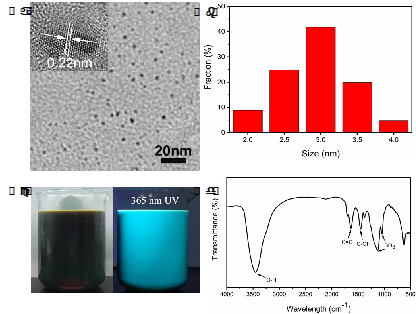
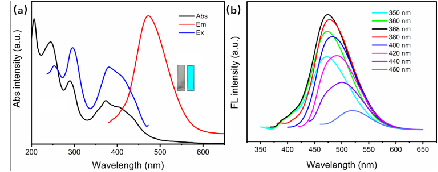
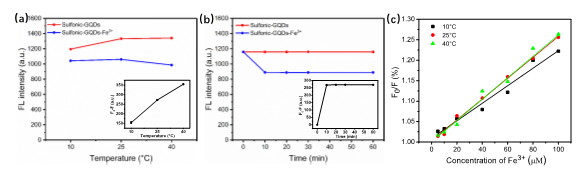
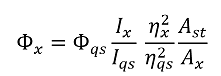Prof. Ming Hong Wu, School of Environmental and Chemical Engineering, Shanghai University, Shanghai 200444, P.R. China. E-mail: mhwu@shu.edu.cn
Prof. Yan Li Wang, Institute of Nano-chemistry and Nano-biology, Shanghai University, Shanghai 200444, P.R. China. E-mail: wangyanli@staff.shu.edu.cn