J. THEO KLOPROGGE
E-mail: j.kloprogge@uq.edu.au
X-ray Photoelectron Spectroscopy study of so-called “Larimar”, blue pectolite from the Dominican Republic
Corresponding Author
Affiliation
- THEO KLOPROGGE,* and BARRY J WOODb
aSchool of Earth Sciences, The University of Queensland, Brisbane, Qld 4072, Australia
bCentre for Microscopy & Microanalysis, The University of Queensland, Brisbane, Qld 4072, Australia
Citation
Jacob Teunis Kloprogge (Theo), X-ray Photoelectron Spectroscopy study of so-called
Abstract
A detailed analysis was undertaken of the X-ray photoelectron spectra of the blue variety of pectolite known by the tradename Larimar from the Dominican Republic in an attempt to elucidate the origin of the blue colour. The survey scan confirmed the chemical composition of pectolite. The O 1s showed a complex set of bands associated with Si-O-Si, Si-O-(Na,Ca) and OH in the crystal structure of pectolite. From the possible elements suspected to cause the colour, such as Cu, Fe, Mn, and V, only Cu and Fe were observed above the detection limit of the XPS instrument. For the first time about 0.02 atom% of Pb was observed was observed in one sample, while another sample from the same block showed the presence of 1.05 atom% Sb. The possible presence of [Pb-Pb]3+ pairs can act as a chromophore analogous to that observed in the blue-green feldspar variety amazonite. This would explain the till now unexplained optical band at 630 nm as well as part of the high hydroxyl concentration. Though other chromophores cannot be excluded, the observation of Pb adds a new possible explanation for the blue colour of Larimar.
Introduction
A blue pectolite (NaCa2Si3O8)(OH) , known under the trade-name Larimar, is found in the Sierra de Baoruco, Barahona Province, southern Dominican Republic [1,2]. Pectolite is a triclinic inosilicate with a structure similar to that of wollastonite [3]. The mineral was originally thought to be discovered in 1974 by a Peace Corps volunteer named Norman Rilling. The material was originally named Travelina by Miguel Mendez of Santo Domingo, but was later changed to Larimar, a combination of the first part of Larissa (Mendez’s daughter) and mar (Spanish for sea, whose colour the material resembles)[1].The mine is located about 10 km north of the main road from Barahona to Bahoruco, about 200 km west of Santo Domingo. The pectolite forms as a hydrothermal alteration product in veinlets, stringers, and irregular masses within an ultramafic volcanic rock (serpentinised basalt)[4]. Associated minerals identified are calcite, zeolites (in particular natrolite), chalcedony, and hematite [4].
The major unanswered question so far has been the origin of the blue colour that only appears at this one particular location. It has been reported that the pectolite contains small particles of native copper and red plumes of iron [1,2,5,6]. Also mentioned is the presence of small amounts of Mn [3], Cr, and V [4] as possible sources for the colour . Limited spectroscopic measurements show a significant intensity maximum at 480-400 nm together with a smaller maximum at 630 nm [2,4]. The first band is associated with the blue colour spectral range suggested to come from Cr3+ or V3+, while no explanation has been given for the second one [4]. Bente et al. [4] showed a direct correlation between the vanadium content and the intensity of the blue colour of the pectolite. In contrast, Woodruff, Fritsch [2] suggested that the absorption might be due to processes involving multiple atoms, probably within small clusters of Cu2+ ions, though the details of the electronic transitions remain unknown. Fluorescence was a very chalky green to long-wave UV for blue pectolite but the intensity became weaker with increasing blue colouring. The greenish pectolite showed yellow fluorescence to long-wave UV. Fluorescence to short-wave UV was much more homogenous and slightly more intense turbid green independent of colour [2]. Correcher et al. [7] used laser-induced fluorescence emission spectroscopy to study the possible origin of the colour. The blue-coloured zones showed the presence of two bands at 510 nm and 810 nm that were assigned to Mn2+ and Cr3+, respectively. Sullasi et al. [8] showed by energy-discriminating X-ray fluorescence the presence of K, Ca, V, Mn, Ni, and Fe in the light blue regions, while in the brown regions additional elements such as Cr, Sr, and Ti were observed. The thermoluminescence emission spectrum exhibited a band between 500 nm and 600 nm. The presence of the second band around 810 nm was not observed here as no Cr was found in this blue sample, in contrast to the results of Correcher et al. [7] who assumed Cr3+ as the fluorescence activator for this band.
X-ray Photoelectron Spectroscopy (XPS) is widely used for studying the surface properties and compositions of solid materials, including minerals [e.g. 9,10]. In this paper an attempt is made to study the blue pectolite (Larimar) with X-ray Photoelectron Spectroscopy in order to find the possible source of the blue colour. Besides the major elements associated with pectolite (Na, Ca, Si, O) special attention was paid to the presence of Cu, Fe, Mn, and Cr.
Materials & Methods
The material used in this study is part of one of authors’ collection (Kloprogge) and consists of a block approximately 10 x 5 x 5 cm and shows a variety of colours from white via light blue to a gentle dark blue. The pectolite forms botryoidal and spherulitic as well as irregular aggregates. The material is very dense and tenacious with Mohs hardness up to approximately 6.
The XPS analyses were performed on freshly crushed samples on a Kratos AXIS Ultra with a monochromatic Al X-ray source at 225 W under ultrahigh vacuum conditions. The samples were finely crushed and, using a clean SS spatula a small amount of sample was loaded onto a special double sided adhesive tape covering ~ 5mm diameter. The area examined by XPS is ~0.7 mm X 0.3 mm. Typical duration for the powders being exposed in air (prior to evacuation in the sample load-lock chamber) was less than 10 min. The samples were not Argon Ion Beam cleaned because the use of Argon Ion Beam cleaning in most cases results in ion-induced damage to the sample, in particular ion-beam reduction. Prior to the analysis the samples were outgassed overnight under vacuum. Each analysis started with a survey scan from 0 to 1200 eV with a dwell time of 100 milliseconds, pass energy of 160 eV at steps of 1 eV with 1 sweep. For the high resolution analysis the number of sweeps was increased, the pass energy was lowered to 20 eV for the major elements and 40 eV for the trace elements at steps of 100 meV and the dwell time was changed to 250 milliseconds. The Kratos Axis Ultra, XPS used for the analysis, has a built-in patented coaxial low energy electron charge compensation system which provides a high flux of electrons of uniform charge density. It uses a magnetic immersion lens situated below the sample and the low energy electrons from a filament located at the base of the photoelectron input lens are injected into the magnetic field. It is operated in such a way that ‘overcompensation’ occurs, which results in full charge neutralization and the photoelectron peaks move down scale a few eV. The principal C 1s peak from advantageous carbon is used as the reference at 284.8 eV to calibrate the spectra.
Results
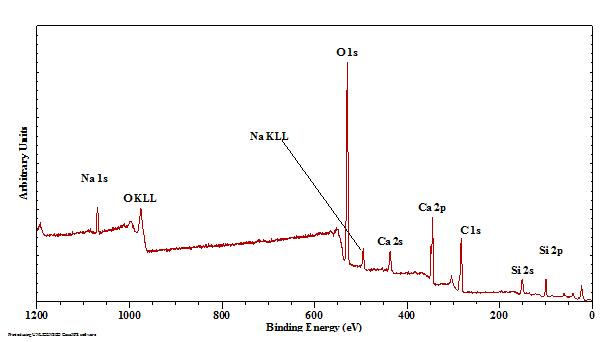
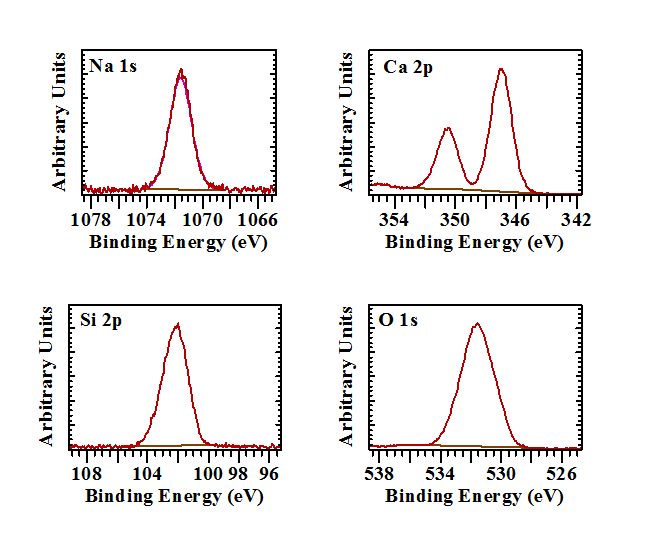
The XPS survey scan of Larimar is characterised by the major elements Na, Ca, O, and Si as expected for pectolite (Figure1). In agreement with the XRD results no other minerals are present. A minor amount of advantageous carbon can be observed due surface contamination. Besides the in this study used Si 2p and Al 2p bands also the Si 2s and Al 2s can be observed around 152 and 118 eV, respectively. Table 1 shows the exact binding energies and atom percentages for the major elements. The ratio of Na:Ca:Si is about 1.0:2.2:3.0, which is close to the ratio of 1:2:3 for pectolite (Table 1). Survey scans of the lighter areas showed a local existence of a compound consisting of Ca, O and C, identified by XRD as calcite and another silicate mineral with higher Na content, Si, Al and O but no Ca. Earlier work has shown the possible presence of natrolite and it is suggested that this survey scan agrees with that identification [4].
Pectolite is a silicate of calcium and sodium, both large cations, and therefore has to deal with the size issue by tilting the tetrahedra until they link up. Unlike wollastonite, pectolite includes sodium atoms that separate strips of Ca-O octahedra. The binding energy of the Si 2p is in the range characteristic for inosilicates. The Si atoms are tetrahedrally coordinated to four oxygen atoms and form Si-O-Si linked in single chains. Analyses indicate that the spectrum has a single Gaussian peak with a binding energy of 102.2 eV, suggesting that the electronic environment around Si atoms in pectolite is different from the higher polymerized SiO4 units in phyllo- and tectosilicates resulting in a considerably shift compared to phylloslicates such as kaolinite (103.1 eV) and tectosilicates such as quartz (α-SiO2, 103.9 eV) [10].
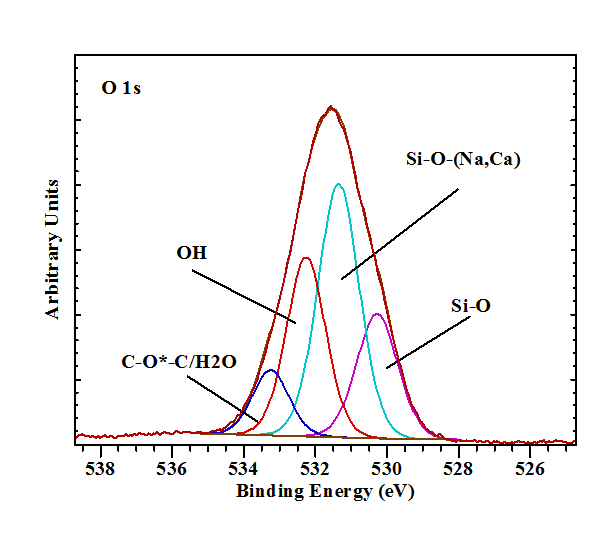
The shape of the O 1s spectrum is rather broad and indicative of the presence of a variety of different oxygen atom environments (Figure 3). First of all there is the presence of a small amount of oxygen associated with the carbon on the surface (C-O*-C, 533.1 eV). The second distinction that can be made in the crystal structure of pectolite is that between the hydroxyl groups and structural oxygen. It is suggested that the O 1s with a binding energy of 532.2 eV is associated with not only the structural OH (4.5 at%) but also with a significant amount of surface hydroxyls (14.7 at%). The structural oxygen is split in two separate bands with slightly different binding energies of 530.2 eV and 531.3 eV at a ratio of 2 to 7. The coordination of the oxygen atom to 0, 1, or 2 Na, or Ca atoms has a distinct effect on the Si-O bond distances and the Si-O-Si angles. Based on the crystal structure four main types of oxygens can be distinguished based on the cations they are linked to: O1 to 1 Si and 2 Ca; O2 to 1 Si, 2 Ca, and 1 Na; O3 and O4 to 1 Si, 1Ca, and 1 Na; O5 and O6 to 1 Si and 3 Ca; O7 and O8 to 2 Si and 2 Na and finally O9 to 2 Si. The Si-O bond lengths are significantly shorter than the Ca-O and Na-O bond lengths. Since the charge density difference for each oxygen atom may not vary significantly, it is not expect to be able to resolve all these binding energy differences. The one oxygen that stands out from the other oxygen atoms is O9 that links the two adjacent SiO4tetrahedra. Based on this it suggested that the band with a binding energy of 530.2 eV is associated with this tetrahedral linking oxygen, while the other band at 531.3 is associated with the other oxygen atoms that are linked not only to Si but also to Na and/or Ca. It seems that the effect of the Ca2+or Na+ on the Si-O linkages is very limited and the different oxygen environments cannot be distinguished.
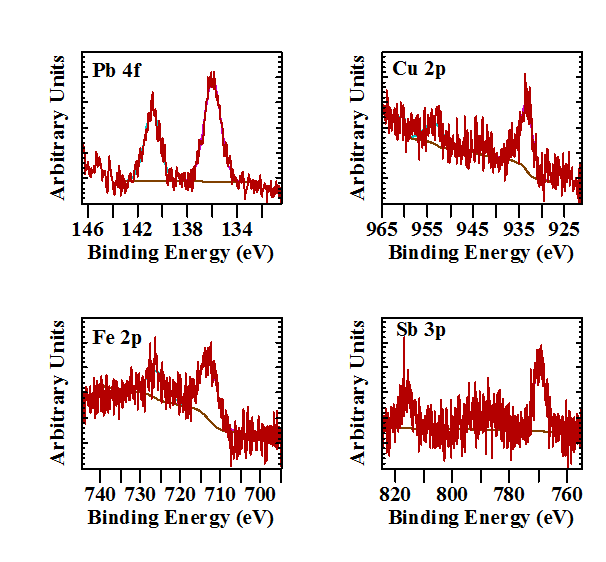
The high resolution spectra of trace elements mentioned in the literature such as Cu and Fe varied per sample. In none of the samples Mn, Cr, Ni, or V was observed. If these elements are present in Larimar they are below the detection limit of the XPS instrument. However in one sample besides a trace of Fe a trace amount of Pb was observed. Figure 4 shows the Pb 4f spectrum. The amount present is in the order of 0.02 atom%. Lead has not been mentioned before as a possible element to cause the blue colour in Larimar. However it has been suspected that trace amounts of lead play a role in the blue-green colour of the feldspar variety amazonite. The unexplained band at 630 nm observed in the optical spectrum of Larimar can be related to the presence of a [Pb-Pb]3+ pair. Earlier work has shown that the [Pb-Pb]3+ pair in amazonite gave a band at this same position [11]. It is interesting that an earlier work on amazonite suggested the substitution of 2 K+ + O2- → Pb2+ + 2 OH-, where the increase in Pb was directly associated with an increase in OH content and that intensely coloured amazonite could have as much as ten times the amount of water compared to the white/yellowish microcline (KAlSi3O8) [12]. This mechanism, if correct, could partly explain the high OH content observed in the O 1s high resolution spectrum.
Another sample from the same block did not show the presence of Pb, but instead it showed the presence of Fe, Cu and surprisingly Sb (Figure 4). Iron is present at a concentration of 0.55 atom%, Cu at 0.27 atom% and Sb at 1.05 atom%. The normally used Sb 3d lines could not be used for the concentration determination as the two weak lines are completely invisible due to strong interference of the intense O 1s signal in the same position. However, the Sb 3p lines are clearly visible. Manganese, chromium, nickel, and vanadium were not observed in this sample.
Cu2+ is a well-known chromophore leading to the blue colour in many minerals (idochromatic with Cu a major part of the mineral structure, e.g. azurite, chrysocolla, or as impurity, i.e. allochromatic e.g. blue smithsonite, aragonite, Paraiba tourmaline). In the case of pectolite Cu2+ is present as a trace and its concentration can be related to the intensity of the blue colour. In Larimar the situation seems to be more complex as other metals can act as chromophores as well. Fe as Fe2+ - Fe3+ Intervalence Charge Transfer (ICT) between two adjacent metal ions usually causes blue-green coloration. They are a major cause of colour in a variety of common and uncommon minerals, e.g. rockbridgeite and vivianite. The Fe2+ - Fe3+ interaction typically results in absorption of light in the red portion of the spectrum. Consequently, most minerals with this interaction are either blue or green when thin. A similar mechanism can play a role causing the blue colour of Larimar. The actual situation is probably a mix of different chromophores acting at the same time. Besides copper and iron as chromophores the combination of Fe and Pb, as observed in amazonite cannot be excluded. Foord, Martin [13] for example attributed the bluish-green amazonite colour to the charge transfer Pb2+ + Fe3+ - Pb3+ + Fe2+. Though not observed in this study, the presence of Mn, Ni, Cr or V can also act as blue chromophores. Antimony does not act as a chromophore in the pectolite and seems to be present just as a trace impurity in certain parts of the Larimar material.
Conclusion
The XPS survey scans and high resolution scans of individual elements have shown that blue Larimar has the same composition as pectolite. The whiter regions in between are composed of mixtures of pectolite and natrolite to pure natrolite, while other regions consist of calcite. The high resolution spectrum of in particular O 1s shows the existence of various species that can be linked to Si-O-Si, Si-O-(Na, Ca), OH and C-O*-C from surface contamination. The high resolutions scans of the elements Mn, and V did not show the presence of these elements above the detection limit of the XPS instrument. However, for the first time a trace amount of Pb was observed in one sample. It is possible that the [Pb-Pb]3+ pair act as a chromophore, similar to that observed in amazonite. This would explain the optical band at 630 nm as well as the high amount of hydroxyl groups compared to the number of hydroxyl groups in the unit cell of pectolite. Other samples showed the presence of Cu, Fe and for the first time Sb. Both copper and iron play a role as chromophores creating the blue colour of Larimar. Overall, the blue colour is probably a mixture of at least three different types of chromophores, all present in varying degrees within even a single sample, causing the changes in intensity of the blue colour over even short distances (in the order of mm to cm).
Acknowledgement
The authors acknowledge the facilities, and the scientific and technical assistance, of the Australian Microscopy & Microanalysis Research Facility at the Centre for Microscopy and Microanalysis, The University of Queensland.
Images and Tables
|
Element |
Binding energy (eV) |
Atom Percentage |
|
Na 1s |
1071.6 |
4.5 |
|
Ca 2p3/2 |
347.0 |
9.7 |
|
Si 2p |
102.2 |
13.3 |
|
O 1s Si-O |
530.2 |
10.1 |
|
O 1s Si-O-(Na,Ca) |
531.3 |
35.4 |
|
O 1s OH |
532.2 |
19.2 |
|
O 1s C-O*C |
533.1 |
6.8 |
References
Woodruff RE: Larimar, beautiful, blue - and baffling. Lapidary Journal 1986, 39: 26-32.
View ArticleWoodruff RE, Fritsch E: Blue pectolite from the Dominican Republic. Gems & Gemology 1989, 1989: 216-225.
View ArticleSchmetzer K: Pektolith aus der Deominikanischen Republik. Zeitschrift der Deutschen Gemmologischen Gesellschaft 1984, 33: 63-64.
Bente K, Thum R, Wannemacher J: Colored pectolites, so-called "Larimar" from Sierra de Baoruco, Barahona Province, southern Dominican Republic. Neues Jahrbuch fur Mineralogy, Monatshefte 1991, 1991: 14-22.
Lizzadro J: The interesting story of a new blue gem material called Larimar. Lizzadro Museum 1987: 13-14.
View ArticleFritsch E, Rossman GR: An update on color in gems. Part 3: colors caused by band gaps and physical phenomena. Gems & Gemology 1988, 24: 81-103.
View ArticleCorrecher V, Carcia-Guinea J, Castillejo M, Oujja M, Rebollar E, Lopez-Arce P: Laser-induced fluorescence and thermoluminescence response of a Na-Ca rich silicate. Radiation Measurements 2006, 41: 971-975.
View ArticleSullasi HL, Khoury HJ, Barros V, Libonati R, Guzzo PL, Asfora V, De Araujo RE, Capriles M, Reyes J: Thermoluminescence response of the larimar rocks. Radiation Measurements 2010, 45: 540-542.
View ArticleKloprogge JT, Duong LV, Wood BJ, Frost RL: XPS study of the major minerals in bauxite: Gibbsite, bayerite and (pseudo)boehmite. Journal of Colloid and Interface Science 2006, 296: 572-576.
View ArticleKloprogge JT, Wood BJ: Chemical bonding and electronic structures of the Al2Si2O5(OH)4 polymorphs kaolinite, dickite, nacrite and halloysite by X-ray Photoelectron Spectroscopy. Clay Science 2015, 19: 39-44.
View ArticlePetrov I, Mineeva RM, Bershov LV, Agel A: EPR of [lPb-Pb 3+ mixed yalence pairs in amazonite-type microcline. American MIneralogist 1993, 78: 500-510.
View ArticlePlyusnin GS: On the coloration ofamazonite (in Russian). Zapiski Vsesoyuznogo Mineralogicheskogo Obshchestva 1969, 98: 3-17.
Foord EE, Martin RF: Amazonite from the Pikes Peak Batholith. Mineralogical Record 1979 I 0,: 373-382.
View Article
