: G.T. Kleinheinz
E-MAIL: kleinhei@uwosh.edu
Seasonal Impacts on Indoor Fungal Concentrations After Mold Remediation
Corresponding Author
Affiliation
G.T. Kleinheinz* and A. Campbell
Environmental Engineering Technology University of Wisconsin – Oshkosh
800 Algoma Boulevard,Oshkosh, WI 54901,Phone – (920) 424-1100,Fax – (920) 424-0832
Article Reviewed By:
Mohamed Fathi(mohamed.fathi@vet.au.edu.eg)
Man Xiao(xiao_man1989@163.com)
Miha Humar(Miha.Humar@bf.uni-lj.si)
Jung-Wan Kim(kjw5864@inu.ac.kr)
Citation
Greg Kleinheinz, Seasonal Impacts on Indoor Fungal Concentrations After Mold Remediation(2017)SDRP Journal of Earth Sciences & Environmental Studies 2(1)
Abstract
Fungal contamination of the indoor air environment is a concern for building owners, residents, landlords, realators, and environmental health professionals. These health effects can vary greatly from person-to-person. These potential health effects are not just related to the concentration of fungi, but also the type of fungi present. There are a number of methods for enumerating indoor fungi, but the use of non-viable Air-O-Cell cassettes has become a common, standardized, and well established method for air sampling during or after mold remediation. The overarching objective of this study was to evaluate the impact of time of year on Air-O-Cell fungal testing results after professional remediation. This project relied on data from 138 sampling events that occurred over the course of several years. The data collected in outdoor samples during the summer months revealed much higher concentrations of fungi when compared to indoor air samples. However, in winter months the outdoor samples were found to have very low concentrations of fungi relative to indoor samples. The mean concentration of fungi in each sample location varied greatly with respect to season of the year. Thus, absolute concentrations of fungi should only be one piece of information used to assess a succesful remediation. Additional information to be considered includes a visual inspection of the remediated site, the season, the relationship of fungal type between sample locations, and the proportion of various fungal types in each sample.
Introduction
Fungal contamination of the indoor air environment is a concern for building owners, residents, landlords, realators, and environmental health professionals. While fungi are ubiquitous in the environment, the indoor air environment should not contain fungal concentrations that are above outdoor concentrations. Some have recommended concentrations of fungi with an upper limit range 200 CFU (Colony Forming Unit)/m3 to > 1000 CFU/m3 (Morey et al. 1984; and Godish 1991) with people reporting concentrations that they consider normal of 1-6,000 CFU/m3 in residential homes (Soloman et al. 1984). This is a substantial range of concentration in light of the fact that elevated levels of fungi can be associated with various adverse health effects (Kuhn and Ghannoum 2003). These health
effects from excessive fungi in air can vary greatly from person-to-person due to differences in age, medical condition, fungal concentration, etc.. These potential health effects are not just related to the concentration of fungi, but also the type of fungi present within the overall concentration. The genus or group of fungi is important due to allergenic effects and the compounds the organisms may produce (i.e., mycotoxins). Thus, adverse effects may be due to allergic reactions to fungi present, portions of fungi that have been lysed or killed in some manner, and by extracellular compounds that may be produced by the fungi.
While there are many methods and equipment used for indoor air sampling from Anderson N6 impaction menthods (Godish and Godish 2008) and qPCR enumeration techniques (Reponen et al 2010), the use of non-viable cassettes is a common, standardized, and well established method for air sampling in mold remediation (AIHA 2001, Godish and Godish 2008, Codina et al 2008, and Zefon International 2016). In the authors experience it has been used in 95% of indoor air quality investigations at the request of remediation contractors or clients due to the speed of analysis and lack of growth bias. Conversely users are giving-up the ability to speciate fungi to species level as the morphological similarities make this impossible in non-viable impaction microscopic analysis (i.e.,Apergillus/Penicillium). Non-viable impaction analysis provides results in spores per meter cubed based on sampling time and pump flow rate (Zefon 2016).
Each building will have its own ‘background’ fungal concentrations. This background concentration of fungi is determined by the building age, location, construction type, and maintenance. Confounding the understanding of this background concentration of fungi is the fact that outdoor concentrations fluxuate throughout the year are highly dependent on weather conditions. Additionally, in the northern part of the United States snow cover can greatly reduce outdoor concentrations of fungi during the winter. Thus, when a mold remediation project is completed a contractor or microbiologist typically not only looking at absolute concentrations of fungi, but also the ratio of fungi between the complaint, non-complaint, and outdoor samples. Again confounding this analysis is the high degree of variability in northern areas of the United States due to seasonality and the variability in housing construction. Thus, recommendations on fungal levels above or below some threshold concentration may not be a good indicator of remediation success or what the levels in that particular location would be ‘normal’.
Numerous groups have proposed levels for fungi to reach in order to deem remediation a success (Rao et al. 1996). However, many of these recommendations are based either on consensus, or on data obtained from a limited sample area, or type of location. It is evident that more data is needed to help answer questions regarding what normal fungi levels are and what these levels should be after a MRC (mold remediation contractor) has completed mitigating an elevated fungal problem.
Several groups have proposed protocols and standard guidelines for mold remediation and testing of remediation (AIHA 2001; Binnie 1990; Federal-Provincial Committee on Environmental and Occupational Health 1995; Indoor Air Quality Association 1995; Malmberg 1991; Reynolds 1990; USEPA 2001; USOSHA 1994; World Health Organization 1989) efficacy. However, these recommendations are generally based upon survey studies of ‘normal’ levels of fungi in homes and not on what is found after a remediation project is complete. There does appear to be consensus that any fungal testing after a remediation project needs to include a sampling of the complaint area (area with the elevated mold levels), a non-complaint area in the same building, and an outdoor sample for comparison (AIHA, 2001, USEPA 2001 Indoor Air Quality Association 1995, Wisconsin State Laboratory of Hygiene 2016).
The overarching objective of this study was to evaluate the impact of time of year on non-viable impaction fungal testing results after professional remediation. Specifically, residential homes that underwent professional mold remediation were sampled using non-viable sampling cassettes using the generally accepted practice of comparisons between complaint, non-complaint, and outdoor samples. Additionally, the sampling events were seperated into seasons to determine if seasonality plays a role in how we should interprit these results between these samples.
Materials & Methods
Sample Site Selection/Strategy
All locations sampled were single family residences in Wisconsin, USA. Each location was selected by a mold remediation contractor due to a significant water intrusion event (leaking pipes, leaking rook, etc.) and contact with the owners. Each site had at least 6 m2 of visible mold present before remediation. At each location a complaint, non- complaint, and outdoor location were identified for sampling. The complaint area was characterized as impacted by excessive fungal growth prior to remediation. The non- complaint area was an area of the same building that was not connected via ducting, hallways, etc. to the complaint area. The non-complaint area was not affected by the amplification event found in the complaint area, such as a water leak. The outdoor sample was collected within 10 meters of the entrance/exit of the building. All samples were collected in sequence with one right after the other and always within 30 minutes of each other. Samples were collected at a height of 1 meter using an aluminum tripod.
Non-Viable Impaction Sampler
The Air-O-Cell sampling cassette is one of the industry leaders in non-viable impaction fungal samplers that uses an adhesive slide to collect fungal elements at the inlet (Zefon). All Air-O-Cell cassettes were obtained from Zefon International, St. Petersburg, FL. A flow rate of 15L min-1 was used (Zefon International, 2016). The air velocity at the inlet is approximately 299 feet per minute (Zefon International, 2016). The air flow rates were verified prior to each use by a digital flow meter (Alicat Scientific, Tucson, AZ). After sampling for 5 minutes, or the equivalent of 75.0 L, the Air-O-Cell cassettes were removed from the sampler and placed in individual sterile bags. The cassettes were then sent to the Wisconsin State Laboratory of Hygiene in Madison, WI for fungal spore counts and identifications following manufacturer recommended procedures (Zefon International 2016). In short, the sample is preapred for analysis by opening the cassette and removing the glass collection slide. The collection slide is placed sample side up onto a clean microscope slide. The sample slide is then affixed to a clean microscope slide. A drop of lacto-phenol cotton blue stain solution is placed on the sample trace and covered by a glass cover slip. The limit of detection (LOD) for Air-O Cell samples was 4 fungal spores per slide. The Air-O-Cell method has the advantage of speed of analysis over culture-based methods, but it is limited to morphological identification of fungi and their components which limits the identification of many fungi to genus level. Furthermore, fungal components are also reported by the laboratory as they can have impacts on allergy symptoms. Even with these limitations, the ease of use, relatively low cost, and rapid results make it the choice of most mold remediation contractors and thus is used as the focus of this study. All air sampling was conducted with a Zefon Z-Lite IAQ Pump. The pump was calibrated before every sampling event using a Model 4046 TSI Primary Flow Pump Calibrators (Zefon Inc.).
Statistical and graphical analysis
Statistical analysis was performed with Systat 11.0. Graphs were generated with Microsoft Excel 2011. Significance of analysis was set at the alpha<0.05.
Results
This project relied on data from 138 sampling events that occurred over the course of several years. Each sampling event was conducted within a 30 minute time (i.e., complaint, non-complaint, and outdoor reference samples) while visiting the remediated site. Due to changes in seasonal ground cover (i.e., snow), precipitation, temperature, etc. results were evaluated on an annualized basis and in three month intervals with the intervals representing the most closely related months (i.e., December, January, and February are the coldest months with snow cover).
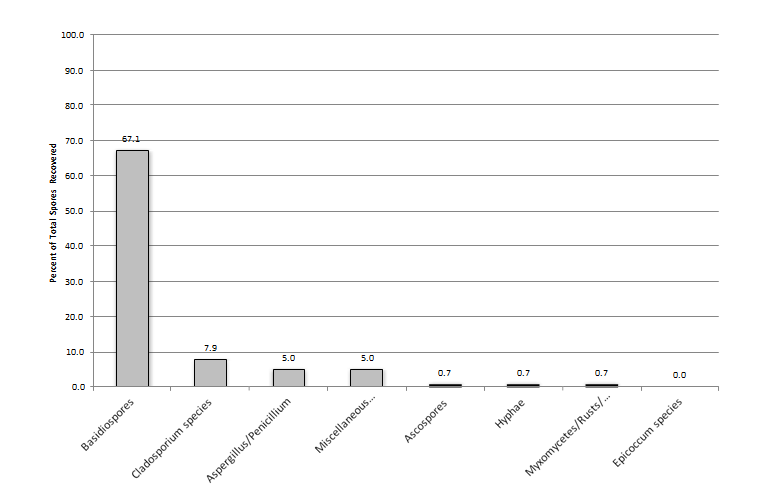
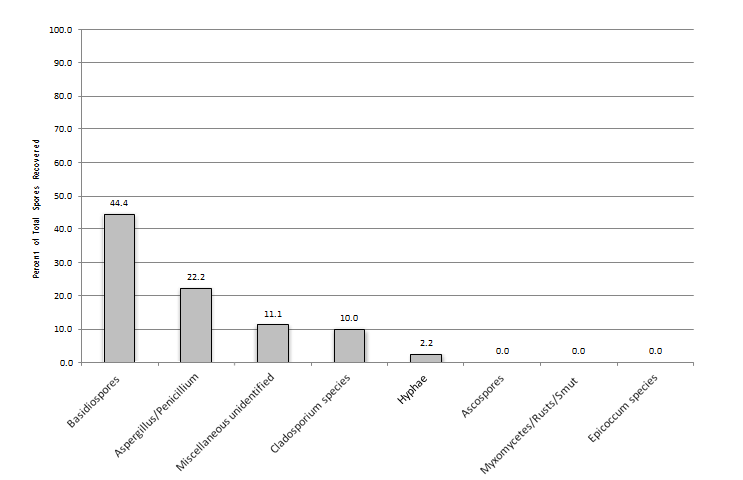
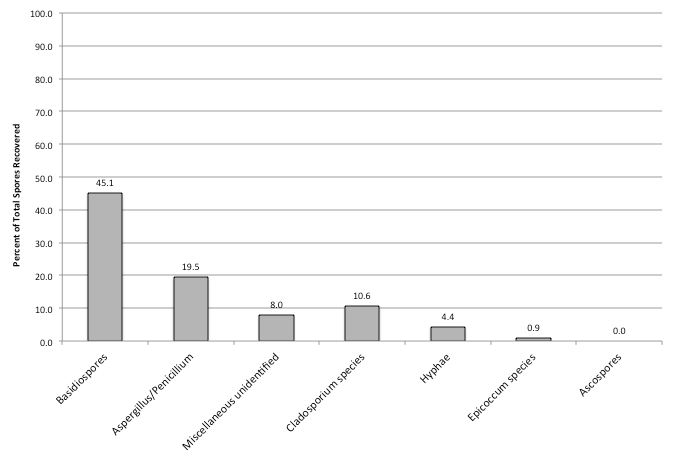
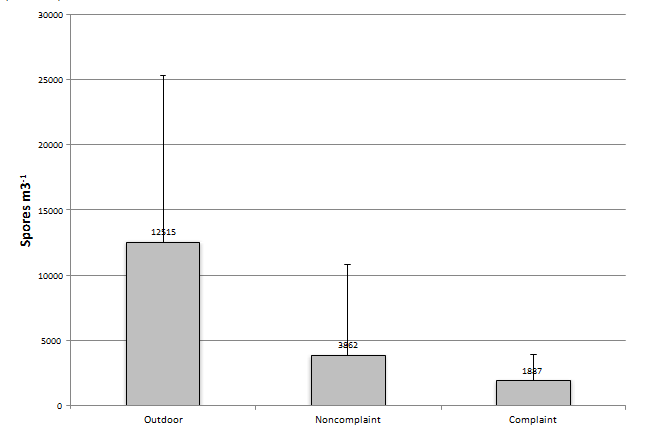
When one looks at all samples collected throughout a year and the percent of spores recovered per individual sample, basidiospores were the most prevalent group in all samples with it comprising 67, 44, and 45 percent of total spores recovered in the outdoor, non-complaint, and complaint samples, respectively (Figures 1-3). Cladosporium was the most prevalent genus in the outdoor samples making-up 8% of the total spores found during the sampling event. This was followed by the Aspergillus/Penicillium group at 5% of the total spores found during the sampling event. Interestingly, both the non-complaint and complaint samples showed Aspergillus/Penicillium as the second most prevalent fungal group at 22% and 20%, respectively. The total spores recovered was greatest in the Outdoor samples with a mean of 12,515 spores/m3 (SD ± 12,914). The non-complaint mean was determined to be 3,862 spores/m3 (SD ± 6,910) and this was followed by the remediated complaint samples at 1,887 spores/m3 (SD ± 2,023) (Figure 4). When comparing the samples collected withing each seasonal grouping (Winter = December-Feb., Spring = March- May, Summer=June-Aug, and Fall=Sept.-Nov) with the other groupings, the complaint and non-complaint samples were found to be significantly different than the outdoor sample (p<0.001) in all groups of data. However, the complaint and non-complaint samples were not significantly different (p=0.183) in all sample groups.
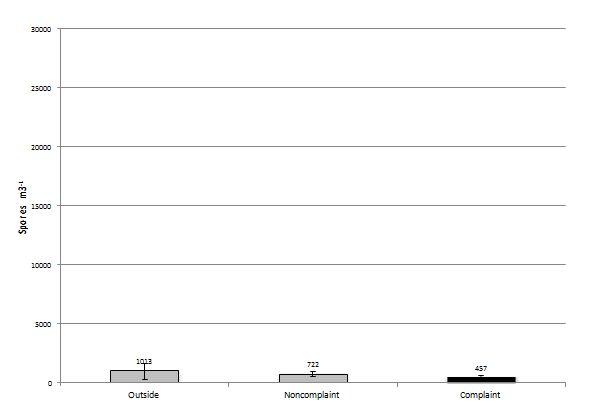
When the sampling events are broken down by three month intervals a different pattern emerges. There are different number of sampling events in each group due to the seasonal effects on the demand for mold remediation. That is, winter and spring have less remediation events than summer and fall. However, the groups represent different weather confounfing factors such as rainfall, temperature, and snow cover. In the December to February group (N=12 or number events in this group) of samples the number of recovered fungal elements drops to 1,013 spores/m3 in the outside sample, 722 spores/m3 in the non-complaint samples, and 457 spores/m3 in the complaint sample (Figure 5). When you look at the most prevalent group of fungi in each outdoor sample event and Aspergillus/Penicillium is found to be the most prevalent (N=2) it comprises 26% of the total, when Cladosporium is the most prevalent (N=2) it makes-up 26% of the total and when it is basidiospores (N=2) it makes-up 17% of the total found during the sampling event. Miscelaneous unidentified made up the most prevalent group in 25% of the samples or N=4. When the most prevalent group of fungi in each non-complaint sample event is Aspergillus/Penicillium (N=5) it is found to be at 26% of the total. When it is Cladosporium (N=3) it is found to make-up 26% of the total spores found during the sampling event and when basidiospores are the most prevalent (N=2) it makes-up 17% of the total spores present. Finally, when the most prevalent group of fungi in each complaint sample event is Aspergillus/Penicillium (N=1) it was found to be at 27% of the total found during the sampling event. When it is Cladosporium (N=1) it is found to make-up 15% of the total spores and when Miscelaneous unidentified (N=7) is the most prevalent it makes-up 42% of the total spores present. When comparing the samples collected on each date with each other, the non-complaint samples were found to be significantly different than the outdoor samples (p<0.05). However, the complaint samples were not signifcantly different than the non-complaint samples (p=0.304) or the outdoor sample (p=0.383).
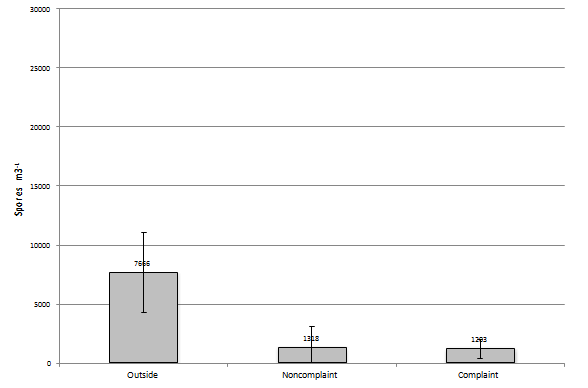
In the March to May group (N=26 or number events in this group) of samples the number of recovered fungal elements increases to 7,666 spores/m3 in the outside sample, 1,318 spores/m3 in the non-complaint samples, and 1,203 spores/m3 in the complaint sample (Figure 6). When you look at the most prevalent group of fungi in each outdoor sample event and basidiospores (N=20) are found to be the most prevalent they comprise 72% of the total found during the sampling event, when Miscelaneous unidentified are the most prevalent (N=3) it makes-up 65% of the total found during the sampling event. When the most prevalent group of fungi in each non-complaint sample event is Aspergillus/Penicillium (N=6) it is found to be at 32% of the total found during the sampling event. When it is Cladosporium (N=2) it is found to make-up 33% of the total spores and when basidiospores are the most prevalent (N=2) it makes-up 48% of the total spores present. Finally, when the most prevalent group of fungi in each complaint sample event is Aspergillus/Penicillium (N=7) it is found to be at 35% of the total. When it is basidiospores (N=7) it is found to make-up 26% of the total spores and when Miscelaneous unidentified (N=7) is the most prevalent it makes-up 19% of the total spores present. When comparing the samples collected from each sampling event within this group with each other, the complaint and non-complaint samples were found to be significantly different than the outdoor sample (p<0.05). However, the complaint and non-complaint samples were not significantly different (p=0.383).
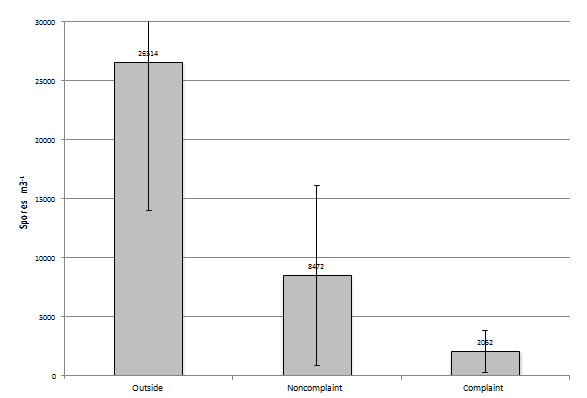
In the June to August group (N=44 or number events in this group) of samples the number of recovered fungal elements increases to 26,514 spores/m3 in the outside sample, 8,472 spores/m3 in the non-complaint samples, and 2,052 spores/m3 in the complaint sample (Figure 7). When you look at the most prevalent group of fungi in each outdoor sample event and Cladosporium (N=29) are found to be the most prevalent they comprise 61% of the total, when Ascospores are the most prevalent (N=10) it makes-up 69% of the total. When the most prevalent group of fungi in each non- complaint sample event is basidiospores (N=17) it is found to be at 60% of the total. When it is Cladosporium (N=14) it is found to make-up 42% of the total spores and when Ascospores are the most prevalent (N=12) they make-up 58% of the total spores present. Finally, when the most prevalent group of fungi in each complaint sample event is Cladosporium (N=12) it is found to be at 67% of the total. When it is basidiospores (N=12), they are found to make-up 59% of the total spores present. When comparing the samples collected from each sampling event within this group with each other, the complaint and non-complaint samples were found to be significantly different than the outdoor sample (p<0.001). However, the complaint and non-complaint samples were not significantly different from one another (p=0.941).
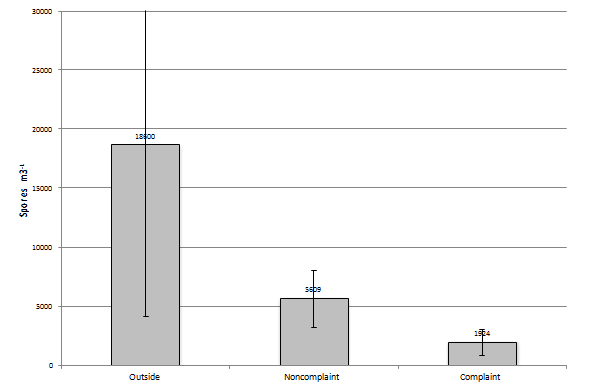
In the September to November group (N=41 or number events in this group) of samples the number of recovered fungal elements decreases to 18,600 spores/m3 in the outside sample, 5,609 spores/m3 in the non-complaint samples, and 1,924 spores/m3 in the complaint sample (Figure 8). When you look at the most prevalent group of fungi in each outdoor sample event and basidiospores (N=34) are found to be the most prevalent they comprise 67% of the total, when Cladosporium is the most prevalent (N=5) it comprises 88% of the total, and when ascospores are the most prevalent (N=2) it makes- up 40% of the total found during the sampling event. When the most prevalent group of fungi in each non-complaint sample event is basidiospores (N=15) it is found to be at 62% of the total found during the sampling event. When it is Cladosporium (N=8) it is found to make-up 62% of the total spores and when ascospores are the most prevalent (N=7) they make-up 60% of the total spores present. Finally, when the most prevalent group of fungi in each complaint sample event is basidiospores (N=23) it is found to be at 70% of the total. When it is Cladosporium (N=9), they are found to make-up 56% of the total spores present. When comparing the samples collected from each sampling event within this group with each other, the complaint and non-complaint samples were found to be significantly different than the outdoor sample (p<0.05) and (p<0.001). However again, the complaint and non-complaint samples were not significantly different from one another (p=0.065).
Discussion
When a professional is called to evaluate the success of a mold remediation activity there are a number of different ways that one can go about this evaluation.
Generally, an air test is conducted after remediation and a environmental health professional uses these results to help assess if the remediation has been a success. There are no Threshold Limit Values (TLV) or Permissible Exposure Limits (PEL) for fungal concentrations in air. It would be difficult to assign such numbers as the type of fungi, environment, and physiological state of each human plays a major role in how fungi affect humans. For this reason professionals usually rely on a relationship of fungi in complaint, non-complaint, and outdoor samples to assist in their determination of success (or not). Generally, successful remediation projects are characterized by fungal concentrations in the remediated complaint that are at the same level or less than the non- complaint area. Furthermore, in the spring, summer, and fall the outdoor contains fungi concentrations that are greater than the indoor samples. The exception is during rain events or during months of snow cover in winter climates. On an annualized basis and perhaps in areas with constant environmental conditions we see that the outdoor sample contains the highest concentration of fungi when compared to any indoor samples. This is plausible as the building envelope acts as a filter of sorts for the fungal spores moving in and out of homes. In the case of this study we are dealing with only successful remediation events. Of the indoor samples, the complaint sample contained the lowest concentrations of fungi. This is also reasonable as this is the area that has been subject to cleaning and HEPA filtration of air during the clean-up. It would be reasonable to expect that the complaint area would have the highest concentration of fungi, or at least different proportions of fungi prior to remediation (Kleinheinz, data not presented 2016). In the Wisconsin climate investigated Basidiospores were the most prevalent fungal group found in the Air-O-Cell samples. This was followed by Cladosporium in the outdoor sample and Aspergillus/Penicillium in the indoor samples (complaint and non-complaint). These results are analogous to previous work described by (Codina et al., 2008 and Fairs et al 2008).
Conclusion
While annually these relationships appear to be consistent with previous work (Kleinheinz et al, 2006, Barnes et al., 2007), when one compares results from different seasons of the year a new pattern can emerge. This same phenomenon was alluded to previously by other researchers (LeBouf, et al. 2008). In this study that pattern held true for months March through November, but when the December through February data was analyzed a new pattern arises. In the months of December through February the outdoor and complaint samples had the lowest concentrations of fungi with the non- complaint as the highest concentration. All samples were relatively low when compared with other times of the year. Again, this does seem plausible as these are the months with the most signififcant snow cover in the upper midwest.
The mean concentration of fungi in each sample location varied greatly with respect to season of the year. Again, this is a plausible outcome due to changes in temperature, precipitation, snow cover, etc. in the upper midwest. Having said that, mold remediation professionals should be aware of these changes in concentrations of fungi and changes in the relationship of concentrations between complaint, non-complaint, and outdoor samples. Only by coupling a visual inspection of the remediation area and having an understanding of these ‘plausible concentrations’ of fungi with respect to the impact of season, can an environmental health professional make a truly informed decision on the success of mold remediation activity. This approach confirms the opions of previous researchers that help professional judgement should couple with fungal air testing resuts to arrive at an opinion as to remediation success (Johnson et al. 2008).
While this study did not evaluate the use of surface sampling methods, such as tape lifts, these methods do not provide significant value in the evaluation of the remediation success due to sample and surface variabilities in building materials. That is the samples are not ‘before’ and ‘after’ samples of the same locations and while sometimes they can be if used for qualitative results, understanding the meaning of these results is problematic.
Acknowledgement
The authors would like to thank the staff at the Bioaerosol Section of the Wisconsin Occupational Health Laboratory for their excellent work on analyzing all submitted samples.
Images and Tables