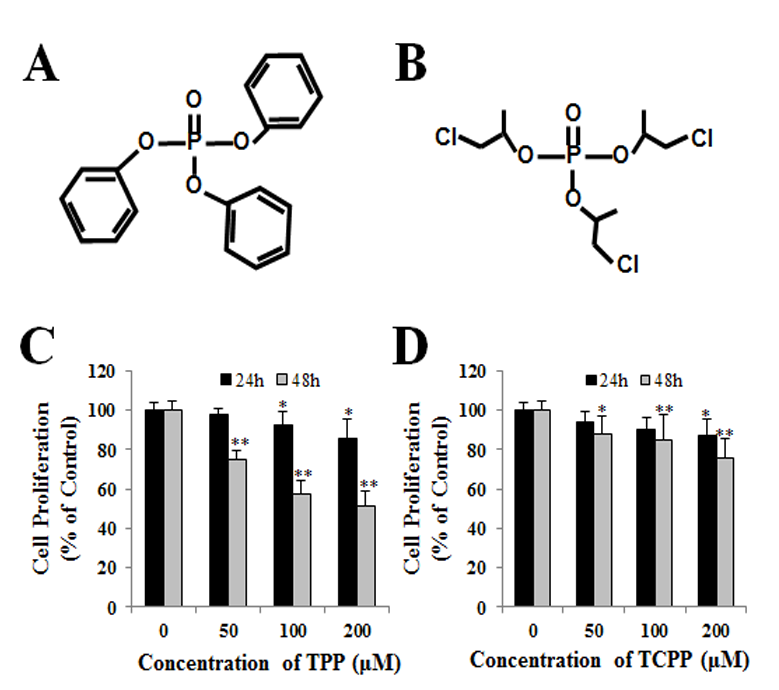
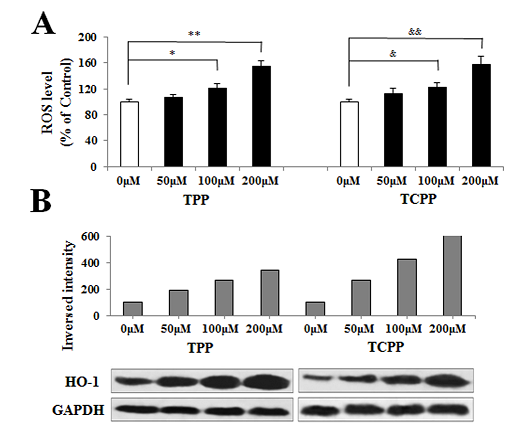
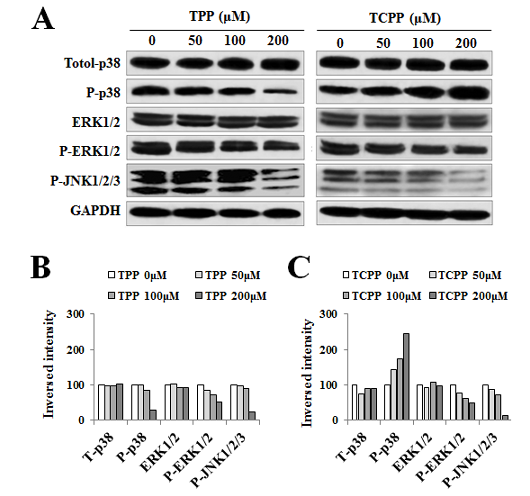
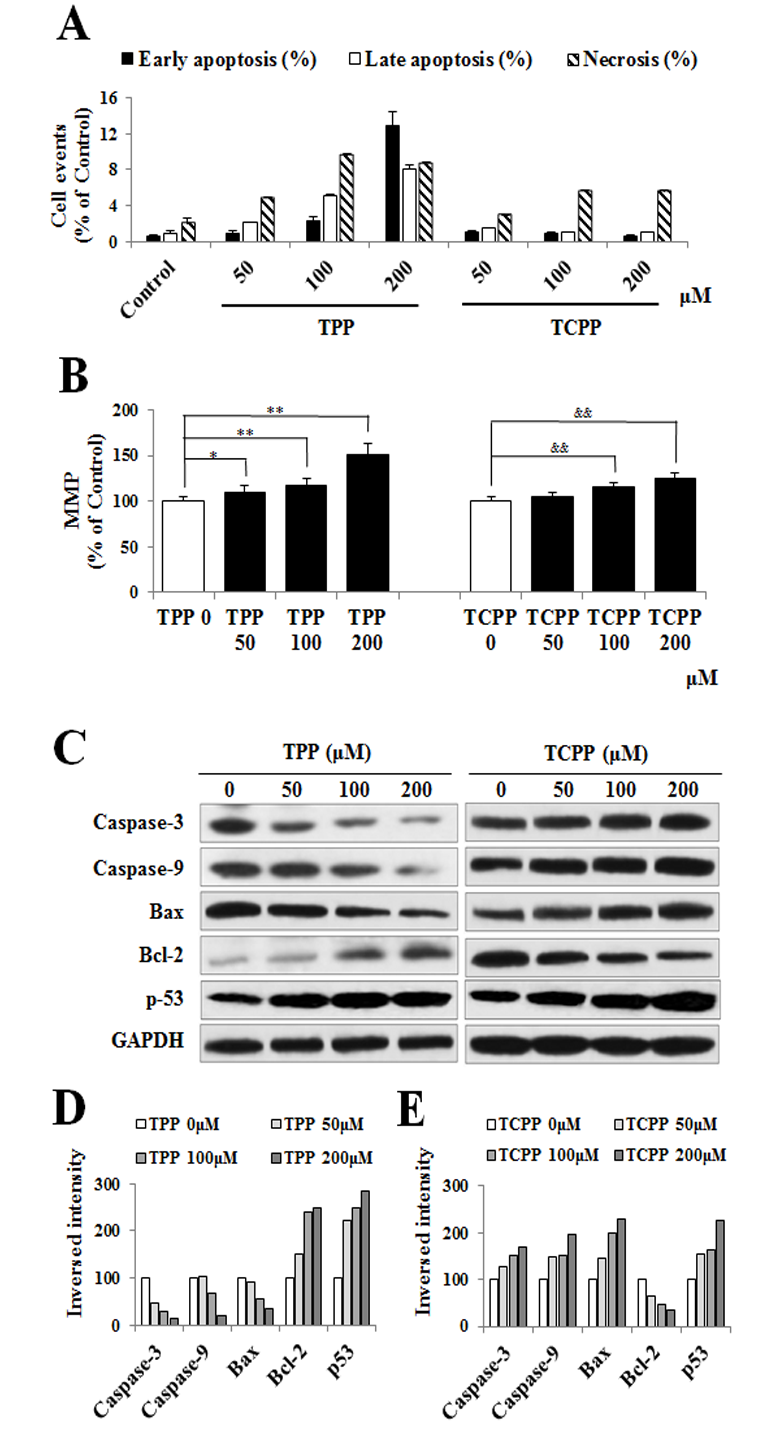
Jing An1, Yu shang2
Institute of Environmental Pollution and Health, School of Environmental and Chemical Engineering, Shanghai University, Shanghai, China;
1Phone:+86-021-66137736; Fax:+86-021-66137736; E-mail: peace74839@shu.edu.cn. ,
2Phone:+86-021-66137736; Fax:+86-021-66137736; E-mail: yushang@staff.shu.edu.cn