediation assumed that most of contamination was in the soil at the site of the spill; from where it was reasoned that the contaminated soil could be treated, and contaminated water. Various physical, chemical, biological, and their combined (e.g. Physico-chemical) technologies have been attempted to remediate or remove the contaminants from soils and groundwater or to reduce the contamination to a safe and acceptable level [7]. Combination of treatment processes may offer the most effective remediation [8].
Improvement in both the desorption efficiency from soils and the mobility and bioavailability in aqueous phase are essential to the remediation of organic contaminants in soils and groundwater. There are several methods used for the clean-up of contaminated soils and groundwater: air sparging or biosparging (aerated accumulation), pulsed air sparging, air/ozone sparging (ozonation), natural attenuation, bioremediation (biological methods), phytoremediation, incineration, off-site disposal, soil flushing, soil washing, flushing and washing of soils, soil vapor extraction, solidification and stabilization, thermal desorption, vitrification, pump and treat, permeable reactive barriers, immobilization, chemical oxidation, and electrokinetic remediation of contaminated-soil, use of this methodology in treatments is cost-effective and easy to apply in different conditions. However, no universal method can be devised for the removal of oil from contaminated sites. Remediation chemical methodologies can be executed in to two different modes are; in situ “in site” (by direct injection of chemical into contaminated site using the subsurface itself as the reactor) and ex-situ “on site” also called prepared beds (physically carried out at the polluted environs by first removing the contaminated area for treatment in aboveground), they are more attractive from an economic point of view [9].
2.1.Preponderant Treatment Trains for Organic Contaminants
It is recognized that no single specific technology may be considered as a panacea for all contaminated site problems [10]. Using just one technology may not be adequate to remediate some contaminated sites with different contaminants and complex site conditions. Under such situations, different technologies are used sequentially or concurrently along with the primary treatment technology to achieve the remedial goals. Such use of multiple remediation technologies is often referred to as “treatment trains” the selection depends on contaminant and site characteristics, regulatory requirements, costs, and time constraints [11]. Since most remediation technologies are site-specific, the selection of appropriate technologies is often a difficult, but extremely important, step in the successful remediation of a contaminated site. Therefore, the successful treatment of a contaminated site depends on proper selection, design, and adjustment of the remediation technology’s operations based on the properties of the contaminants, soils and on the performance of the system. The typical treatment trains used in organic contaminated includes surfactant flushing accompanied by water washing to enhance air sparging.
2.1.1. Air Sparging (AS)
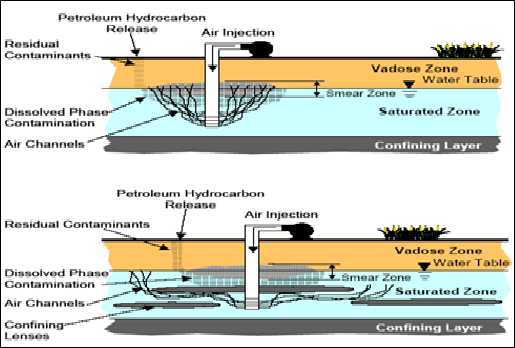
With the growing interest in environmental remediation, various approaches have been proposed for treating petroleum hydrocarbon contaminated sites. More cost-effective methods are needed to clean up sites that are contaminated with hydrophobic organic compounds. One such technique that has proven to be successful is air sparging, also known as biosparging [12]. Air sparging first used in Germany in the mid-1980s, air sparging is the subsurface equivalent of a surface air-stripping system, involves the injection of atmospheric air, under pressure into the contaminated zone to volatilize contaminants and to promote biodegradation by increasing subsurface oxygen concentrations as shown in Figure (1) [13].
Air sparging were conducted at a site where soils and groundwater or removal residual source (smear zone) were contaminated by petroleum hydrocarbons, organic hydrocarbons originating from leakage of petroleum or hazardous chemical storage facilities have been a main contamination source in the unsaturated soil environment [14]. It has been used for the past 30 years for the remediation of volatile organic compounds (VOCs) which trapped in soil pores in the saturation and capillary zones both above and below the groundwater table, because they readily transfer from the dissolved to the gaseous phase [15]. For semi-volatile and non-volatile contaminants, such as diesel, jet fuels and lubricating oils, air sparging is not the removal mechanism [16].
2.1.1.1. Air Sparging Vs. Contaminant Removal Mechanisms
Even though air sparging has been successfully applied at several contaminated sites, the airflow distribution in saturated media and the interactions of various physical, chemical and microbial processes during air sparging operations are still not well understood. This is mainly due to the difficulty of obtaining, at reasonable costs, enough information on the phenomena occurring within the contaminated saturated zone to be treated by air injection [17]. Suthersan [18] suggested three contaminant removal mechanisms that occur during air sparging include: (1) stripping of dissolved VOCs, based on the mass transfer of dissolved contaminants to the vapor phase through the air-water interface created by the injection of air within the water-saturated media, (2) direct volatilization, based on the mass transfer from the trapped and adsorbed phase below the water table in the capillary fringe to the vapor phase, through the air-water interface created by the injection of air within the water-saturated media, and (3) aerobic biodegradation, based on the disruption of the contaminants molecule, through the action of microorganisms that use (and consume) oxygen as electron donor.
2.1.1.2. Air Sparging System Advantages
Issues relating to air sparging systems include the following advantages:
- Application of the technology is widely recognized by the regulatory community as an effective remedial technology for removing volatile contaminants from soil and groundwater.
- Implementation is relatively simple, because only readily available commercial equipment is utilized, the major components of a typical air sparging system include an air injection well, a compressor or blower to supply air, monitoring points and polyvinyl chloride (PVC) well casing, etc. [16]. The equipment is relatively easy to install and causes minimal disturbance to site operations.
- Cleanup times are relatively short, this technique, in fact, was found to accelerate the remediation process for non-aqueous phase liquid (NAPL) contaminated aquifers [19].
- Use of low-cost, direct-push well installation techniques is possible. Direct-push technologies are most applicable in unconsolidated sediments and at depths of less than 30 ft. Although, in relatively coarse-grained lithologies, direct-push rigs may experience some difficulty in obtaining good material recovery and specialized equipment may be needed to obtain relatively undisturbed samples from depths greater than 10 ft [20].
- In situations where subsurface contamination is located under a building or runway (i.e. needing a nominal above ground footprint) noninvasive horizontal or angled sparge wells may be used.
- Air sparging may be effectively implemented to intercept and treat a migrating contaminant plume [21].
- Effect of air temperature: the increase in the soil air temperature would enhance the microbial degradation of the hydrocarbon contaminants [22], optimal biodegradation occurs between 20 and 40 °C. Many sites have been successfully remediated using air sparging [23]. Air sparging is often used as part of a larger system of treatment processes and works in tandem with other technologies at complicated remediation sites.
- Biodegradation is one weathering process whereby oil-degrading microbes convert the hydrocarbons to simpler components [24]. Air sparging provides oxygen for aerobic microbial degradation of contaminants, an effective mechanism in the later stages of remediation [19].
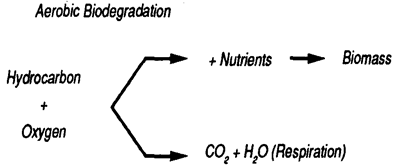
As an added benefit, air sparging may boost biodegradation by increasing the amount of oxygen available to microorganisms such as bacteria, which live in the soil or groundwater to break down contaminants into harmless substances as illustrated in Figure (2). The increased airflow through the subsurface provided by air sparging also stimulates the biodegradation of contaminants (i.e. supplying oxygen or increased dissolved oxygen to oxygen deprived soil microorganisms by injecting air through unsaturated contaminated soil at a low flow rate that can promote aerobic biodegradation). Bioremediation of soils can be contemplated as a polishing stage after a chemical treatment step [25]. The use of bioremediation techniques in conjunction with chemical and physical treatment processes, i.e., the use of a “treatment train,” is an effective means for comprehensive site-specific remediation [26]. A promising application of air sparging is for enhancement of biodegradation of volatile, semivolatile and residual nonvolatile chemicals in soils [27].
2.1.1.3. Limitations of Air Sparging
Despite these advantages, however, these are important observations related to the performance of air sparging technology:
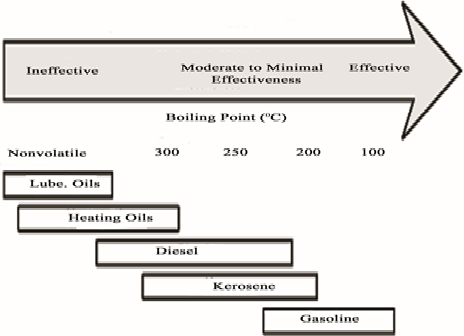
- Air sparging typically, not effective for recovering light non-aqueous phase liquid (LNAPL) layers greater than one foot thick or recovering dense non-aqueous phase liquid (DNAPL) free product. In general, constituents in petroleum products with boiling points less than 250 ºC to 300 ºC are sufficiently volatile for removal by air sparging as shown in Figure (3). Nearly all gasoline constituents and a portion of kerosene and diesel fuel constituents can be removed by air sparging. Heating and lubricating oils cannot be removed by air sparging. However, air sparging can promote biodegradation of semivolatile and nonvolatile constituents [27].
- Sites with dispersed areas of pollution may be difficult to clean up because of the limited sparging radius of influence. Because air sparging increases the rate of contaminant volatilization, it is important to be aware of the potential for migration of VOCs impacted vapor to human and ecological receptors at potential levels of concern. A soil vapor extraction (SVE) system can be used to reduce or eliminate vapor migration problems, but the proximity of the site to buildings or other structures should be taken into careful consideration.
- Areas containing stratified soils with low permeability will restrict vertical air flow and possibly spread the contaminant laterally, bringing about the need for groundwater control and increased costs. Hydrocarbon vapors released through this process can spread to uncontaminated areas in the unsaturated zone if the SVE system isn’t properly designed to capture the vapors.
- Confined aquifers cannot be treated by this remediation technique due to possible off-site migration of the volatilized contaminants [9].
- Biosparging can be used at most types of petroleum contaminated sites, but it is least effective on heavy petroleum because of the length of time required [29].
- This technology is ineffective in the case of non-strippable and non-biodegradable contaminants [13].
- Heterogeneous geologic conditions, with the presence of low permeability layers overlying zones of higher permeability, reduce the effectiveness of the system.
- Air sparging works best in uniform coarse-grained soils, such as sand and gravel and can enhance the aerobic biodegradation in the subsurface by transferring oxygen [30]. Silt and clay sediments are not appropriate for this technology [31].
- Other site factors that influence the applicability of air sparging include the thickness of the saturated zone and the depth of the groundwater [32].
Air sparging used most often at sites with mid-weight and lighter petroleum. These higher molecular weight petroleum constituents require long periods of time to degrade. Heavier products do not evaporate during aeration but are broken down by microorganisms present in the soil at the treatment site. Biodegradation tend to take longer to reduce heavier products, but this does not mean that this technology cannot be employed for them [33]. The presence of a high concentration of heavy metals inhibit microbial growth [10].
In general, air sparging is more effective for constituents with greater volatility and lower solubility and for soils with higher permeability. At present, models used in predicting the results of air sparging are simple and limited in scope. Overall, while air sparging is a promising technology, questions remain unanswered and the technology needs further research and field testing.
2.1.1.4. Air Sparging System Supplemental
The organic hydrocarbons and poly-aromatic hydrocarbons (PAHs) are bind strongly to the soils and sediments, and sparingly soluble in water, thus their removal by various treatments is difficult and hence soil remediation depends on the ability to desorb them from the soil and to incorporate them in the bulk aqueous phase. An effective remediation technique for such situations is the surfactant-solution flushing approach [35]. The replacement and disposal of surfactant solution or the need for supplemental groundwater and soils flushing plays a large role in treatment economics. So, air sparging with surfactant enhancement are used with low and high pollution concentration. In the lab or in the plant, interfacial area can be increased and energy added to the water interface by mixing, shaking, etc. in an aquifer however, the only means of adding this energy is the pressure drop across the drop created by the pressure gradient between the injection and extraction wells up-gradient and down-gradient of the drop. Surfactant-enhanced soil remediation has been proposed as an alternative method for recovering residual organic materials from contaminated soils. Remediation with surfactants may be the primary action to remove large amounts of oil from the soil matrix, followed by or used to enhance other remediation techniques such as bioremediation [36].
2.1.2. Surfactants and Pollutant Interaction
Surfactants or surface active agents are amphiphilic compounds all share interesting physicochemical characteristics at surfaces and interfaces. Surfactants represents a heterogeneous and long-chain molecule containing both (polar) hydrophilic or water soluble (head) group that has a strong attraction for water, and (non-polar) hydrophobic or water insoluble (tail) group, usually consists of aliphatic or aromatic hydrocarbons with very little attraction for water moieties on the same molecule. Dual nature causes surfactants to adsorb at interfaces or align at the interface between two immiscible phases (air–water and water–oil) by positioning at the interface, thereby reducing the interfacial energies [37]. As they are amphiphilic molecules which making them ideal for solubilization of hydrophobic compounds, depending on concentration and surfactant type, by monomers accumulate at interfaces present in the system (e.g., air−water, oil−water, soil−water), or acting as a bridge between the oil and water interface, can reduce the surface tension of water [38]. Thus, this promotes the mobilization of contaminants from unsaturated soils, or thereby making the contaminant more available for remediation.
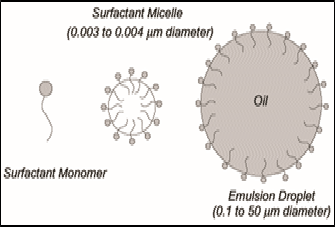
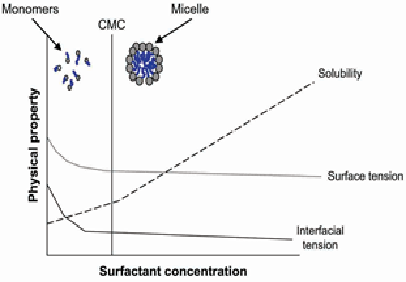
As the interfacial areas are satisfied and the aqueous surfactant concentration increases the monomers aggregate to form micelles as shown in Figure (4). At low concentrations surfactants exist solely as monomers, but the concentration at which surface tension becomes constant or the concentration at which micelles first begin to form is known as the critical micelle concentration (CMC) as illustrated in Figure (5).
This concentration corresponds to the point where the surfactant first shows the lowest surface tension. Surfactant monomers orient themselves into structures called micelles (i.e. the monomers aggregate to form micelles), which are formed by a shell of hydrophilic ends. Hydrophobic ends form the interior of the micelle, called the hydrophobic pseudophase. Micelles are on the order of 0.003 μm to 0.004 μm in diameter [40]. Hydrophobic compounds can partition into the interior of micelles by reducing their surface tension, increasing their effective solubility in the aqueous phase [41].
2.1.2.1. Role of Surfactants in Soil Flushing
Surfactants are the active ingredients found in soaps and detergents with ability to concentrate at the air–water interface and are commonly used to separate oily materials from a media. The use of surfactants to enhance the removal of soil contaminants has received increasing attentions in recent years. Petroleum users have traditionally been the major users, as in enhanced oil removal applications [43]. With the growing interest of surfactants applications in environmental remediation, Barathi and Vasudevan [44], and McCray et al. [45] have studied the removal of single and double components of petroleum hydrocarbons through soil flushing using aqueous surfactant solutions and conclude that it is important to evaluate the characteristics of the contaminated soils such as soil particle size distributions, organic and inorganic materials contents.
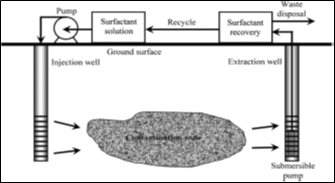
High interfacial tension (IFT) and low solubility in water the most physical characteristics of organic contaminants commonly found (because of their inability to compete with the strong hydrogen bonds between water molecules), which decrease the efficiency of soil remediation during water flushing and bioremediation process [46]. Flushing with water alone may take decades to achieve the desired level of removal [47]. To overcome this problem, the use of surfactants is a commonly employed to increase the desorption rates of sorbed contaminants from soil, hence also making them available for remediation [48, 49]. These surfactants can reduce the time to treat a site compared to the use of water alone. Consequently, beginning in the late 80s, there has been an increasing willingness to consider the use of chemical enhancements to accelerate the remediation effort while cleaning up more sites with less money [50]. This brings us to the emerging role of surfactants in remediation, flushing of soils has been proposed as a promising innovative remediation technology due to its potential for treating not only oils contaminated soils but also those contaminated by heavy metals [46]. The schematic presentation of the surfactant enhanced remediation (SER) process is shown in Figure (6). For this remediation scheme, the surfactant solution is injected below the ground to enhance the extraction of the contaminant.
2.1.2.2. Mechanisms of Surfactants Contaminant Recovery
Surfactants enhance organic contaminant recovery through two mechanisms. First, occurs below the critical micelle concentration (soil rollup mechanism or mobility) as the surfactant is dissolved in water, the surface tension between water and contaminants that slows the mobility of the organic components of the aqueous phase is greatly decreased and the ability of viscous force to displace the oil is favored [52]. Therefore, surfactants can transfer the hydrophobic organic compounds (HOCs) to the mobile phase [53].
Surfactant monomers are responsible for the soil rollup mechanism which occurs in two steps. In the first step surfactant monomers accumulate at the soil-contaminant and soil−water interfaces and increase the contact angle between the soil and the contaminant (i.e. change the wettability of the system). Surfactant molecules adsorbed on the surface of the contaminant cause repulsion between the head group of the surfactant molecule and the soil particles, thereby promoting the separation of the contaminant from the soil particles. In the second step, convective currents create agitation and abrasion which provides the energy necessary to create additional surface area of the oil phase and thus displace the oil from the soil. Both steps are necessary for the soil rollup mechanism to be significant [41].
Second, occurs above the critical micelle concentration, surfactants can form aggregates known as micelles. Which could increase the apparent solubility of the oil in water, thus Surfactants can increase the solubility of hydrophobic compounds significantly through a process known as solubilization [54].
2.1.2.3. Emulsions and Emulsification of NAPL
An emulsion is a two-phase system consisting of an insoluble phase dispersed as droplets within an external, continuous phase [55]. Addition of surfactants to an oil/water system can promote the formation of emulsions. There are many texts about emulsions that are available for reference [56]. Mobilization of NAPL residual as an emulsion has been studied as an enhanced oil recovery (EOR) technique [57]. Emulsions have applications in many industries, more recently, use of emulsions has been considered for environmental remediation techniques [39].
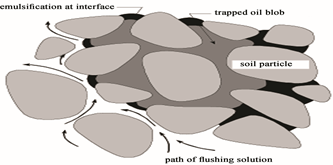
Influence of interfacial area during surfactant enhanced soil flushing is depicted in Figure (7). Emulsification of residual NAPL is believed to occur at interfaces between residual NAPL and the surfactant solution. As seen in the Figure (7), surface area of the complex blob is relatively small as compared to blob volume. It is hypothesized that removal of a residual blob by emulsification processes is limited by interfacial area and diffusion, like limitations described by Wilson et al. [58] and Powers et al. [59] for dissolution during water flushing alone. In general, surfactant soil remediation is done by detaching organic molecules adsorbed on soil and trapped in the pores, followed by their encapsulation within micelles formed at a concentration greater than the CMC [60].
2.1.2.4. Technical Challenges of Surfactant Flushing
Technical challenges to the successful use of surfactant flushing include locating and delineating the NAPL source zone and obtaining an accurate estimate of the initial NAPL mass and its spatial distribution. Additional requirements include characterizing the hydraulic properties of the aquifer, delivering and distributing the injected chemicals to the targeted zone, and designing the optimum chemical formulation for a given NAPL composition and soil type. The implementability of surfactant flushing will depend on site-specific geologic conditions and on the type of NAPL present at the site [61]. Soil flushing tends to work best at sites with soils that have spaces that permit the movement of the flushing solution through it; high permeability soils are favored over low permeability soils [62]. If the soil has a high percentage of silt or clay, the flushing solution cannot move through the soil, and it cannot easily contact the contaminants; this reduces the effectiveness of the flushing process [63].
The research in the area for metal removal is still quite limited, particularly where metal removal is concerned. Even though organic and metal contaminations are major concerns, very few technologies can deal with both types of contaminants. As previously mentioned, surfactants can be used to assist in the remediation of numerous types of hydrocarbon contaminants. Only recently, it has been shown that surfactants can be used to enhance metal removal. In most cases, heavy metals are often present in soil as a component of mixed contamination with organics. This can cause serious limitations for treatment technologies. It was postulated that surfactants could enhance oil remediation by lowering the IFT between the oil and the soil; also, metals that are associated with the oil may be removed with oil simultaneously. The surfactant concentration is another important factor to be considered. It was found that metal removal efficiencies would increase linearly with the increasing surfactant concentration below the CMC, and remain relatively constant above the CMC [64]. However, for organic contaminants the solubility increased when a higher surfactant concentration above the CMC was used [65], as illustrated in Figure (5).
Important observations related to the performance of soil flushing technology are:
- Simplicity, minimal equipment requirements.
- Safety, neither personnel nor the public in the vicinity are exposed to contaminants.
- Cost-effectiveness requires low electrical energy leading to lower overall cost.
- Wide range of contaminants can be used for metals, organic compounds, or combinations of these contaminants (mixed contaminants, e.g., organic compounds combined with heavy metals).
- Hydrophobic contaminants require surfactants or organic solvents for their removal from the soil. Also, the successful application of surfactants to enhanced oil recovery has been demonstrated [66].
- Recovered groundwater may require treatment to meet the appropriate discharge standards [67].
- If soil flushing is used to extract VOCs, air emissions should also be treated [68].
- Flexibility can be used as an in-situ or ex-situ remediation system and is easily integrated with other remediation technologies such as bioremediation “treatment train” or stand-alone technology depending on site-specific cleanup objectives. As an in-situ technology, it eliminates the need to excavate, handle, and transport contaminated media.
- Low permeability or heterogeneous soils is difficult to treat [69]. Remediation times are usually lengthy because of the slowness of diffusion processes in the liquid phase.
- This technology requires hydraulic control to avoid the movement of contaminants off-site [70].
2.1.2.5. Guidelines for Surfactant Selection
Besides the site characteristics and contaminant speciation, selection of a surfactant for a specific application requires the evaluation of many inherent properties including solubility, toxicity, foamability (foam generation), biodegradability, turbidity, chemical stability, reactivity, wetting ability, and corrosivity [71]. Obviously economic considerations will dictate the preferred surfactant system [72]. The selection of surfactants was based on their origin (synthetic or chemically based materials and biosurfactants or biologically based materials), ability to solubilize specific contaminants, and on their environmental and health effects [73]. Some of the undesirable features of surfactant, such as sensitivity to highly saline brine, temperature, contaminant type, and retention, need to be considered when surfactants are selected for a given site.
The hundreds of different surfactants commercially available can be grouped into one of four categories: anionic, nonionic, cationic, and zwitterionic (consist of positive and negative moieties). Generally, only anionic and nonionic surfactants are acceptable for most flushing applications. Even within these two categories, there is hundreds of surfactant structures derived from synthetic and natural sources. Factors to consider in selecting surfactants include: prior application experience, potential effectiveness for the desired application, cost, public and regulatory perception, biodegradability and degradation products, toxicity to humans, animals, plants and ability to handle resulting extracted fluids.
Several factors can influence the efficiency of soil flushing with surfactants; groundwater that is too hard may be detrimental to the effectiveness of a surfactant [74]. Surfactants can adsorb onto clay fractions, reducing their availability. Too quick biodegradation can inactivate the surfactant although some degradability is required to avoid accumulation. Removal of the surfactant from the recovered water from flushing can be difficult and lead to high consumption rates. Selection of a surfactant solution is a process requiring laboratory batch and column studies and field trials. A considerable amount of laboratory work is required before implementation can commence [61]. Overall, the main factors should be considering for the surfactant selection in this process are cost, biodegradability, low toxicity, solubility at groundwater temperatures, effective at concentrations not higher than 3%, low soil dispersion, low adsorption to soil and low surface tension and low CMC [42]. If selected surfactant effective at concentrations higher than 3%, the used surfactant must be recovered and reused for the process to be economic.
Biodegradability: Many types of surfactants are considered toxic, biodegradable surfactants which could be degraded by microorganisms in the soil may be desirable. Several studies have shown that biodegradation was enhanced in the presence of surfactants, while in other cases inhibitions were induced or no effects were observed [75]. Such phenomena were found to be related to the nature of surfactants. Nonionic surfactants generally donate greater enhancement whereas anionic surfactants are inhibitorier [76]. Ideally, the surfactant would not destroy the microorganisms that have evolved in the contaminant site to metabolize the contaminants; further the residual surfactant would ideally act as a carbon source for the growing microbes and speed up the final bio-polishing of the site for residual contaminants.
Lower CMC: For some soil−contaminant systems the surfactants may perform similarly irrespective of the surfactant or concentration. In this case the lower CMC of nonionic surfactants may again be attractive candidates. Generally, increase in temperature decreases the CMC of some nonionic surfactants, but increases solubility of ionic surfactants. Salt addition reduces the CMC of ionic surfactant while those of nonionic are only slightly affected [77]. Both soil rollup and solubilization mechanisms were active during soil flushing, with the magnitude of each effect being a function of the extent of surfactant sorption and the soil grain size distribution. For a given soil-contaminant system if the soil rollup mechanism is significant and produces desirable results then the lower CMC of nonionic surfactants may render them economically preferred.
Turbidity: Where clay content was not very high, the nonionic surfactant solutions were clearly less turbid than the anionic surfactant solutions because the higher clay content lead to great cation exchange capacity. Upon appropriate alteration of conditions, such as temperature and additives, the micellar solutions become turbid due to the loss in aqueous solubility of surfactant molecules [54]. Micellar solutions of proper nonionic surfactants are homogeneous and isotropic at ambient temperature.
Low toxicity: Many surfactants are of low toxicity to humans but can affect animals and plants. The ingestion hazard to humans of many anionic or nonionic surfactants is low (e.g., swallowing residual dish soap is not harmful, and ingestion of larger amounts may result in diarrhea). However, some of these surfactants may be hazardous to aquatic life. For example, ethoxylated phenols (which have been used in remediation projects and are common in detergent formulations, including dish soap and laundry detergent) are toxic to fish but are not a significant environmental concern when used as household detergents because they readily degrade in biological wastewater treatment systems.
Foaming power (FP): It can be concluded that, the surfactant, which has the lowest foam height and the lowest foam stability, has a good detergency power. In general, the nonionic surfactants form unstable foams. The nonionic surfactants show foam volume and foam stability smaller than the anionic surfactants in aqueous medium, due to the larger surface area per molecule and the absence of highly charged films in these foams. If the rate of contaminant removal can be enhanced by foam generation, then the use of surfactants in soil remediation processes can be reduced. Foam can be created either by injection of air and surfactant solution simultaneously or alternately into porous media, so foam consists of thousands of tiny bubbles (foam containing 70–90% air). This technique can be used at large scale to enhance contaminated soil remediation of NAPLs by pulsed operation followed by aerobic biodegradation. Another advantage of using foam in soil treatment processes is that high volumes of air per unit volume of foam are injected into the soil [78].
If foam is to be used for remediation purposes, it must have sufficient stability to provide enough time for injection. In other words, the rate of bubbles collapsing must be much less than the generation rate. A variety of surfactants were evaluated for their ability to form foam before testing by soil injection. Many surfactants either were unable to generate foam or the stability of generated foam was very low.
Sorption to soil: The surface of the soil matrix provides a large sink for the loss of surfactant through adsorption of the surfactant on the soil [79]. Accumulation and adsorption of surfactant monomers on soil surfaces can result in significant reduction of the surfactants aqueous phase concentration. Sorption of surfactants to soil particle surfaces can have adverse effects on remediation processes. The formation of admicelles, which are like micelles but form as a layer on soil surfaces, can result in reductions in soil permeability [52]. Interactions between the hydrophilic and hydrophobic ends of surfactant monomers and charged soil surfaces affect the potential for adsorption [80].
Adsorption of anionic surfactants is not very significant because they are repelled by the negatively charged surfaces of soil particles. However, cationic surfactants are readily adsorbed to soil surfaces, nonionic surfactant which had less potential for sorption to soil media [53]. The sorption of nonionic surfactants to the soil was not affected by pH, while sorption of anionic surfactants increased as the pH decreased.
Solubility in groundwater: Besides selecting the surfactant to produce either a solubilization system or a mobilization system, the surfactant must also be matched to other the characteristics of the groundwater and the soil matrix in which the groundwater is found [81]. It is critically important that the surfactant be active at the temperature of the aquifer, a primary consideration is that groundwater can be expected to cold, temperatures below 10 ºC are common. Many aquifers also contain a height level of total dissolved solids (TDS), especially when the dissolved solids contain substantial concentrations of multivalent ions like calcium, magnesium, or multivalent iron any anionic surfactant will be susceptible to precipitation. However, conjunction with a nonionic surfactant can reduce precipitation and CMC values [82]. It is critical, therefore, to the potential for success in a surfactant-enhanced remediation project that the surfactant be known to be resistant to precipitation by the hardness ions present in the aquifer at the aquifer conditions.
Factors affecting treatment costs: The initial cost of the surfactant plays an important role in the overall cost of flushing method, and highly variable due to many factors. The main influencing factors include the site characteristics (specifically the size of the treatment area), contamination speciation (number of soil flushing cycles required), surfactant type and concentration (i.e. costs increase as surfactant dosages increase), other additives, scale of application, required treatment level, and regulatory factors [83]. Surfactant adsorbed at the soil/water interface, however, is not available either for making micelles to solubilize the NAPL, or for lowering the NAPL/water interfacial tension.
It is important to the over-all economics of the process that the surfactant be chosen to show a minimum amount of adsorption on the solid matrix. The cost of the surfactant makes it important that surfactant biodegrades at a sufficiently slow rate, the replacement of the lost surfactant not causes a major expense [76]. Laboratory work has also demonstrated applicability to NAPL-containing mineral oils, but it may be less cost-effective than alternate technologies for these contaminant groups [84].
In summary, the desirable surfactant system must show high hardness tolerance, low toxicity, biodegrade rapidly and low adsorption, higher solubilizing ability for contaminants and less sorption to soil [85]. These criteria must apply at the actual conditions in the aquifer, not just in the laboratory. So, nonionic surfactants were selected to flushing different soils contaminated with petroleum hydrocarbons.
Iturbe et al. [86] explored the use of nonionic surfactants for washing highly contaminated sandy soils, and the successful use of soil flushing techniques for remediation of oil-contaminated soils. Nonionic surfactant is applied to enhance the extraction of NAPL from soil [87]. In general, nonionic surfactants are commonly used for soil flushing since they are environmentally agreeable and cost effective. Furthermore, they possess lower CMC values, and have a lower tendency to flocculate clay particles in the soil compared to ionic surfactants, low toxicity, and low sorption onto the soil. The addition of nonionic surfactant to soil/water system increase desorption of PAH compounds, it was also observed that the desorption rate of PAH compounds depend on their molecular weight. The three and four ring PAH compounds showed higher and faster desorption rates than the five and six ring PAHs [88].
2.1.3. Soil Washing as Polishing Stage after Soil Flushing
In this technique, polluted soil is scrubbed by water through mechanical agitation to remove the hazardous contaminants or reduce their volume. Soil washing uses liquids (usually water) and it is often combined with other technologies. Soil washing belongs to the category of volume reduction techniques in which the contaminants are concentrated in a relatively small mass of material. Soil washing is cost-effective because it reduces the quantity of material that would require further treatment by another technology.
Important observations related to soil washing performance are:
- Complex waste mixtures require a combination of additives.
- Organics adsorbed onto clay particles are difficult to remove [89].
- Since soil washing does not destroy or immobilize the contaminants, the resulting soil must be disposed of carefully.
- Wash water needs to be treated before its final disposal.
- Soil washing is most effective for soil that does not contain a large amount of silt and clay.
- The average cost for this technology, depending on site-specific conditions and the target waste quantity and concentration [90].
Images and Tables
References
John Scullion, (2006): Remediating polluted soils. Naturwissenschaften 93, 51?65.
Winterhalder, K., (1996): Environmental degradation and rehabilitation of the landscape around Sudbury, a major mining and smelting area. Environ Rev 4, 185?224.
Helmisaari, H.S., Derome, J., Fritze, H., Nieminen, T., Palmgren, K., Salemaa, M., VanhaMajamaa, I., (1995): Copper in Scots pine forests around a heavy-metal smelter in south-western Finland. Water Air Soil Pollut 85:1727?1732.
Siebe, C., (1996): Heavy metal availability to plants in soils irrigated with wastewater from Mexico City. Water Sci Technol 32:29?34.
Collins, C., Laturnus, F., Nepovim, A., (2002): Remediation of BTEX and trichloroethene-current knowledge with special emphasis on phytoremediation. Environ Sci Pollut Res 9, 86?94.
vanBrummelen, T.C., vanGestel, C.A.M., Verweij, R.A., (1996): Long-term toxicity of five polycyclic aromatic hydrocarbons for the terrestrial isopods Oniscus asellus and Porcellio scaber. Environ Toxicol Chem 15, 1199?1210.
RAAG, (2000): Evaluation of risk based corrective action model, Remediation Alternative Assessment Group, Memorial University of Newfoundland, St John?s, NF, Canada.
Armishaw, R., Bardos, R.P., Dunn, R.M., Hill, J.M., Pearl, M., Rampling, T., Wood, P.A., (1992): Review of innovative contaminated soil cleanup processes. Warren Springs, Stevenage.
Reddy, K.R., (2008): Physical and chemical groundwater remediation technologies. C.J.G. Darnault (ed.), Overexploitation and Contamination of Shared Groundwater Resources. pp. 257- 274.
Khan, F.I., Tahir H., Ramzi H., (2004): An overview and analysis of site remediation technologies. Journal of Environmental Management 71, 95?122.
Reddy, K.R., Admas, J.F., Richardson, C., (1999): Potential technologies for remediation of Brownfield. Practice Periodical of Hazardous, Toxic, and Radioactive Waste Management 3(2), 61?68.
Semer, R., Adams, J.A., Reddy, K.R., (1998): An experimental investigation of air flow patterns in saturated soils during air sparging. Geotechnical and Geological Engineering, 16, 59?75.
Benner, M.L., Mohtar, R.H., Lee, L.S., (2002): Factors affecting air sparging remediation systems using field data and numerical simulations. Journal of Hazardous Materials 95(3), 305?329.
Lee, C.H., (2000): Site Assessment and In-Situ Remediation Technology Application for Contaminated Sites, Ph.D. Thesis, School of Earth and Environmental Sciences, Seoul National University, 198 pp.
Adams, J.A., Reddy, K.R., (2003): Extent of benzene biodegradation in saturated soil column during air sparging. Ground Water Monitoring and Remediation 23(3), 85?94.
NFESC (Naval Facilities Command Center) (2001): Final Air Sparging Guidance Document. Technical Report TR-2193-ENV. Prepared for NFESC by Battelle. August.Washington, DC 20374-5065.
Braida, W., Ong, S.K.., (2000): Influence of porous media and airflow rate on the fate of NAPLs under air sparging. Transp. Porous Media 38(1?2), 29?42.
Suthersan, S.S., (1999): In situ air sparging. Remediation Engineering: Design Concepts; CRC press LLC: Chapter 4.
Johnson, R.L., Johnson, P.C., McWhorter, D.B., Hinchee, R.E., Goodman, I., (1993): An overview of in situ air sparging, Ground Water Monitoring Review, Fall, 127?135.
Kram, M., (2001): Performance Comparison: Direct-Push Wells Versus Drilled Wells. NFESC Technical Report TR-2120-ENV. NFESC, Washington, DC.
Reddy, K.R., Adams, J.A., (2000): Effect of groundwater flow on remediation of dissolved-phase VOC contamination using air sparging. Journal of Hazardous Materials 72, 147?165.
Lee, D.H., Cody, R.D., Kim, D.J., Choi, S., (2002): Effect of soil texture on surfactant-based remediation of hydrophobic organic-contaminated soil, Environ. Int. 27, 681?688.
Reddy, K.R., Adams, J.A., (2001): Cleanup of chemical spills using air sparging, Chapter 14, in: Hand Book of Chemical Spill Technologies, M. Fingas, ed., Mc-Graw Hill Company, pp. 14.1?14.29.
Prince, R.C., (1998): Crude oil biodegradation. In: The Encyclopaedia of Environmental Analysis and Remediation, vol. 2. John Wiley, New York, pp. 1327?1342.
Kulik, N., Goi, A., Trapido, M., Tuhkanen, T., (2006): Degradation of polycyclic aromatic hydrocarbons by combined chemical pre-oxidation and bioremediation in creosote contaminated soil. J. Environ. Manage. 78, 382?391.
Ross, D., Marziarz, T.P., Bourquin, A.L., (1988): Bioremediation of Hazardous Waste Sites in the USA: Case Histories. In: Superfund 88, Hazardous Materials Control Research Institute, Silver Spring, MD, pp. 395-397.
Harper, B.M., Stiver, W.H., Zytner, R.G., (2003): Non-equilibrium nonaqueous phase liquid mass transfer model for soil vapor extraction systems. Journal of Environmental Engineering 129(8), 745?754.
Hinchee, R., (1989): Enhanced Biodegradation Through Soil Venting. Presented at Workshop on Soil Vacuum Extraction held at US. EPA Robert S. Kerr Environmental Research Laboratory, Ada, OK, April 27-28 (Dominic DiGiulio, Technical Coordinator).
USEPA (United State Environmental Protection Agency), (1995a): How to Evaluate alternative cleanup technologies for underground storage tank sites. Office of Solid Waste and Emergency Response, US Environmental Protection Agency. Publication # EPA510-B-95-007, Washington, DC.
Kirtland, B.C., Aelion, C.M., Widdowson, M.A., (2001): Long-term AS/SVE for petroleum removal in low-permeability Piedmont saprolite. J. Environ. Eng.-ASCE. 127(2), 134?144.
Kirtland, B.C., Aelion, C.M., (2000): Petroleum mass removal from low permeability sediment using air sparging/soil vapor extraction: impact of continuous or pulsed operation. Journal of Contaminant Hydrology 41, 367?383.
Tomlinson, D.W., Thomson, N.R., Johnson, R.L., Redman, J.D., (2003): Air distribution in the Borden aquifer during in situ air sparging. J. Contam. Hydrol., 67 (1-4), 113-132.
Chaineau, C.H., Yepremian, C., Vidalie, J.F., Ducreux, J., Ballerini, D., (2003): Bioremediation of a crude oil-polluted soil: biodegradation, leaching and toxicity assessments. Water, Air, and Soil Pollution 144(1?4), 419?440.
Marley, M., Hazenbronck, D.J., and Walsh, M.T., (1992): The application of in situ air sparging as an innovative soils and groundwater remediation technology. Ground Water Monitoring Review. Vol. 12, No. 2, pp. 137-145.
Gannon, O.K., Bibring, P., Raney, K., Ward, J.A., Wilson, D.J., Underwood J.L., Debelak, K.A., (1989): Soil clean up by in-situ surfactant flushing. III. Laboratory results. Separation Sci. and Technol. 24, pp. 1073?1094.
Couto, H.J.B., Guilio Massarani, Evaristo, C., Biscaia, Jr., Geraldo L., Sant?Anna Jr., (2009): Remediation of sandy soils using surfactant solutions and foams. Journal of Hazardous Materials 164, 1325?1334.
Urum, K., Pekdemir, T., ?opur, M., (2004): Surfactants treatment of crude oil contaminated soils. Journal of Colloid and Interface Science 276, 456?464.
Myers, D., (1999): Surfaces, Interfaces, and Colloids: Principles and Applications, third ed. Wiley-Vch, New York.
Duggan, J.W., Bruell, C.J., Ryan, D.K., (1994): In situ emulsification and mobilization of gasoline range hydrocarbons using surfactants. J. Soil Contam. Vol. 3, No. 2, pp: 159-182.
West, C.C., Harwell, J.F., (1992): Surfactant and subsurface remediation. Environ. Sci. Technol. 26, 2324?2330.
Rosen, M.J., (1989): Surfactant and Interfacial Phenomena. Wiley, New York, pp. 170?202.
Mulligan, C.N., Yong, R.N., Gibbs, B.F., (2001): Surfactant-enhanced remediation of contaminated soil: a review. Eng. Geol. 60, 371?380.
Venditti, F., Angelico, R., Ceglie, A., Ambrosone, L., (2007): Novel surfactant based adsorbent material for groundwater remediation. Environ. Sci. Technol., 41, 6836.
Barathi, S., Vasudevan, N., (2001): Utilisation of petroleum hydrocarbons by Pseudomonas fluorescens isolated from a petroleum-contaminated soil. Environ. Int. 26 (5?6), 413?416.
McCray, J.E., Bai, G., Maier, R.M., Brusseau, M.L., (2001): Biosurfactant-enhanced solubilisation of NAPL mixtures. J. Contam. Hydrol. 48 (1?2), 45?68.
Mulligan, C.N., Eftekhari F., (2003): Remediation with surfactant foam of PCP contaminated soil, Eng. Geol. 70, 269?279.
Palmer, C.D., Fish W., (1992): Chemical enhancements to pump-and-treat remediation. Ground Water Issue January, 1?19.
Park, S.W., Lee, J.Y., Yang, J.S., Kim, K.J., Baek, K., (2009): Electrokinetic remediation of contaminated-soil with waste-lubricant oils and zinc. Journal of Hazardous Materials. 169 (1-3), 1168-1172.
Baek, K., Kim, D.H., Park, S.W., Ryu, B.G., Bajargal, T., Yang, J.S., (2009): Electrolyte conditioning-enhanced electrokinetic remediation of arsenic-contaminated mine tailing, Journal of Hazardous Materials 161, 457-462.
NRC (National Research Council) (1994): Alternatives for ground water cleanup, National Academy Press, Washington, DC, ISBN 0-309-04994-6.
Cheng, H.F., Sabatini, D.A., (2007). Separation of organic compounds from surfactant solution: a review. Separation Science and Technology, 42(3), 453?475.
Abdul, A.S., Gibson, T.L., Rai, D.N., (1990): Selection of surfactants for the removal of petroleum products from shallow sandy aquifers. Ground Water 28, 920?926.
Cheah, E.P.S., Reible, D.D., Valsaraj, K.T., Constant, W.D., Walsh, B., Thibodeaux, L.J., (1998): Simulation of soil washing with surfactants. J. Hazard. Mater. 59 (2?3), 107?122.
Rosen, M.J., (2004): Surfactants and Interfacial Phenomena. New York: John Wiley & Sons.
Clayton, W., (1935): The Theory of Emulsions and Their Technical Treatment, 3rd Edition. P. Blakiston's Son & Co. Inc., Philadelphia.
Schramm, L.L., (1992): Petroleum Emulsions: Basic Principles. In Emulsions Fundamentals and Applications in the Petroleum Industry. Edited by L.L. Schramm. American Chemical Society, Washington, DC. pp: 1-49.
Lake, L.W., (1989): Enhanced Oil Recovery. Prentice-Hall, Engelwood Cliffs, N.J.
Wilson, J.L., Conrad, S.H., Mason, W.R., Peplinski, W., Hagan, E., (1990): Laboratory Investigations of Residual Liquid Organics from Spills, Leaks, and Disposal of Hazardous Wastes in Groundwater. EPA/600/6-90/004, U.S. EPA, U.S. Government Printing Office, Washington, D.C.
Powers, S.E., Abriola, L.M., Weber W.J., Jr., (1994): An experimental investigation of NAPL dissolution in saturated subsurface systems: transient mass transfer rates. Water Resourc. Res. 30, 321?332.
Lee, M., Kang, H., Do, W., (2005): Application of nonionic surfactant-enhanced in situ flushing to a diesel contaminated site. Water Research 39, 139?146.
ITRC (Interstate Technology & Regulatory Council) (2003): Technical and regulatory guidance for surfactant/cosolvent flushing of dnapl source zones. DNAPL-3. www.itrcweb.org/Documents/DNAPLs-3.pdf
Juhasz, A.L., Smith, E., Smith, J., Naidu, R., (2003): In situ remediation of DDT-contaminated soil using a two-phase co-solvent flushing fungal biosorption process. Water, Air, and Soil Pollution 147(1?4), 263?274.
Reddy, K.R., Saichek, R.E., (2003): Effect of soil type on electrokinetic removal of phenanthrene using surfactants and co-solvents. Journal of Environmental Engineering 129(4), 336?346.
Doong, R.A., Wu, Y.W., Lei, W.G., (1998): Surfactant enhanced remediation of cadmium contaminated soils. Water Sci. Technol. 37, 65?71.
Liu, Z., Laha, S., Luthy, R.G., (1991): Surfactant solubilization of polycyclic aromatic hydrocarbon compounds in soil? water suspensions. Water Sci. Technol. 23, 475?485.
Oma, K.H., Clarke, A.N., Mutch, R.D., Jr., Wilson, D.J., (1991): Surfactant flushing/washing: Design and preliminary testing of an innovative process for recovery and recycle of surfactant, Paper presented at the I and EC Special Symposium, American Chemical Society, Atlanta, GA, October.
Otterpohl, R., (2002): Options for alternative types of sewerage and treatment systems directed to improvement of the overall performance. Water Science and Technology 45(3), 149?158.
CPEO, (1998d): Soil flushing. Center for Public Environmental Oversight. 425 Market Street, San Francisco, CA,
View ArticleFRTR, (1999h): Soil flushing. (Federal Remediation Technologies Roundtable). USEPA, 401 M Street, S.W., Washington, DC,
View ArticleJohnston, C.D., Fisher, S., Rayner, J.L., (2002): Removal of petroleum hydrocarbons from the vadose zone during multi-phase extraction at a contaminated industrial site, IAHS-AISH Publication, number 275.
Admas, C.D., Spitzer, S., Cowan, R.M., (1996): Biodegradation of nonionic surfactants and effects of oxidative pretreatment. J. Environ. Eng. 122, 477?483.
Deshpande, S., Shiau, B.J., Wade, D., Sabatini, D.A., Harwell, J.H., (1999): Surfactant selection for enhancing ex situ soil washing. Water Resource 33 (2), 351?360.
Urum, K., Pekdemir, T., Ross, D., Grigson, S., (2005): Crude oil contaminated soil washing in air sparging assisted stirred tank reactor using biosurfactants. Chemosphere 60, 334?343.
AAEE (American Academy of Environmental Engineers), (1993): In: Anderson, W.C. (Ed.), Soil washing/soil flushing, Innovative Site Remediation, vol. 3, WASTEC.
Deschenes, L., Lafrance, P., Villeneuve, J.P. and Samson, R., (1996): Adding sodium dodecyl sulfate and Pseudomonas aeruginosa UG2 biosurfactants inhibits polycyclic aromatic hydrocarbon biodegradation in a weathered creosote-contaminated soil. Appl. Microbiol. Biotechnol. 46, pp. 638?646.
Rouse, J.D., Sabatini, D.A., Suflita, J.M., Harwell, J.H., (1994): Influence of surfactants on microbial degradation of organic compounds. Crit. Rev. Environ. Sci. Technol. 24, 325?370.
Urum, K., Pekdemir T., (2004): Evaluation of biosurfactants for crude oil contaminated soil washing. Chemosphere 57, 1139?1150.
Rothmel, R.K., Peters, R.W., Martin, E.S., Deflaun, M.F., (1998): Surfactant foam/bioaugmentation technology for in situ treatment of TCE-DNAPLs. Environ. Sci. Technol. 32, 1667?1675.
Rouse, J.D., Sabatini, D.A., Harwell J.H., (1993): Minimizing surfactant losses using twin-head anionic surfactants in subsurface remediation. Environmental Science and Technology 27(10), 2072?2078.
Brickell, J.L., Keinath, T.M., (1991): The Effect of Surfactants on the Sorption Partition Coefficients of Napthalene on Aquifer Soils. Water Sci. Tech., Vol. 23, pp: 455-463.
Sabatini, D.A., Lipe, K.M., Hasegawa, M.A., Harwell, J.H., (1996): Micellar-enhanced ultrafiltration and air stripping for surfactant contaminant separation and surfactant reuse. Ground Water Monit. Rem. 16, 85?92.
Sabatini, D.A., Knox, R.C., Harwell, J.H., (1995): Emerging technologies in surfactant-enhanced subsurface remediation. In: Sabatini, D.A., Knox, R.C., Harwell, J.H. (Eds.), Surfactant-Enhanced Subsurface Remediation, Emerging technologies. ACS Symposium Series 594. American chemical Society, Washington DC, pp.1-9.
Lowe, D.F., Oubre, C.L., Ward, C.H., (2000): Reuse of surfactants and cosolvents for NAPL remediation. Lewis Publishers, Boca Raton.
Alter, S.R., Brusseau, M.L., Piatt, J.J., Ray, M.A., Wang, J.M., Cain, R.B., (2003): Use of tracer tests to evaluate the impact of enhanced solubilization flushing on in-situ biodegradation. Journal of Contaminant Hydrology 64(3?4), 191?202.
Ahn, C.K., Kim, Y.M., Woo, S.H., Park, J.M., (2008): Soil washing using various nonionic surfactants and their recovery by selective adsorption with activated carbon. Journal of Hazardous Materials 154, 153?160.
Iturbe, R., Flores, C., Castro, A., Torres L.G., (2007): Sub-soil contamination due to oil spills in six oil pipeline pumping stations in northern Mexico, Chemosphere, 68, 893-906.
Kujawski, W., Koter, I., Koter, S., (2009): Membrane-assisted removal of hydrocarbons from contaminated soils - laboratory test results. Desalination 241, 218-226.
Ahn, C.K., Kim, Y.M., Woo, S.H., Park, J.M., (2007): Selective adsorption of phenanthrene dissolved in surfactant solution using activated carbon. Chemosphere 69, 1681?1688.
CPEO, (1997): Soil washing. Center for Public Environmental Oversight.425 Market Street, San Francisco,CA, wash.html
View ArticleFRTR, (1999a): Soil washing. (Federal Remediation Technologies Roundtable). USEPA, 401 M Street, S.W. Washington,DC,
View Article
