M.A Ochuka
E-mail: ochukamolly@gmail.com
© 2019 Sift Desk Journals. All Rights Reserved
VOLUME: 4 ISSUE: 4
Page No: 668-680
M.A Ochuka
E-mail: ochukamolly@gmail.com
M.A Ochuka a, C.O. Ikporukpo b, G.M, Ogendi c, J.Y. Mijinyawa d
a Department of Agricultural and Environmental Engineering, Pan African University, Life and Earth Sciences Institute, University of Ibadan, Ibadan, Nigeria.
b Department of Geography, University of Ibadan, Ibadan, Nigeria.
c Department of Environmental Science, Egerton University, Njoro, Kenya.
d Department of Agricultural and Environmental Engineering, University of Ibadan, Ibadan, Nigeria.
Tony Venelinov(TVenelinov_fhe@uacg.bg)
T Ribeiro(thierry.ribeiro@unilasalle.fr)
Ran Yu(yuran@seu.edu.cn)
Athanasios Alexopoulos(alexopo@agro.duth.gr)
M.A Ochuka, C.O. Ikporukpo, G.M, Ogendi, J.Y. Mijinyawa, Temporal Variations in Nutrients Loading in Lake Baringo Basin, Kenya.(2019)Journal of Earth Sciences & Environmental Studies 4(4)
Seasonal variation in nutrient loading in Lake Baringo were examined by investigating 10 limnological variables which are; Temperature, pH, electrical conductivity (EC), total dissolved solids (TDS), salinity, total nitrogen (TN), nitrates (NO3), nitrites (NO2), Ammonia (NH3), total phosphates (TP) and orthophosphates (OP). Sampling was carried out in January (dry season), April (beginning of rainy season), August (mid rainy season) and October (end of rainy season) from 10 different stations and analyzed using both chemical and statistical methods. All parameters varied seasonally except salinity which was statistically significant. Temperature mean values ranged from (27.8±4.94) to (31.01±1.97), pH (7.91±0.44) to (8.44±0.59), EC (332±83.78) to (540±1.69), TDS (243.88±1.73) to (302.63±2.00), Salinity (0.08±0.05) to (0.2±2.97), TN (2.52±1.79) to (3.52±1.75), NO3 (1.21±1.22) to (2.37±1.10), NH3 (0.14±0.06) to (0.41±0.21), TP (0.74±0.33) to (0.85±0.90), OP (0.20±0.08) to (0.41±0.59) while Nitrites was below the detectable limits <0.005. This result suggests that seasonal variations had significant negative effects on water quality of the basin which is adversely impaired by discharges from agricultural, industrial and domestic sources. These findings will assist and guide management decisions.
Keyword: Temporal Variation, Nutrient Loading, Pollution, Physicochemical Parameters.
Water is the most significant natural resource, essential for life and for the sustenance of mankind and the ecosystem (NWSS , 2015). Anthropogenic activities have greatly altered nutrients flows from watersheds into aquatic ecosystems, thereby increasing primary production and causing widespread eutrophication. Globally, Pollution of surface water with chemical pollutants and eutrophication of lakes and rivers are of significant environmental concern (Ouyang et al.,2006). More than 40% of water bodies are affected by eutrophication worldwide (Zamparas, 2015). Changes in Land use and unsteady growing seasons leads to the increased use of fertilizers which later leaches into rivers and lakes contributing to the risk of eutrophication and biodiversity loss (Bhateria and Jain, 2016) .Landscape disturbance over several decades, and the resulting increase in sedimentation is the dominant cause of the ongoing eutrophication in many of the lakes in Eastern Africa (Olago and Odada, 2007).The main concern of deteriorated water quality over time in Lake Baringo is turbidity, which has increased because of high rates of sedimentation resulting from increased soil erosion in the catchment (Odada et al., 2006).
Previous erosion studies extrapolated from Perkerra catchment reveal that Lake Baringo basin estimated sediment yield is 10.38 million metric tons/year (Onyando et al., 2005).Increased sedimentation has resulted to low oxygen concentrations, high salinity, turbidity and eutrophication and this has rendered the bed of the lake in the open waters nearly devoid of invertebrate life (Aloo, 2006).Additionally, water abstractions for irrigation along the riparian corridors in Lake Baringo basin has contributed to reduced stream flow’s which has resulted in a reduction in the lake depth and thus only a limited number of aquatic animals can survive under such conditions (Odada et al., 2006).Seasonal changes play a significant role in structuring characteristics of lakes and the variation of condition of lakes of the same region with similar local weather pattern might be expected to follow a similar temporal pattern (Rask and Ruuhij, 2002).The release of nitrate, transport, exchanges and transformations within the catchment are controlled by a complex set processes which varies in time (Jarvie et al., 2002).For instance, during rainy seasons, phosphates are washed from soils into drainage ditches and canals (Boman et al., 2018). Extreme seasonal variations have had significant ecological influences on Lake Baringo Basin, resulting to water quality change (Ouma and Mwamburi, 2014).
Management of water resources is critically important for the economy, the environment and society. Safe water must be considered as a basic human right and should be available and affordable to all. Therefore there is a need to save the Lake from further deterioration, to avoid the disastrous consequences of these threats and lead to a future where the full potential of Africa’s water resources can be readily unleashed as envisaged in the new Africa Water Vision for 2025. United Nations recognized water security as one (Goal 6) of the seventeen sustainable development goals (SDGs), which aims at ensuring availability and sustainable management of water and sanitation for all by 2030.Therefore there is a great need to understand the cycle of water deterioration for water resources management underpinned by the 2016 Water Act in light of aiming to achieve Kenya’s Vision 2030 ambitions. Water quality investigations provide information on the health of water bodies and for developing strategies that help in better management of catchment and water resources ( Brainwood et al., 2004). Hence, the purpose of this study is to investigate seasonal changes in surface water quality in lake Baringo.
Study Area
Lake Baringo is a freshwater lake located in a tectonically active closed basin in the Eastern arm of the Rift Valley in Kenya and is an internationally recognized important world Ramsar site. It is located between 00°300N and 00°450N, and 36°000E and 36°100E, lying 60 km north of the equator at an altitude of 975 m above sea level. It has a surface area of 130 km² and an altitude of about 1100m a.s.l..It drains a total area of 6,820 km2. The lake’s water catchment includes the Mau Hills and Tugen Hills. Several seasonal rivers drain into the lake, including Ol Arabel, Makutan, Tangulbei, Endau and Chemeron. River Perkerra and Molo are perennial rivers, although with significantly reduced water discharges during dry seasons. It has no visible outlet and it is believed to have an underground seepage at Kapedo to the north and the water flows out as geysers and hot springs into Lake Turkana.It is characterized by a very high annual evaporation rate that ranges from 1,650 to 2,300 mm and annual rainfall that varies between 1,100 mm and 2,700 mm .The long rains occur in the months of April to August, whereas the short rains fall from October to November.
Population Density
According to the population and housing census conducted in 2009, the population size of Baringo County was 555,561, consisting of 279,081 males and 276,480 females. The county’s intercensal growth rate is 3.3 per cent per annum, which is above the national average of 3 per cent. The population of the county was estimated to be 613,376 in 2012 and is further projected to increase to 677,209 and 723,411 in 2015 and 2017, respectively.
Economic Activity
The study area is inhabited by three major ethnic groups namely the Turgens to the east, Pokots, to the north, Ilchamus/Njemps South and eastern sides. All groups are agro-pastoralists. Pokot who inhibit the northern part of the area are heavily dependent on livestock keeping and in particular goats while the Njemps are mainly fishermen. The Turgen settled up on the humid hills with good conditions for rain fed farming are in most cases agriculturalist. Perkerra Irrigation scheme has increased horticultural production and led to the growth of Marigat town. Additionally, large scale sisal farming has led the establishment of sisal industry in the catchment. The community heavily relies on charcoal burning as one of the major socio-economic activity alongside Bee-Keeping and brewing of local liquor as an alternative source of income. Donkey abattoir in Mogotio, the Goldox Company, is one of the latest company for large scale production of donkey meat for export to china.
Lake Baringo Catchment is also an international tourist site and tourism has become one of the economic activities in the catchment. The study area is a critical habitat and refuge for more than 500 species of birds and fauna, seven species of fresh water fish, several species of animals including the hippo, Crocodiles and many more freshwater organisms. The lake has major seven islands namely Ruko, Rongena, Linage, Samatian, Olkokwa, Parmalok and the Devil’s Island. The largest being Ol-Kokwa Island which is extinct volcanic centre and has several hot springs. Ruko Island on the other hand is a park having a variety of wildlife such as Zebra and Giraffe.

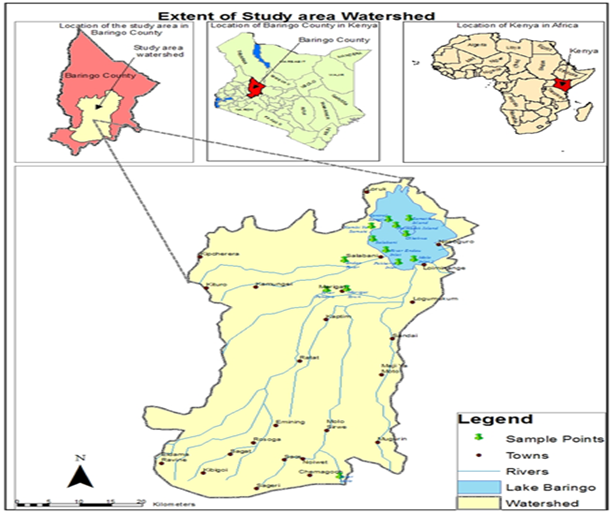
Figure 1: Lake Baringo
Sampling
Samples were collected in four seasons; beginning of dry season (January), end of dry season (April), rainy season (August) and end of rainy season (October) during the year 2018. Ten water quality sampling points were selected in areas where human activities were high. The sampling sites were designed to cover a wide range of determinant that reasonably define the spatial extent and water quality of the catchment. The sampling sites were River Molo inlet (Site 1), River Perkerra inlet (Site 2), River Endau Inlet(3), Samatian Island (Site 4), Ol-kokwa Island (Site 5), Parmalock Island (Site 6), Kampi ya Samaki (Site 7), River Endau (Site 8), River Perkerra (Site 9) and River Molo (Site 10). However, river Endau being a seasonal river was sampled once in the month of August when the rains were high. The sampled water quality parameters included water temperature, Water PH, Total Dissolved Solids, Electrical Conductivity, Salinity, Total Nitrogen, Nitrates, and Nitrites, Ammonia, Total phosphate and Orthophosphate.
Physical and Chemical Analysis.
Physical parameters were measured in insitu including: Temperature, Electrical conductivity, PH, salinity and Total Dissolve Solids using HACH, USA (Model: sension™+ EC71) portable multi-meter. While the Chemical parameters such as Total phosphorus (TP) and total nitrogen (TN) concentrations were analyzed according to APHA 2005. Total nitrogen (TN) was analyzed with unfiltered water samples, via digestion with concentrated sulphuric acid (by autoclave procedure) to convert organic nitrogen to ammonium nitrogen (NH4-N), with subsequent analysis for TN carried out, as outlined for NH4-N. Nitrate and nitrite were measured with the cadmium reduction method and ascorbic acid method (using HACH DR2800 spectrophotometer); Ammonium was determined with the phenol hypochlorite method, using nitroprusside as a catalyst. Total phosphorus was analyzed by oxidizing unfiltered water samples with hot 5% potassium per sulfate (K2S2O8) in distilled water. The tubes (samples, standards and blanks) were autoclaved for 30 min. They were further cooled to room temperatures with the tube caps slightly loosened. The TP concentration was then determined using the methods described above for inorganic phosphate. Phosphate phosphorus (PO4-P) was measured with the ascorbic acid method.
Statistical Analyses
Descriptive statistics such as mean and standard deviation (mean ± SD) of physicochemical parameters were calculated using Microsoft Excel and SPSS package. Inferential Statistics like the one way analysis of variance (ANOVA) was used to analyze the seasonal variations in the physico- chemical parameters at a significance level of p< 0.05.
Physico-Chemical analysis was carried out on samples for four seasons; January (dry season), April-August (rainy season) and October (End of rainy season). The release of nutrient including nitrates, organic phosphorus and ammonia into the water body from agricultural lands, industries, water treatment plants contributed to the algal blooming; and this conditions are seasonal (El-sheekh, 2016). All physical and chemical parameters of Lake Baringo and its catchment demonstrated statistical insignificance (P >0.05) except salinity. A summary of the range, mean value, standard deviation of measured variables are provided in Table 1.
Table 1: Monthly Means of the Physico-Chemical Parameters.
|
Physico-chemical Parameters |
||||||||||
|
Months |
TN |
NO3 |
NH3 |
TP |
OP |
TEMP |
PH |
EC |
TDS |
Salinity* |
|
January |
|
|
|
|
|
|
|
|
|
|
|
Mean±SD |
3.43±2.84 |
1.24±1.66 |
0.41±0.21 |
0.83±0.74 |
0.41±0.59 |
31.01±1.97 |
8.05±0.66 |
540±1.69 |
302.63±2.00 |
0.2±2.97 |
|
Range |
1.36-9.98 |
0.28-5.11 |
0.12-0.77 |
0.4-2.6 |
0.13-1.84 |
28.2-33.8 |
7.2-8.73 |
538-543 |
300-305 |
0.00-0.2 |
|
April |
||||||||||
|
Mean±SD |
3.52±1.75 |
1.21±1.22 |
0.27±0.12 |
0.74±0.33 |
0.20±0.08 |
29.04±2.17 |
7.91±0.44 |
537.5±9.26 |
287.63±34.86 |
0.15±0.09 |
|
Range |
2.07-6.63 |
0.09-3.21 |
0.11-0.53 |
0.49-1.48 |
0.09-0.32 |
24-31 |
7.18-8.5 |
520-552 |
202-303 |
0-0.2 |
|
August |
||||||||||
|
Mean±SD |
2.52±1.79 |
1.23±1.45 |
0.17±0.14 |
0.76±0.31 |
0.35±0.16 |
27.8±4.94 |
8.18±0.53 |
332±83.78 |
297.5±113.11 |
0.09±0.04 |
|
Range |
1.4-6.86 |
0.32-4.29 |
0.05-0.44 |
0.54-1.37 |
0.18-0.63 |
21.3-32.6 |
7.4-8.78 |
224-438 |
183-433 |
0-0.1 |
|
October |
||||||||||
|
Mean±SD |
3.17±1.40 |
2.37±1.10 |
0.14±0.06 |
0.85±0.90 |
0.31±0.07 |
28.83±3.03 |
8.44±0.59 |
430.88±4.91 |
243.88±1.73 |
0.08±0.05 |
|
Range |
1.89-6.02 |
0.59-4.2 |
0-0.19 |
0.25-2.91 |
0.19-0.4 |
22.8-32.7 |
7.4-8.92 |
420-435 |
241-246 |
0-0.1 |
*Significantly varies at P<0.05. Source: Researcher’s Survey, 2018
|
Abbreviation |
Description |
|
TN |
Total Nitrogen |
|
NO3 |
Nitrate |
|
NH3 |
Ammonia |
|
TP |
Total Phosphate |
|
OP |
Orthophosphate |
|
TEMP |
Temperature |
|
EC |
Electrical Conductivity |
|
TDS |
Total Dissolved Solids |
|
SD |
Standard Deviation |
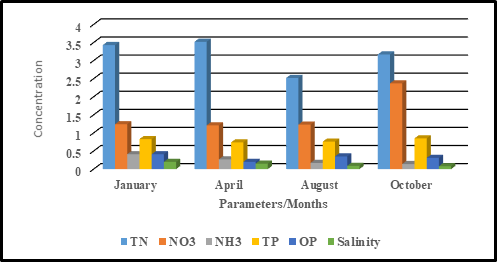
Figure 2. Seasonal Variation in the concentration of TN, NO3, TP, OP and Salinity.
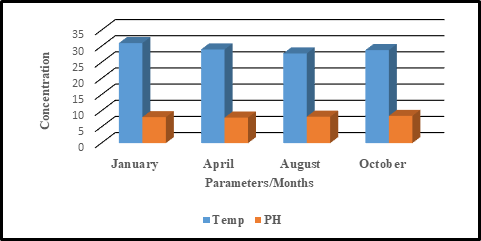
Figure 3. Seasonal Variation in the concentration of Salinity and Temperature Value.
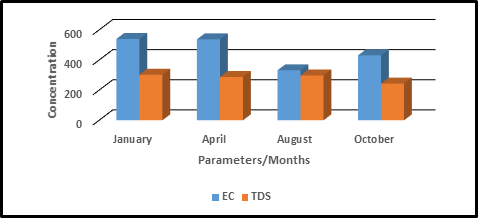
Figure 4. Seasonal Variation in the concentration of TDS and EC.
The temperature of any given water controls the rate of all chemical reactions and affects fish growth, reproduction and immunity (Bhateria and Jain, 2016).Temperature ranged from 28.2 to 33.8oC with mean value of 31.01±1.97oC for dry season while in wet season, temperature ranged from 21.3-32.6oC with mean value of 27.8±4.94°C. The mean value of temperature in dry season was higher than wet season. The temperature in dry season was above the acceptable range (25°C –32 °C) as recommended by World Health Organization. Similar results were observed in Tigris River which showed highest temperatures during the summer months and lowest during the winter months (Varol et al., 2011). A study in Nile water of Egypt, showed low temperatures values during the winter season and increase toward the summer season (Ali et al., 2014) The general high temperatures in Lake Baringo could be attributed to high solar radiation considering the fact that the study area is in semi-arid part of Kenya. The variation in temperature could be attributed to time and season of sampling. Early morning samples and late evening recorded lower temperatures compared to midday sampling. Additionally, the high temperature recorded during the dry season due to the depth of the water; when the water level is low, light rays from the sun can penetrate through the water and it gets heated to high temperatures. The high water temperatures could also be attributed to effluent discharges from the tourist lodges and surface runoffs from large scale Perkerra irrigation scheme in the catchment. Previous studies in the same Lake reported high temperature ( Omondi, et al 2011). Dallas (2017) water temperature variations results from thermal discharges, agricultural run offs, river regulation, industrial effluents, domestic discharge and global warming. The rise in temperature of water accelerates chemical reactions, raises metabolic activity of organisms and decreases solubility of gases (Umarani et al., 2011). Increased metabolism instantaneously lowers oxygen availability creating a noxious condition that could quickly cause stress and mortality and ecosystem breakdown (Andrew and Gedan, 2014). Cold water contains more oxygen than warm water (El-sheekh, 2016). Leading to the proliferation of invasive species in an aquatic system (Havel and Kats, 2015). Future changes in climate patterns may also intensify current and generate new water quality challenges which may have unknown long-term impacts on the world’s ecosystems (UNEP, 2016).According to Ouma and Mwamburi, (2014) higher temperature contributed to chlorophyll-a concentration in Lake Baringo. When the results were subjected to statistical analyses, there was no seasonal significant difference (P>0.05).
Aquatic organisms are affected by pH because most of their metabolic activities are pH dependent. pH of an aquatic system is an important indicator of the water quality and the extent of pollution in the watershed areas (Kumar, 2011). The pH values ranged between 7.2-8.73 with a mean value of 8.05±0.66 for dry season while in wet season, the pH ranged from 7.4-8.78 with a mean value of 8.18±0.53.The pH values were higher in wet season than dry season. The pH range both in dry and wet season were within the pH range of (6.5 to 8.5) required value for drinking and domestic purposes by World Health Organization (WHO, 2011) and Kenya Bureau of Standards (KEBS) limit for natural water (6.0-8.5) .Optimal pH range for sustainable aquatic life is pH 6.5-8.2 (Adeyemo et al., 2008). The result agrees with (Atobatele and Olutona, 2013). The high pH during the wet season can be linked to the process of photosynthesis; Photosynthesis increases pH value in the surface water through the removal of CO2. A similar finding was obtained by (Edokpayi et al., 2015).However (Wilbers et al., 2014) reported a slightly lower pH in the wet season than in the dry season in their studies of spatial and temporal variability of surface water pollution in the Mekong Delta, Vietnam. High pH values may be attributed to fertilizers from the intensive irrigated horticultural activities in the catchment and large scale sisal farms at Mogotio. Additionally, the high pH could be attributed to lack of any surface outlet and high concentrations of carbonate salts, typically sodium carbonate. However, low pH in the catchment could be attributed to discharges from car washing within the Lake and its tributaries and decomposition of organic matter from wastewater originating from nearby towns such as Mogotio town and Marigat can lead to acidification. Decrease in pH of water increases the solubilization of some metals, which may be toxic to fish and render the water unsuitable for other uses. pH variation was statistically insignificant (P>0.05).
Electrical Conductivity (EC) is directly related to total solids and is a function of the geology of the area. It also indicates the presence of dissolved ions in water (Odiyo et al., 2012). EC is a numerical expression ability of an aqueous solution to carry electric current. Electrical conductivity (EC) values season ranged from 538-543 µS/cm for dry with a mean value of 540±1.69 µS/cm while wet season ranged from 224-438 µS/cm with a mean value of 332±83.78 µS/cm.It is evident that the mean value for dry season was higher than the wet season.The obtained values were within WHO limits of 600µS/cm (WHO, 2011). The findings were in conformity with (Deepa et al., 2016; Vaishali et a.l., 2013 ).This result contradicted the normal expected results of a wet season which is characterized with heavy rainfall having high EC values due to high surface runoff from agricultural lands (Shabalala et al., 2013). Anhwange et al., (2012) reported a decrease in conductivity during the dry season
EC is controlled by geology of the area where the water body is situated, the size of the watershed, wastewater from sewage treatment plants, wastewater from septic systems, urban runoff from roads and agricultural runoff. Baringo catchment streams run through areas with mainly clay loam with alluvial deposits which tend to have higher conductivity because of the presence of materials that ionize when washed into the water. Additionally, the high Conductivity during the dry season could also be attributable to increased evaporation rates due to the arid nature of the catchment. It could also be due to increased decomposition and mineralization of organic materials (Ecjhao and Begum, 2008). Agricultural surface runoffs and domestic effluent discharges into the Lake from the Tourist Lodges on the Island such as Samatian Island, and at the shores such as Soi lodge and from Kampi ya Samaki Health Center are attributed to the high EC. Increased deforestation due to intensive charcoal burning in the study area weakens the soil structure making it susceptible to erosion and are drained into water bodies thus increasing ion concentration. The lower value obtained during the wet seasons could be related to the dilution effect by rain water. Dissolved ions can alter the taste of water and also contribute to the hardness of water. Water with high EC values is not suitable for domestic purposes and irrigation as it can lead to the salinity of agricultural soils. All the sampling seasons exhibited high EC and statistical analysis of the data obtained for both seasons showed no significant difference (p > 0.05).
Duffy and Weber (2007) reported that the concentration and composition of Total Dissolved Solids (TDS) in natural water is determined by the geology of the drainage, atmospheric precipitation and the water balance. TDS vales ranged from 300-305mg/l with a mean value of 302.63±2.00 mg/l during dry season while in the wet season, the values ranged from 183-433mg/l with a mean value of 297.5±113.11 mg/l.The results indicates that the TDS values were higher in dry season than wet season. A previous study in Lake Baringo revealed similar results of high TDS (Mwamburi, 2015; Kumar et al.,2015).However,(Islam and Azam, 2015;Manohara et al., 2019) in their studies observed high TDS values during the wet seasons resulting from run-off in wet seasons. TDS values were within the acceptable National Environmental Management Authority (NEMA, Kenya) limits of 1200 mg/l for natural water. Statistics analysis indicates no significant difference at (P>0.05). Similar
Primary sources for elevated TDS level in river water are agricultural runoff, leaching of soil contamination and point source water pollution discharge from industrial and sewage treatment plants particularly during dry season with low water level. Lower TDS during wet season could be attributed to the dilution effect. Electrical conductivity, BOD and turbidity of water increases with increase in TDS and this influences the existence of aquatic life a river can support. Waters with high (TDS) are unpalatable and potentially unhealthy. High TDS levels gives the water a musty taste and impairs its aesthetic values through discolorations. Sedimentation has affected the water quality of lake Baringo with approximately 400 tonnes of silt deposited into the lake during every rainy season turning the appearance of the lake muddy and brown in appearance (Josephine, 2014).
Salinity refers to the total dissolved salts which are usually but not limited to sodium and chloride ions as well as others such as potassium and bicarbonate ions (UNEP, 2010).Salinity is a dynamic water quality indicator of the nature of the exchange system. The Salinity value ranged from 0.00-0.2ppt with a mean value of 0.2±2.97 for the dry season and wet season ranged from 0 to 0.1 with a mean value of 0.09±0.04.It is evident that the dry season had higher salinity value compared the wet season .Generally, the salinity level in the study area was low. High salinity in dry season could be attributed to the high evaporation rates increasing the concentration of the water constituents while the low salinity can be related to the dilatation effects of the incoming flowing water from the catchment during the rainy season. The semi-arid nature of Lake Baringo Catchment could also be a contributing factor to higher salinity. Salinization of water bodies has always been related to agricultural activities especially in arid and semi-arid environments throughout the world (Plessis, 2019).Increases in precipitation and runoff combined with warmer sea surface temperatures and lower currents increase the intensity of stratification ( Ranjan et al., 2013).The finding is in agreement with a previous study serthalaikadu Lagoon, east coast of India where salinity values of monsoon season had very low than those of during the summer season (Nagalakshmi et al., 2016). Increased salinity increases the tension on freshwater organisms by affecting metabolic function and oxygen saturation levels. Riparian and emergent vegetation can also be altered by rising salinity. The seasonal Variation in salinity was statistically significant p<0.05.
Total nitrogen is the sum of total kjeldahl nitrogen (ammonia, organic and reduced nitrogen) and nitrate-nitrite. It can be derived by monitoring for organic nitrogen compounds, free-ammonia, and nitrate-nitrite individually and adding the components together (McGill et al., 2010). A Total Nitrogen values ranged from 1.36-9.98 with a mean value of 3.43±2.84 for dry season while the wet season the values ranged from 1.4-6.86 with a mean value 2.52±1.79. It is evident that the values in dry season were higher than the values obtained in wet season. This result is consistent with the findings of (Gosine and Mahon, 2015; Balarabe and Auta, 2012 ).Gosine and Mahon, (2015) reported higher concentration of potential sources of coastal pollution during dry days at 1.4 mg/l than on wet days in Coset Bay watershed .Similarly, Balarabe and Auta (2012) discovered a disparity in nutrients obtained from Nguru Lake during the dry seasons to be significantly higher than values recorded in the rainy seasons.
Total Nitrogen concentrations showed clear seasonal variation and exhibited a tendency to decrease during the wet season and increased in the dry season. This may be attributed to the dilution of nutrients as a result of the heavy rains High River flows and the resultant dilution effect in the wet seasons. Similarities in the concentration irrespective of the seasons resulted from dry season irrigation farming and concentrations of these nutrients due to evaporation and increased surface runoffs during the wet season. Additionally, aquatic macrophytes growing in riparian wetlands and shallow water bodies can absorb and accumulate large quantities of contaminants in the wet season (Xiaolong et al., 2010). However, the seasonal variations in the TN concentrations was not statistically significant P>0.05
Nitrate is a form of nitrogen and a vital nutrient for the growth, reproduction and survival of organisms (Adeyemo et al., 2008). Estimation of Nitrate levels is important in evaluating the potential biological productivity of surface water (Venkatesharaju et al., 2010).The nitrate values for the dry season ranged from 0.28-5.11with a mean value of 1.24±1.66 while the wet season values ranged from 0.32-4.29 with a mean value of 1.23±1.45.The results indicate that the dry season had a higher mean value that the wet seasons. The concentrations of NO3 did not exceed the WHO threshold limits for domestic water use and National Environmental Authority in Kenya (NEMA).Similar results were reported by Mbui et al.,(2017); Atobatele and Olutona (2013) and Adeyemo et al.,(2008). During the dry seasons, nitrates are usually built up and that high levels of nitrates are only observed during early rainy seasons. This is because initial rains flush out deposited nitrate from near surface soils and nitrate level reduces drastically as rainy season progresses (Symader and Bierl, 2010). However ,a contradicting results have been reported by (Deepa et al., 2016). High nitrate detected can be attributed use of fertilizers Perkerra irrigation scheme which get leached and eroded to streams. The presence of nitrate originate from the contamination by surface runoff from riverine agricultural irrigated horticultural farms (Maghanga et al.,2013). Nitrate is contributing significantly to the water pollution (Singh, 2013). Brewing Local Traditional Spirits commonly known as “Chang’aa” in Kiswahili at the Lake shores is a common phenomenon in Lake Baringo catchment particularly at Kataran Village. They use the Lake water as a condenser and pouring the molasses in the lake. The by-products (e.g. mash, yeast surplus) generated are discharged into the Lake. This causes water pollution because the distillery wastewater contains nutrients in the form of nitrogen and potassium. Recent studies in Lake Baringo reflected presence of algae at river Molo inlet due to rich inflow of nutrients into the lake from the catchment areas (Ndiba et al., 2018).
Increase in Nitrates causes deterioration in both the hygienic and aesthetic aspects of water quality due the proliferation of aquatic weeds. Nitrates render a water body eutrophic consequently creating a number of human health problems (Fennessy and Cronk, 1997). Consumption of drinking water contaminated with high levels of nitrates by infants below six months old and certain young animals causes “oxygen debt” in the blood, a fatal condition known as methemoglobinemia or blue baby syndrome and miscarriages in pregnant women (Weselak et al., 2007).The difference in the mean values of the concentrations observed during the dry and wet season was statistically insignificant (P>0.05).
Nitrite is the intermediate oxidation state between ammonia and nitrate, can appear as a transient species by the oxidation of ammonia or by the reduction of nitrate. In present study, nitrite was below the detectable limits .Nitrites are relatively short-lived because they are quickly converted to nitrates by bacteria.
Ammonia comes from the biodegradation of waste and inputs from domestic, agricultural and industrial, and that is why it is a good indicator of contamination of water ways. The Ammonia is abundant in surface waters and its concentrations is highly variable during lake seasonal cycles .The most important source of ammonia is the ammonification of organic matter. Ammonia is rapidly oxidized by certain bacteria, in natural water systems, to nitrite and nitrate, a process that requires the presence of DO. Ammonia concentration varied seasonally across the sampling periods. The ammonia values for dry season ranged from 0.12-0.77 with a mean value of 0.41±0.21 while in the wet season, the values ranged from 0.05-0.44 with a mean value of 0.17±0.14.I .It is evident that the value of ammonia was higher in the dry season that in wet season. A similar study in Dongting Lake, China also revealed that ammonia concentrations during the dry season were higher than those during the wet season (Tian et al., 2017).Additionally, Barakat et al.,(2016) observed a higher ammonia concentration in summer compared to the winter season in Oum Er Rbia River, Morocco. The higher ammonia concentration in dry season can be attributed to the reduced dilution of effluents. The residents in the study area are majorly pastoralists and this is attributable to the presence of ammonia in the water due to inputs of decomposing animal dung during animal watering. In contrast to the results, previous finding in the upper Mississippi River revealed that Total ammonia nitrogen concentrations in surface water were greatest in winter and lowest in summer (Razier et al., 1996).Generally, the ammonia levels were low due to the oxidation of ammonia by nitrifying bacteria coupled with ammonia assimilation by plants and planktonic algae. This process reduces ammonia concentrations in overlying surface water. Increase in ammonia content depicted the disposal of sewage disposal or effluent discharge of industries used ammonia liquor or ammonia gas for process of its production (Vaishali and Punita, 2013). Ammonia provides nutrient for algae and other forms of plant life and thus contribute to overloading of natural systems and cause eutrophication. Ammonia can block oxygen transfer in the gills of fish, thereby causing immediate and long term gill damage. There was no significant difference p>0.05.in the ammonia concentration across the sampling periods. In this study, ammonia levels were above the EPA recommended limit of 0.02 mg/L for freshwater environments.
Phosphate can be found as a free ion in water systems and as a salt in terrestrial environments. In most water bodies phosphorous is recognized as the limiting nutrient and considered to be the most significant among the nutrients responsible for eutrophication of lakes, as it is the primary initiating factor. Phosphates can be in organic form (organically-bound phosphates) or inorganic form (including orthophosphates and polyphosphates).Phosphate comes from fertilizers, pesticides, industry and cleaning compounds. It also occurs naturally originating from phosphate containing rocks. A modest increase in phosphorus under the right conditions can cause eutrophication. During the dry season, the phosphate values ranged from 0.4-2.6 with a mean value of 0.83±0.74 while in wet season, the values ranged from 0.54-1.37 with a mean value of 0.76±0.3.The values were higher in wet season than in dry seasons. It exceeds the permissible limit of 0.3 mg/l, and hence, the risk of eutrophication. Similar result was Observed by (Atobatele et al.,2013; Singh and Meetei,2013;and Anhwange et al., 2012).Additionally, a study in northern Spain (Europe) river catchments revealed that phosphates increased in summer and autumn, with a maximum mean value of 0.23 mg/l in summer, and decreased in spring and winter, with a minimum mean value of 0.11 mg/l in spring (Álvarez-Cabria et al.,2016). On the contrary, Kumar et al., (2015) recorded highest phosphate values during post-monsoon season could be attributed to the heavy rainfall in Pichavaram mangrove forest water, south- eastern India. The high value of phosphate in wet season are mainly due to the use of detergents, weathering of rock minerals, decomposition of organic matter, detergents, fertilizers, industrial waste, household waste and phosphate minerals, domestic discharges especially use of based detergents for laundry. The residents around the lake bath directly into the Lake and even wash clothes right inside the lake. Apart from that the car washing activities observed at Kampi ya Samaki Beach, River Molo and River Endau increased the flow of soap into the streams contributing to phosphate increase. A small amount of phosphorous may enhance the growth of algal blooms leading to eutrophication and ultimately produce harmful toxins. When additional phosphorus becomes available to the aquatic system, it stimulates the growth of algae and other aquatic plants. Phosphate value above 0.025 mg/L is found to accelerate eutrophication process in rivers and lakes (Howell, 2010). Degradation of water bodies consequently kills fishes and other aquatic animal due to oxygen deficient condition. Orthophosphate values in dry season ranged from 0.13-1.84 with a mean value of 0.41±0.59 in dry season while in wet season, the values ranged from 0.18-0.63 with a mean value of 0.35±0.16. There was no significant difference p>0.05 in phosphate and orthophosphate concentration across the sampling seasons.
The study shows that seasonal variations had great influence on the water quality. The deterioration in water quality is found due to higher amount of organic pollutants into the water. Anthropogenic activities influenced water quality within the basin. The study reveals that in dry season the level of pollution was much higher than in wet season. We therefore have the following recommendation for the relevant stakeholders.
This research was supported by research grants from the African Union Commission through flagship institution Pan African University (PAU). We acknowledge Nakuru Water and Sanitation Services Company (NAWASSCO) Laboratory for analyzing the parameters. Thanks also are due to those who reviewed this manuscript.
Adeyemo, O. K., Adedokun, O. A., Yusuf, R. K., & Adeleye, E. A. (2008). Seasonal Changes In Physico-Chemical Parameters And Nutrient Load Of River Sediments In Ibadan City, Nigeria. Global Nest Journal, 10(3), 326–336.
Ali, E. M., Shabaan-Dessouki, S. A., Soliman, A. R. I., & Shenawy, S. El. (2014). Characterization Of Chemical Water Quality In The Nile River , Egypt. International Journal Of Pure &Applied Bioscience, 2(3), 35–53. Retrieved From Www.Ijpab.Com
Aloo, P. A. (2006). Effects Of Climate And Human Activities On The Ecosystem Of Lake Baringo, Kenya. 335–347.
View ArticleÁlvarez-Cabria, M., Barquín, J., & Peñas, F. J. (2016). Modelling The Spatial And Seasonal Variability Of Water Quality For Entire River Networks: Relationships With Natural And Anthropogenic Factors. Science Of The Total Environment, 545–546, 152–162.
View ArticleAndrew H. Altieri And Keryn B. Gedan. (2014). Climate Change And Dead Zones. 1–12.
View ArticleAnhwange, B. A., Agbaji, E. B., & Gimba, E. C. (2012). Impact Assessment Of Human Activities And Seasonal Variation On River Benue, Within Makurdi Metropolis. International Journal Of Science And Technology, 2(5), 248–254.
Atobatele, E. O., & Olutona, G. O. (2013). Spatio-Seasonal Physico-Chemistry Of Aiba Stream , Iwo , Nigeria. Afican Journal Of Biotechnology, 12(14), 1630–1635.
View ArticleBalarabe, M. L., & Auta, J. (2012). Effects Of Physico-Chemical Factors On Seasonal Dynamics Of The Phytoplankton In Nguru Lake ,. 2(8), 74–82.
Barakat, A., El, M., Rais, J., Aghezzaf, B., & Slassi, M. (2016). International Soil And Water Conservation Research Assessment Of Spatial And Seasonal Water Quality Variation Of Oum Er Rbia River ( Morocco ) Using Multivariate Statistical Techniques ☆. International Soil And Water Conservation Research, (November), 0–1.
View ArticleBarrado, E., & Deba, L. (1998). Assessment Of Seasonal And Polluting Effects On The Quality Of River Water By Exploratory. 32(12).
Bhateria, R., & Jain, D. (2016). Water Quality Assessment Of Lake Water: A Review. Sustainable Water Resources Management, 2(2), 161–173.
View ArticleBoman, B. J., Wilson, P. C., & Ontermaa, E. A. (2018). Understanding Water Quality Parameters For Citrus Irrigation And Drainage Systems 1 Obtaining A Water Sample. 1–11.
Brainwood, M. A., Burgin, S., & Maheshwari, B. (2004). Temporal Variations In Water Quality Of Farm Dams : Impacts Of Land Use And Water Sources. 70, 151–175.
View ArticleDallas, H. (2017). The Effect Of Water Temperature On Aquatic Organisms : A Review Of Knowledge And Methods For Assessing Biotic Responses To Temperature The Effect Of Water Temperature On Aquatic Organisms : A Review Of Knowledge And Methods For Assessing Biotic Responses .
Deepa, P., Raveen, R., Venkatesan, P., Arivoli, S., & Samuel, T. (2016). Seasonal Variations Of Physicochemical Parameters Of Korattur Lake , Chennai , Tamil Nadu , India. 4(3), 116–123.
Deng, A., Ye, C., & Liu, W. (2018). Spatial And Seasonal Patterns Of Nutrients And Heavy Metals In Twenty-Seven Rivers Draining Into The South China Sea. Water (Switzerland), 10(1).
View ArticleDuffy, L. K., & Weber-Scannell, P. K. (2007). Effects Of Total Dissolved Solids On Aquatic Organisms: A Review Of Literature And Recommendation For Salmonid Species. American Journal Of Environmental Sciences, 3(1), 1–6. Retrieved From Http://Pebblescience.Org/Pdfs/Tdsalaskastudy.Pdf
Ecjhao, C., & Begum, A. (2008). Study On The Quality Of Water In Some Streams Of Cauvery River. E- Journal Of Chemistry, 5(2), 377–384.
Edokpayi, J. N., Odiyo, J. O., Msagati, T. A. M., & Potgieter, N. (2015). Temporal Variations In Physico-Chemical And Microbiological Characteristics Of Mvudi River , South Africa. 3, 4128–4140.
View ArticleEl-Sheekh, M. M. (2016). Impact Of Water Quality On Ecosystems Of The Nile River.
View ArticleFennessy, M. S., & Cronk, J. K. (1997). The Effectiveness And Restoration Potential Of Riparian Ecotones For The Management Of Nonpoint Source Pollution, Particularly Nitrate. Critical Reviews In Environmental Science And Technology, 27(4), 285–317.
View ArticleGosine, T., & Mahon, R. (2015). Mapping Land-Use , Human Activities And Vegetation In The Conset Bay Watershed , Barbados , To Determine Potential Sources Of Coastal Pollution.
Havel, J. E., Kovalenko, K. E., & Kats, L. B. (2015). Aquatic Invasive Species : Challenges For The Future.
View ArticleHowell, J. A. (2010). The Distribution Of Phosphorus In Sediment And Water Downstream From A Sewage Treatment Works. Bioscience Horizons, 3(2), 113–123.
View ArticleJarvie, H. P., Wade, A. J., Butterfield, D., Whitehead, P. G., Tindall, C. I., & Virtue, W. A. (2002). Modelling Nitrogen Dynamics And Distributions In The River Tweed , Scotland : An Application Of The INCA Model The Tweed Catchment. 6(3), 433–454.
Josephine, K. (2014). Implications Of Climate Change On The Management Of Rift Valley Lakes In Kenya . The Case Of Lake Baringo Implications Of Climate Change On The Management Of Rift Valley Lakes In Kenya . The Case Of Lake Baringo . (June), 133–138.
Kumar, R. P., Ranjan, R. K., AL, R., Singh, S. K., & Srivastava, P. K. (2015). Geochemical Modeling To Evaluate The Mangrove Forest Water. Arabian Journal Of Geosciences, 8(7), 4687–4702.
View ArticleKumar, V. (2011). A Study On Physico-Chemical Charactersitics Of Yamuna River Around Hamirpur (UP), Bundelkhand Region Central India. International Multidisciplinary Research Journal, 1(5), 14–16. Retrieved From Http://Www.Scopemed.Org/?Mno=194172
Maghanga, J. K., Kituyi, J. L., Kisinyo, P. O., & Ng’Etich, W. K. (2013). Impact Of Nitrogen Fertilizer Applications On Surface Water Nitrate Levels Within A Kenyan Tea Plantation. Journal Of Chemistry, 2013.
View ArticleManohara, B. M., Kiran, B. R., & Thirumala, S. (2019). Studies On The Dynamics Of Temperature, Turbidityand Tds In The Water Of Bhadra Reservoir, Karnataka: A Review. 6(4), 15–23. Retrieved From Www.Jetir.Org
Mbui, D., Chebet, E., Kamau, G., & Kibet, J. (2017). The State Of Water Quality In Nairobi River, Kenya. Asian Journal Of Research In Chemistry, 9(11), 579.
View ArticleMcgill, W., Rutherford, P., Figueiredo, C., & Arocena, J. (2010). Total Nitrogen. Soil Sampling And Methods Of Analysis, Second Edition, (415), 3432.
View ArticleMwamburi, J. (2015). Comparative Evaluation Of The Concentrations Of Lead , Cadmium And Zinc In Surficial Sediments From Two Shallow Tectonic Freshwater Lake Basins , Kenya. 9(June), 531–544.
View ArticleNagalakshmi, R., Prasanna, K., & Prakash Chandar, S. (2016). Water Quality Analysis Using Gis Interpolation Method In Serthalaikadu Lagoon, East Coast Of India. Rasayan Journal Of Chemistry, 9(4), 634–640.
Ndiba, R., Omondi, R., Nyakeya, K., Abwao, J., & Oyoo-Okoth, Eo. (2018). Environmental Constraints On Macrophyte Distribution And Diversity In A Tropical Endorheic Freshwater Lake. 6(3), 251–259.
Odada, E. O., Onyando, J., & Obudho, P. A. (2006). Lake Baringo. World Lake Basin Management Initiative: Experience And Lessons Learned Briefs, 1, 31–43.
Odiyo, J. O., Chimuka, L., Mamali, M. A., & Fatoki, O. S. (2012). Trophic Status Of Vondo And Albasini Dams; Impacts On Aquatic Ecosystems And Drinking Water. International Journal Of Environmental Science And Technology, 9(2), 203–218.
View ArticleOlago, D. O., & Odada, E. O. (2007). Sediment Impacts In Africa ’ S Transboundary Lake / River Basins : Case Study Of The East African Great Lakes. 10(1), 23–32.
View ArticleOmondi, R., Yasindi, W. A., & Magana, A. (2011). Spatial And Temporal Variations Of Zooplankton In Relation To Some Environmental Factors In Lake Baringo, Kenya. Eger. J. Sci. Technol, 11(2011), 29–50.
Online, I. P., Islam, S. M. D., & Azam, G. (2015). Seasonal Variation Of Physicochemical And Toxic Properties In Three Major Rivers ; Shitalakhya , Buriganga And Turag Around Dhaka City , Bangladesh. 7(3), 120–131.
Onyando, J. O., Kisoyan, P., & Chemelil, M. C. (2005). Estimation Of Potential Soil Erosion For River Perkerra Catchment In Kenya. Water Resources Management, 19(2), 133–143.
View ArticleOuma, H., & Mwamburi, J. (2014). Spatial Variations In Nutrients And Other Physicochemical Variables In The Topographically Closed Lake Baringo Freshwater Basin (Kenya). Lakes And Reservoirs: Research And Management, 19(1), 11–23.
View ArticleOuyang, Y., Nkedi-Kizza, P., Wu, Q. T., Shinde, D., & Huang, C. H. (2006). Assessment Of Seasonal Variations In Surface Water Quality. 40, 3800–3810.
View ArticlePlessis. (2019). Primary Water Quality Challenges , Contaminants And The World ’ S Dirtiest.
View ArticleRanjan, M., Chun, C., Palani, S., & Tkalich, P. (2013). Temporal Variability And Climatology Of Hydrodynamic , Water Property And Water Quality Parameters In The West Johor Strait Of Singapore. Marine Pollution Bulletin, 77(1–2), 380–395.
View ArticleRask, M., & Ruuhij, J. (2002). Temporal Coherence In Water Temperature And Chemistry Under The Ice Of Boreal Lakes ( Finland ). 36, 3949–3956.
Razier, B. R. E. F., Aimo, T. E. J. N., & Andheinrich, M. A. R. K. B. S. (1996). Un-Ionized Ammonia Nitrogen In Sediment Pore Water From The Upper Mississippi River. 15(2), 92–99.
Republic of Kenya Ministry of Water And Irrigation The National Water Services Strategy ( NWSS ). (2015). (June 2007).
Shabalala, A. N., Combrinck, L., & Mccrindle, R. (2013). Effect Of Farming Activities On Seasonal Variation Of Water Quality Of Bonsma Dam, Kwazulu-Natal. South African Journal Of Science, 109(7–8), 1–7.
View ArticleSingh, A. (2013). Nitrate And Phosphate Contamination In Water And Possible Remedial Measures. Environmenta Problems And Plant, (June), 44–56.
Singh, T. A., Meetei, N., & Meetei, L. (2013). Seasonal Variation Of Some Physico-Chemical Characteristics Of Three Major Rivers In Imphal, Manipur: A Comparative Evaluation. Current World Environment Journal, 8(1), 93–102.
View ArticleSYMADER, W., & BIERL, R. (2010). The Heterogeneity Of Runoff And Its Significance For Water Quality Problems. Hydrological Sciences Journal, 43(1), 103–113.
View ArticleTian, Z., Zheng, B., Wang, L., Li, L., Wang, X., Li, H., & Norra, S. (2017). Long Term (1997-2014) Spatial And Temporal Variations In Nitrogen In Dongting Lake, China. Plos ONE, 12(2), 1–16.
View ArticleUmarani, K., Shanthi, K., Lakshmanaperumalsamy, P., Ayyasamy, P. ., & Usharani, K. (2011). Physico-Chemical And Bacteriological Characteristics Of Noyyal River And Ground Water Quality Of Perur, India. Journal Of Applied Sciences And Environmental Management, 14(2).
View ArticleUNEP. (2010). Fresh Water For The Future.
UNEP. (2016). A Snapshot Of The World’s Water Quality : Towards A Global Assessment. United Nations Environment Programme, Nairobi, Kenya. 162pp. In A Snapshot Of The World’s Water Quality.
View ArticleVarol, M., Gökot, B., Bekleyen, A., & En, B. Ş. (2011). Water Quality Assessment And Apportionment Of Pollution Sources Of Tigris River ( Turkey ) Using Multivariate Statistical Techniques — A Case Study.
View ArticleVenkatesharaju. (2010). Physico-Chemical And Bacteriological Investigation On The River Cauvery Of Kollegal Stretch In. 6(I), 50–59.
Vvaishali, P., & Punita, P. (2013). Assessment Of Seasonal Variation In Water Quality Of River Mini , At Sindhrot, Vadodara. International Journal Of Environmental Sciences, 3(5), 1424–1436. Http://doi.org/10.6088/Ijes.2013030500013aishali, P., & Punita, P. (2013). Assessment Of Seasonal Variation In Water Quality Of River Mini , At Sindhrot, Vadodara. International Journal Of Environmental Sciences, 3(5), 1424–1436.
View ArticleWeselak, M., Arbuckle, T. E., Wigle, D. T., & Krewski, D. (2007). In Utero Pesticide Exposure And Childhood Morbidity. Environmental Research, 103(1), 79–86.
View ArticleWilbers, G. J., Becker, M., Nga, L. T., Sebesvari, Z., & Renaud, F. G. (2014). Spatial And Temporal Variability Of Surface Water Pollution In The Mekong Delta, Vietnam. Science Of The Total Environment, 485–486(1), 653–665.
View ArticleWorld Health Organization. (2011). Guidelines For Drinking-Water Quality 4th Ed., WHO, Geneva, P. 340. World Health Organization. 00006-6
View ArticleWorld, S., & Vol, J. (2010). Nitrate And Phosphate Levels In River Jakara , Kano State , Nigeria . Science World, 5(3), 23–27.
Xiaolong, W., Jingyi, H., Ligang, X., & Qi, Z. (2010). Spatial And Seasonal Variations Of The Contamination Within Water Body Of The Grand Canal, China. Environmental Pollution, 158(5), 1513–1520.
View ArticleZamparas, M. G. (2015). Development Of Physicochemical Methods For Restoring Eutrophic Water Bodies. 187.
