Vladimir M. Shkumatov
Fax: +37517 209 54 61 , Tel: +37517 200 82 72 , E-mail: biopharm@bsu.by
© 2019 Sift Desk Journals. All Rights Reserved
VOLUME: 1 ISSUE: 2
Page No: 38-40
Vladimir M. Shkumatov
Fax: +37517 209 54 61 , Tel: +37517 200 82 72 , E-mail: biopharm@bsu.by
Jan U. Panada1, Yaroslav V. Faletrov1,2, Vladimir M. Shkumatov1,2*
1 Faculty of Chemistry, Belarusian State University, Minsk, Belarus
2 Research Institute for Physical Chemical Problems, Belarusian State University, Minsk, Belarus
*Research Institute for Physical Chemical Problems, Belarusian State University, Leningradskaya str. 14, 220030, Minsk, Belarus
Vladimir M. Shkumatov, Synthesis and in silico evaluation of 16?-azido pregnenolone analogue as a clickable substrate for steroid-converting enzymes(2016)SDRP Journal of Computational Chemistry & Molecular Modelling 1(2)
Synthesis and characterization of 16β-azidopregn-5-en-3β,17α-diol-21-one are reported. The azido steroid was studied by docking simulations as a substrate for a range of steroid-converting enzymes, including bacterial cholesterol oxidase and mammalian cytochromes P450 17-hydroxylase/17,20-lyase (CYP17A1) and 21-hydroxylase (CYP21A2). The derivative was calculated to localize similarly to natural substrates with binding energy values ranging from -7.2 to -11.9 kcal·mol-1. A fluorescent triazole derivative of 16β-azidopregn-5‑en-3β,17α-diol-21-one has been obtained using click chemistry approach. The azido steroid is proposed as a suitable molecular tool for studies of steroid biotransformations.
Keywords: azido steroid, azide-alkyne cycloaddition, fluorescent labeling, molecular docking, cholesterol oxidase, CYP17A1, CYP21A2
Abbreviations
AHPE: 16β-azidopregn-5-en-3β,17α-diol-21-one; CHOX: cholesterol oxidase; CuAAC: copper-catalyzed azide-alkyne cycloaddition; CYP17A1: steroid 17-hydroxylase/17,20-lyase; CYP21A2: steroid 21-hydroxylase; HPE: pregn-5-en-3β,17α-diol-21-one; NBD: 7-nitrobenzo-2-oxa-1,3-diazol-4-yl.
16-substituted steroids are of interest to medicinal chemistry as potential anti-proliferative and anti-cancer pharmacological agents [1]. Moreover, such steroids can be further functionalized with either pharmacophore moieties or labeling fragments. Although a number of molecular modeling studies on 17‑substituted steroids are available there are few reports on corresponding 16‑compounds [2, 3]. Here we report the synthesis and docking simulations of 16β-azidopregn-5-en-3β,17α-diol-21-one as well as its fluorescently labeled derivative. The enzymes of choice consisted of bacterial cholesterol oxidase and mammalian cytochromes P450 17-hydroxylase/17,20-lyase (CYP17A1) and 21-hydroxylase (CYP21A2). CYP17A1 and CYP21A2 were chosen due to their implication into pathogenesis of prostate cancer and some others human steroidogenesis disorders, whereas cholesterol oxidase is involved in initial stages of cholesterol catabolism by some pathogenic bacteria, e.g. Gordonia species [4-6].
2.1 General procedures
The following reagents were used: 16α,17α-epoxypregn-5-en-3-ol-21-one (Sigma), sodium azide (Sigma); ascorbic acid, copper(II) sulfate pentahydrate, ethanol, isopropanol, glacial acetic acid, dimethylsulfoxide, chloroform, ethyl acetate and petroleum ether were supplied from local sources. All solvents except for ethanol were used as supplied by manufacturer. Unless indicated otherwise TLC analysis was performed using teflon-coated plates in a mixture of chloroform-ethyl acetate (9:1). Chromatograms were visualized by spraying with 53% sulfuric acid and illumination at 365 nm. GC-MS analysis was performed on Shimadzu GCMS‑2010 equipment. ESI-MS spectrum was recorded using Shimadzu LCMS-2020 chromatomass spectrometer. IR spectrum was obtained on a Bruker Alpha (ATR‑DI) apparatus in the range of 4000‑400 cm-1. Absorption and fluorescence spectra were recorded on a Solar CM2203 spectrofluorimeter (Belarus).
2.1. Synthesis of 16β-azidopregn-5-en-3β,17α-diol-21-one
The employed method was adopted from Winternitz and Engel with minor modifications [7]. To the solution of 16α,17α-epoxypregn-5-en-3-ol-21-one (50.5 mg, 1.53·10-4 mol) and sodium azide (64 mg, 1.01·10-3 mol) in a mixture of ethanol (2.2 ml) and dimethylsulfoxide (0.9 ml) were added 0.3 ml of ethanol containing 3 μl of acetic acid. The colourless solution was refluxed and stirred until complete consumption of starting material as indicated by TLC. Additional amounts of sodium azide (3 mg) and acetic acid (2.5 μl) in 300 μl of ethanol were added every day. Upon completion the solvent was removed in vacuo and the residue was washed with 10% NaCl-NaHCO3 solution several times. The crude product was recrystallized from chloroform-petroleum ether (1:1) to afford 40 mg of white powder (70% yield). TLC: RF = 0.34. IR (powder): 3350 (O-H), 2950, 2840 (C-H), 2098 (N=N=N), 1692 (C=O), 1620 (C=C), 1430 (C3-OH), 1376 (C17-OH), 1272 (N=N=N), 1109 (C17-OH), 1042 (C3-OH) cm-1. GC-MS: m/z = 327 [M-N2-H2O]+, 345 [M‑N2]+.
2.2. Azide-alkyne cycloaddition
Ascorbate-catalyzed CuAAC between AHPE and N-NBD-prop-2-ynamine was done using a modification of the reported method [8] (Figure 1).
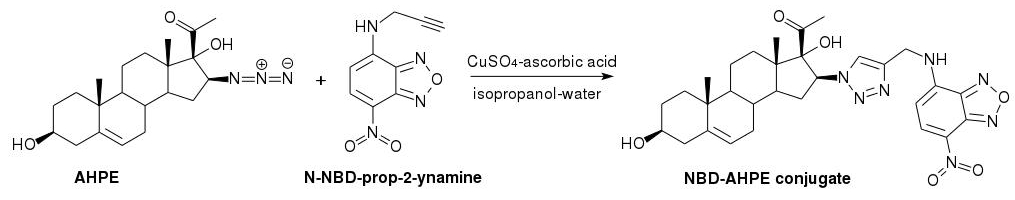
Figure 1: Synthesis of fluorescent NBD-AHPE conjugate.
The reaction was performed on nanomolar scale in the presence of 5-fold excess of CuSO4 and 20-fold excess of ascorbic acid. After heating for 3 hours a fluorescent substance with RF about zero was obtained. The product was purified by elution in ethyl acetate on a silica plate and extraction with ethanol (RF = 0.17) to give 8.2 nmol of NBD-AHPE conjugate as determined by photometry (82% yield). The excitation and emission wavelengths were characteristic of amino-NBD fluorophore (absorbance maximum at 463 nm, excitation and emission maxima at 465 nm and 535 nm, respectively; LC-MS data: m/z= 590.2 [M-H]-, 626.2 [M+Cl]-).
2.3. Docking simulations
Docking simulations were done using AutoDock Vina 1.1.2 software in the rigid receptor approximation [9]. Calculations were carried out using default forcefield parameters. Cholesterol oxidase from Brevibacterium sterolicum (PDB ID 1COY), human CYP17A1 (PDB ID 3RUK) and CYP21A2 (PDB ID 4Y8W) structures were obtained from RCSB Protein Data Bank.
In every case AHPE was found to localize favorably in active sites with binding energies comparable to those of endogenous ligands (Table 1). The 16-azido group did not contribute significantly to any binding mode although several possible donor-acceptor interactions were identified. The latter may include hydrogen bonding between the terminal nitrogen atom of AHPE and the hydroxyl group of Ser-109 of CYP21A2.
The relative calculated orientations of AHPE in active sites of CYP17A1 (Figures 2) and cholesterol oxidase (Figures 3) were found to be similar to the ones reported for endogenous ligands.
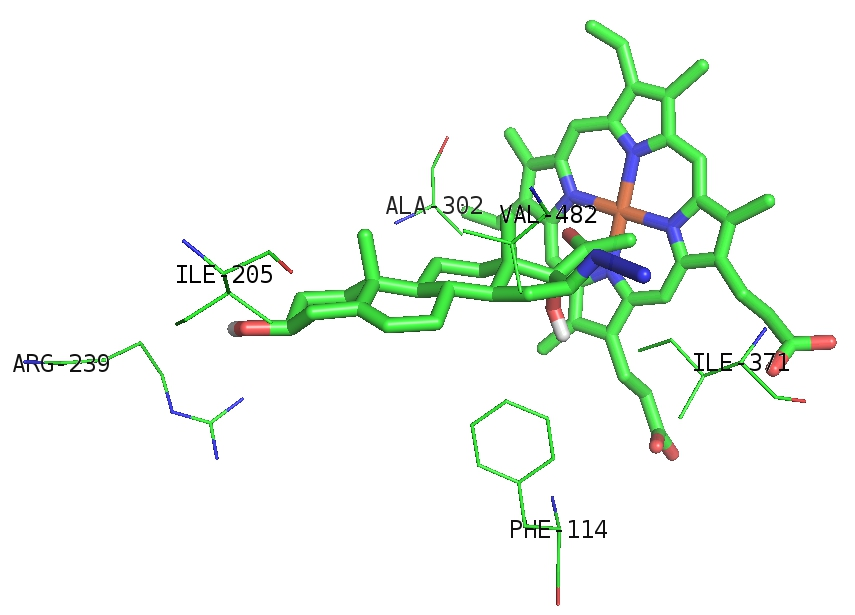
Figure 2: Computed localization of AHPE in the binding site of CYP17A1. Carbon backbones are displayed in green. Dark blue and red correspond to nitrogen and oxygen atoms, respectively.
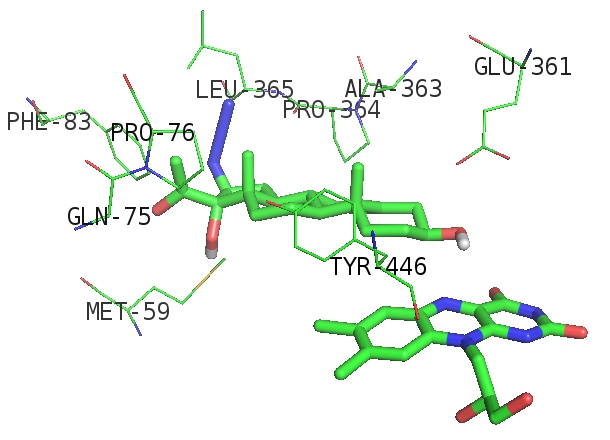
Figure 3: Side view of computed localization of AHPE in the active site of cholesterol oxidase. For atoms color map see capture to Figure 2
According to the results AHPE is a less suitable substrate for selected enzymes than reference compounds. In the case of cholesterol oxidase the chief reason for this appears to be high electron density due to carbonyl group of Pro-364 located next to azide group. No major steric hindrances appear to be involved. Similar structural relationships were observed by our group previously for fluorescent side chain-labeled cholesterols 22-NBD-cholesterol and 25-NBD-cholesterol [10], showing potential of artificial 3β-hydroxysteroids as substrates for cholesterol oxidases. In this work the postulate has been confirmed for AHPE in silico, indicating a prerequisite for experimental studies.
Table 1. Characteristics of binding modes of substituted pregnenolones
|
Enzyme |
Ligand |
-E, kcal·mol-1 |
∆r, Å (ligand atom-coenzyme atom) |
|
|
CHOX |
AHPE |
7.2 |
3.3 (C3-N5) |
|
|
HPE |
8.4 |
3.1 (C3-N5) |
||
|
Pregnenolone |
10.1 |
3.4 (C3-N5) |
||
|
CYP17A1 |
AHPE |
8.9 |
5.4 (O17-Fe) |
3.2 (O20-Fe) |
|
8.5 |
4.4 (O17-Fe) |
3.8 (O20-Fe) |
||
|
HPE |
11.0 |
3.6 (O17-Fe) |
2.7 (O20-Fe) |
|
|
10.7 |
4.7 (O17-Fe) |
2.8 (O20-Fe) |
||
|
Pregnenolone |
11.1 |
- |
4.2 (O20-Fe) |
|
|
CYP21A2 |
AHPE |
10,8 |
4.5 (C21-Fe) |
|
|
HPE |
11.9 |
4.2 (C21-Fe) |
||
|
Pregnenolone |
11.9 |
4.2 (C21-Fe) |
||
A 16β-azido pregnenolone derivative was prepared and characterized. It was predicted to interact with some enzymes involved in steroid bioconversions, namely CYP17A1, CYP21A2 and bacterial cholesterol oxidase. The fluorescent NBD triazole derivative of AHPE has been obtained using click chemistry approach. The results imply that AHPE is a promising artificial substrate for monitoring of human steroidogenesis and bacterial steroid catabolism.
The authors thank Prof. Dr. O. I. Shadyro and Dr. E. V. Grinyuk from the Faculty of Chemistry of Belarusian State University for providing the possibility to perform MS and IR experiments.
Bansal, R., Guleria, S., Thota, S., Bodhankar, S. L., Patwardhan, M. R., Zimmer, C., Hartmann, R. W., Harvey, A. L., 2012. Design, synthesis and evaluation of novel 16-imidazolyl substituted steroidal derivatives possessing potent diversified pharmacological properties. Steroids 77, 621-629. PMid:2236
View Article PubMed/NCBIAggarwal, S., Thareja, S., Bhardwaj, T. R., Kumar, M., 2010. Self-organizing molecular field analysis on pregnane derivatives as human steroidal 5α-reductase inhibitors. Steroids 75, 411-418. PMid:20170668
View Article PubMed/NCBIChen, H., Li, Q., Yao, X., Fan, B., Yuan, S., Panaye, A., Doucet, J. P., 2003. 3D-QSAR and docking study of the binding mode of steroids to progesterone receptor in active site . QSAR Comb. Sci. 22, 604-613.
View ArticlePorubek, D., 2013. CYP17A1: a biochemistry, chemistry, and clinical review . Curr. Top. Med. Chem. 13, 1364-1384 . PMid:23688130
View Article PubMed/NCBIMiller, W. L., Auchus, R. J., 2011. The molecular biology, biochemistry, and physiology of human steroidogenesis and its disorders . Endocr. Rev. 32, 81-151 . PMid:21051590
View Article PubMed/NCBIDrzyzga, O., de las Heras, F., Morales, V., Llorens, J. M. N., Perera, J., 2011. Cholesterol degradation by Gordonia cholesterolivorans. Appl. Environ. Microbiol. 77, 4802-4810. PMid:21622796
View Article PubMed/NCBIWinternitz, F., Engel, C. R., 1965. Steroids and the synthesis of related products. XXIV. The synthesis of 17α-amino 20-keto steroids. Part 1. Steroids 6, 805-840. 90102-9
View ArticleFehér, K., Balogh, J., Csók, Z., Kégl, T., Kollár, L., Skoda-Földes, R., 2012. Synthesis of ferrocene-labeled steroids via copper-catalyzed azide–alkyne cycloaddition. Reactivity difference between 2β-, 6β- and 16β-azido-androstanes. Steroids 77, 738-744. PMid:22521424
View Article PubMed/NCBITrott, O., Olson, A. J., 2010. AutoDock Vina: improving the speed and accuracy of docking with a new scoring function, efficient optimization and multithreading. J. Comput. Chem 31, 455-461. PMid:19499576
PubMed/NCBIFaletrov, Y V., Frolova, N. S., Hlushko, H. V., Rudaya, E. V., Edimecheva, I. P., Mauersberger, S., Shkumatov, V. M., 2013. Evaluation of the fluorescent probes Nile Red and 25-NBD-cholesterol as substrates for steroid-converting oxidoreductases using pure enzymes and microorganisms. FEBS J. 280, 3109 3119. PMid:23551929
View Article PubMed/NCBI