Ravi Gooneratne
Tel. +64 3 423 0636
E-mail address: Ravi.Gooneratne@lincoln.ac.nz
© 2019 Sift Desk Journals. All Rights Reserved
VOLUME: 4 ISSUE: 2
Page No: 629-644
Ravi Gooneratne
Tel. +64 3 423 0636
E-mail address: Ravi.Gooneratne@lincoln.ac.nz
Nadir Saleeb a, Brett Robinson b, Jo Cavanagh c, A K M Mofasser Hossaina, Ravi Gooneratne a,*
a Wine, Food and Molecular Biosciences Department, PO Box 85084, Lincoln University, Lincoln 7647, New Zealand
b College of Science, University of Canterbury, Private Bag 4800, Christchurch 8140, New Zealand
c Manaaki Whenua Landcare Research, PO Box 69040, Lincoln 7640, New Zealand
Cuncang Jiang(jcc2000@mail.hzau.edu.cn)
Chongping Huang(hcping@zju.edu.cn)
Snjezana Mihaljevic(mihaljev@irb.hr)
Lyuben I Zagorchev(lzagorchev@biofac.uni-sofia.bg)
Ravi Gooneratne et al., Biochemical changes in sunflower plant exposed to silver nanoparticles / silver ions(2019) SDRP Journal of Food Science & Technology 4(2)
When soil is contaminated with silver (Ag), plants take up Ag and is concentrated in roots and leaves, with its effects reflected in crop health and yield. This study investigated the toxicity of silver nanoparticles (AgNPs) and silver nitrate (AgNO3 as Ag+) to sunflower seeds grown in soils amended with 150 mg/kg of Ag either as AgNPs or AgNO3. Exposure of the sunflower seeds to soils amended with Ag increased plant lipid peroxidation, activities of antioxidants enzymes (catalase, superoxide dismutase, glutathione-S-transferase), peroxidases (glutathione peroxidase pyrogallol peroxidase, guaiacol peroxidase), oxidases (ascorbate oxidase), urease, total phenolic compounds, vitamins (retinols, alpha-tocopherol and L-ascorbic acid) but inhibited chlorophyll, total carotenoids, total soluble carbohydrates, phenolic compounds and total soluble proteins. In general, AgNO3 increased the above-mentioned parameters in sunflower more than did AgNPs, except for the tested vitamins, which were more affected by AgNPs. The results showed that Ag accumulation in the root > leaf > stem and human food security risk is enhanced in sunflower seeds exposed to Ag compounds.
Keywords: Antioxidants, Chlorophyll, Silver ions, Silver nanoparticles, Sunflower, Total soluble protein, Silver toxicity, Oxidative stress.
Silver nanoparticles (AgNPs) are one of the most widely used industrial nanomaterials and are reported to be an efficient antimicrobial agent [1]. The special characteristics of AgNPs that have led to their large-scale use in a variety of consumer products are also those which raise concern. Although sizes vary, by definition AgNPs are between 1 and 100 nm in three dimensions, with positive or negative charge depending on their coating material [2]. In general, nanoparticles (NPs) have a higher surface-to-volume ratio, which increases their reactivity with biological fluids [3]. The increased use of AgNPs in different industries may cause toxic effects on the environment because of improper discharge of wastes, and they may end up as contaminants in soil when sewage sludge is used as a fertilizer, with uncertain effects on plant crops and may cause soils to become infertile [4, 5].
The sunflower (Helianthus annuus L.; Asteraceae) is an herbaceous annual, grown commercially for its edible oil-bearing seeds. Each flower head produces hundreds of grey-white conical-shaped seeds, which are an excellent source of health benefitting nutrients like water- and fat-soluble vitamins. Each 100 g of seeds contains c. 35.17 g of alpha-tocopherol, essential macro-elements such as magnesium (Mg) and phosphorus (P), and micro-minerals such as copper (Cu), manganese (Mn), and selenium (Se). Packed with essential polyunsaturated fatty acids (linoleic acid, oleic acid), sunflower seeds are also a good source of proteins and amino acids that are essential for growth. Because sunflower plants are to some extent able to extract toxic elements from soil, they have been planted in contaminated soils to clean the soil from toxic elements [6]. However, exposure of sunflowers to AgNPs/Ag+ is known to cause oxidative stress and create reactive oxygen species (ROS), and as a result the activity levels of antioxidant defence enzymes and lipid peroxidation (LPO) increase.
Studies have looked at the effect of other heavy metals on sunflower and an excess of Fe, Cu or Cd has been found to produce oxidative damage in sunflower leaves [7]. Treatment of sunflower with 0.5 mM iron (Fe2+), Cu2+ or cadmium (Cd2+) for 12 h increased LPO and lipoxygenases when exposed to sunlight, but decreased chlorophyll A and B and glutathione (GSH) concentrations by 30%, 40%, and 15% respectively [7]. Free radical scavengers such as sodium benzoate or mannitol protected many of these enzymes from inactivation, but not superoxide dismutase (SOD). Exposure of sunflower to Fe2+ and Cd2+ ions decreased SOD while the Cu2+ ions increased SOD. However, all these metals increased a variety of other antioxidant enzymes such as catalase (CAT), ascorbate peroxidase, glutathione reductase and dehydroascorbate reductase.
Sunflower plants exhibit a strong potential to form Ag+ from AgNPs compared to Basella alba (Basellaccea), Oryza sativa, Saccharum officinarum, Sorghum bicolor and Zea mays (Poaceae) [8]. Sewalem et al. 2014 [9], studying the role of sunflower as a potential phytoremediator of heavy metals, found that 88.8% of Cd accumulated in roots, inducing a low level of LPO, membrane leakage and poor root growth, whereas 71.4% of Pb was translocated to the shoots and hence affected photosynthesis.
Silver nanoparticles at concentrations of 20, 40, 60, 80 and 100 mg kg-1 impacted on plant growth parameters such as leaf surface area, chlorophyll, and carbohydrate, all of which affected growth of the common bean (Phaseolus vulgaris) and corn (Zea mays), promoting plant growth at low doses and inhibition at high concentrations [10].
Exposure of Bacopa monnieri to Ag+ and AgNPs enhanced peroxidase and catalase activity [11]. Qian and colleagues [12] studied the effect of Ag on the transcription of antioxidant and aquaporin genes, and the balance between antioxidant and oxidant status, and concluded that AgNPs were more toxic than Ag+ (in AgNO3). When watermelon plants were exposed to 0, 30, 60, 90 and 200 mg L-1 of Ag+ applied at intervals of 8 days throughout the crop cycle, a dose responsive increase in Ag concentration occurred in both roots and shoots and the amount of antioxidant in fruits increased by 3-fold in those exposed to 30 mg L-1 Ag+ but the lycopene concentration declined [13].
This study therefore aimed to investigate the effects of AgNPs and Ag+ (as AgNO3) on the sunflower plant and compared their effect on the activities of antioxidant defence enzymes [SOD, CAT, glutathione peroxidase (GPx), glutathione-S-transferase (GST)], other peroxidases (guaiacol peroxidase, pyrogallol peroxidase), ascorbate oxidase, urease, and LPO, antioxidant vitamins (A, E and C), chlorophyll A and B, total carotenoids, carbohydrate and protein concentration.
2.1. Silver nanoparticles synthesis and quantification
AgNPs were freshly prepared on the day of the experiments. Citrate-coated AgNPs were synthesized from AgNO3, reduced by ferrous sulphate hepta-hydrate, and coated with tri-sodium citrate dihydrate [14]. This method produced spherical-shaped particles, which did not aggregate due to the electrostatic repulsion of the citrate coating agent.
The Ag concentration in AgNPs was determined by flame atomic absorption spectroscopy (FAAS) (Model A.6200, Shimadzu, Tokyo, Japan), calibrated with the Merck ICP certiPUR multi-element standard (Sigma-Aldrich, Castle Hill, New South Wales).
2.2. Charge measurement and particle size
The average mean particle size and zeta potential of AgNPs were determined using a Zetasizer Nano ZS (Malvern Instruments, Worcestershire, UK) at 25°C. The conditions employed were: He/Ne laser (wavelength = 633 nm), scattering angle 90°, refractive index 1.33, and viscosity 0.887 mPa. Prior to the measurements, the concentration of the AgNPs was diluted 200-fold with water.
Silver nanoparticle morphology and size were determined using Transmission Electron Microscopy (TEM). Silver nanoparticles were diluted with deionized water to obtain a more uniform distribution and transferred directly onto a TEM gold-carbon grid for size characterization, using a high-resolution TEM (Philips CM200, Amsterdam) fitted with a Gatan digital camera.
2.3. Soil amendment and plant sampling/preparation
Templeton Silt Loam (TSL) soil (Table 1) (c. 100 kg) was collected from the Johnson Memorial Laboratory grounds, Lincoln University, Canterbury, New Zealand (43°38'44.67"S, 172°27'3.19"E), sieved and dried. The soil was divided into three portions: one portion was mixed with 150 mg/kg of Ag as AgNPs, the second with an equivalent amount of Ag as AgNO3. (This dose was chosen based on a study by N. Saleeb, which found that Ag at 200 mg/kg was mildly toxic to ryegrass) and the third portion was not treated (control). Four 8-L pots per treatment (control, AgNPs, AgNO3; n = 12) were each filled with 5 kg of soil from their respective portion (c. 20 kg per treatment).
Three sunflower seeds (Helianthus annus, family Asteracear) were planted in each pot then water added to field capacity. Pots were watered daily to field capacity. After 2 weeks, two sunflower plants were removed from each pot, leaving one per pot. The plants were harvested after 53 days, the roots separated, and the stem divided into four equal sections (labelled from the base to the top of the plant; S1, S2, S3, S4). All leaves from each plant were collected along with the flower, and seeds. The different sets of plant parts were then allocated to two groups. One half of each set of plant parts was kept in plastic bags and stored at -200C for measurement of LPO, total soluble protein, total phenolic compounds (TPC), total soluble carbohydrates, chlorophyll A and B, total carotenoids, antioxidant vitamins A, E and C, and activities of antioxidant enzymes, urease, and peroxidases (POX). The frozen sunflower leaves were homogenized with liquid nitrogen (N2) and the powder stored at −20°C until analysis. Leaf material (1 g) in liquid N2 was homogenized with 3 ml of 0.1 M phosphate buffer (pH 6.8) using a homogenizer (YunlinLi FJ-200, Shanghai, China) in ice and centrifuged (Beckman J2-MI, USA) at 16,000 rpm (30,390g RCF) for 20 min at 4°C and the supernatant collected for antioxidant enzyme analysis. All the parameters were measured using spectrophotometry (Shimadzu UV-VIS 1280, Tokyo, Japan). The other half was dried at 65°C for 72 h and then powdered in a tissue analyzer (Tissuelyser II Qiagen, USA). The powdered plant parts were subjected to acid (a mixture of 3 parts of HNO3 and I of HCl acids) digestion to determine the Ag concentration using inductive coupled plasma-optical emission (ICP-OES).
Table 1. Properties of Templeton Silt Loam soil used in the study [15, 16].
|
Parameter |
Mean (SEM) |
|
Sand/silt/clay (%) |
76/20/4 |
|
pH (H2O) |
5.1 |
|
CEC (meq/100 g) |
12.3 |
|
C (%) |
3.3 (0.03) |
|
N (%) |
0.3 (0.00) |
|
P (mg/kg) |
732 (11) |
|
S (mg/kg) |
383 (6) |
|
Ca (mg/kg) |
3329 (58) |
|
Mg (mg/kg) |
3426 (71) |
|
K (mg/kg) |
2541 (279) |
|
Cd (mg/kg) |
0.13 (0.00) |
|
Zn (mg/kg) |
70 (2) |
|
Cu (mg/kg) |
5 (0) |
|
B (mg/kg) |
7.3 (1.0) |
|
Fe (mg/kg) |
17727 (353) |
|
Mn (mg/kg) |
357 (20) |
|
Ag (mg/kg) |
<2 |
2.4. Analytical methods for parameter measurement
The enzyme activities were calculated as reported in supplementary data 1.
Catalase (CAT): Following [17], 50 µl of leaf extract was added to 3 ml of a solution containing 50 mM potassium phosphate buffer (pH 7) and 20 mM hydrogen peroxide (H2O2). The decrease in absorbance was measured at 240 nm wavelength every 30 s for 2 min (Ƹ = 43.6 M/cm).
Superoxide dismutase (SOD): Following [18], 50 µl of leaf homogenate was mixed with 2.95 ml solution of 50 mM potassium phosphate solution (pH 7.8) containing 26 mM L-methionine, 75 µl nitroblue tetrazolium chloride (NBT), 100 µM EDTA and 20 µM riboflavin. The reaction was performed in a chamber under illumination of a 30 W fluorescent lamp at 25°C. The reaction was started by turning the fluorescent lamp on and then turning off after 5 min. The blue formazan produced by NBT photoreduction was measured as an increase in absorbance at 560 nm every 30 s for 2 min (Ƹ = 2640 M/cm).
Glutathione-S-transferase (GST): Based on a conjugation reaction between GST and 1-chloro-2,4-dinitobenzene (CDNB), [19], 0.1 ml of homogenized leaves was added to 2.8 ml of phosphate buffer (pH 6.5) to which 0.1 ml of CDNB was added. The increase in absorbance was recorded at 340 nm, every 30 s for 2 min (Ƹ = 9.6 M/cm).
Glutathione peroxidase (GPx): Based on [20] with slight modifications, 0.4 ml of 0.4 .M sodium phosphate buffer (pH 7) was mixed with 0.1 ml of 10 mM sodium azide, 0.2 ml of 4 mM reduced glutathione, 0.1 ml of 2.5 mM H2O2, 0.2 ml of water and 0.5 ml of leaf homogenate. Then 1 ml of 5,5-dithiobis-2-nitro benzoic acid in 1% sodium citrate reagent was added. The absorbance of the colour developed was measured at 412 nm every 30 s for 2 min (Ƹ = 6,422 M/cm).
Lipid peroxidation (LPO): Based on [21] with slight modifications, this assay was based on measurement of malondialdehyde (MDA), which is formed by the reaction of MDA with thiobarbituric acid. The reaction mixture was prepared by dissolving 3.75 g trichloro acetic acid in 10 ml of 0.25N hydrochloric acid and 0.094 g of thiobarbituric acid in 2 ml of methanol. The two solutions were mixed and completed to 25 ml with 0.25 N hydrochloric acid. Leaf homogenate (1 ml) was mixed with 2 ml of reaction mixture and boiled in a water bath for 15 min, allowed to cool and centrifuged at 4577 RCF for 5 min. The pink colour developed was measured at 532 nm (Ƹ = 163.8 M/cm).
Ascorbate oxidase activity: Based on [22], 8.8 mg of ascorbic acid was dissolved in 300 ml of phosphate buffer (pH 5.6). Then 100 µl of leaf homogenate was added to 3 ml of extraction buffer and the decrease in absorbance measured at 265 nm every 30 s for 5 min (Ƹ = 7 M/cm).
Pyrogallol peroxidase activity: Following [23], 0.1 ml of leaf homogenate was added to the reaction mixture composed of 0.1 M phosphate buffer (pH 6.8) containing 0.05 M pyrogallol and 1% H2O2. The increase in absorbance was measured at 430 nm every 30 s for 2 min (Ƹ = 4.5 M/cm).
Guaiacol peroxidase: Following [24], 2 ml of reaction mixture (20 mM guaiacol, 20 mM H2O2e, 50 mM phosphate buffer; pH 6.8)) was added to 25 ul of leaf extract, and incubated at 30°C for 10 min. The reaction was stopped by adding 0.5 ml of sulphuric acid and the absorbance recorded at 480 nm every 30 s for 2 min (Ƹ = 26,600 M/cm).
Chlorophyll and total carotenoids: Following [25], 0.5 g of homogenized leaf was mixed with 10 ml of methanol for 15 min at 4°C and centrifuged at 4577 RCF (4700 rpm) for 10 min. Then 0.5 ml of supernatant was mixed with 4.5 ml of methanol and analysed for chlorophyll A and B and also total carotenoids. Absorbance was measured at 470 nm for total carotenoids, at 665.2 for chlorophyll A (CA) and at 652.4 nm for chlorophyll B (CB).
Equations used for chlorophyll and total carotenoids calculations are shown in supplementary data 3.
Total soluble carbohydrate (TSC): Following [26], 1 g of homogenized leaf was mixed with 3 ml of 0.1 M phosphate buffer (pH 6.8), centrifuged as described for chlorophyll. Next, 100 µl of supernatant was mixed with 900 µl of phosphate buffer (pH 6.8) followed by addition of 5 ml of concentrated sulphuric acid. The mixture was incubated on a hot block at 50ºC for 10 min. The red colour developed was measured at 490 nm. The concentration of TSC was calculated from a calibration curve using D-glucose as the standard (within the range 0–10 mg/ml).
Total phenolic compounds (TPC): Following [27], 0.2 g of homogenized leaf was mixed with 0.8 ml of ice-cold methanol and incubated at room temperature in the dark for 48 h, then centrifuged at 10,000 × g for 5 min. Next, 0.1 ml of supernatant was mixed with 0.1 ml of Folin-Ciocalteu’s phenol reagent (CF; Sigma-Aldrich). The working reagent was 1 ml of CF reagent and 9 ml of water. The solution was vortexed, 0.8 ml of 700 mM sodium carbonate added and incubated at room temperature for 2 h. The absorbance was measured at 765 nm. Gallic acid was used as the standard (within the range 0–200 ug/g).
Total soluble protein: Following [28] with minor modifications, 950 µl of water was added to 10 µl of leaf sample (1 g of homogenized leaf mixed with 3 ml of phosphate buffer (pH 6.8) to which1 ml of diluted Bradford reagent (1-part reagent + 4 parts water) was added. The solution was mixed and incubated at room temperature for 5 min. Absorbance was measured at 595 nm. The calibration curve was constructed using serum albumin within the range 0.312 to 10 µg/ml.
Urease: The assay was based on the alkaline phenol reaction [29]. Briefly, 0.25 g of leaf sample powder was homogenized with 5 ml of 30% ethanol in a 50-ml centrifuge tube. The sample was vortexed at 300 rpm for 30 min at 8°C and centrifuged at 50,000 × g for 10 min. Next, 20 µl of supernatant was added to 896 µl of hypochlorite solution (12% sodium hypochlorite, 0.4 M Na2HPO4 and 0.37 M NaOH). The pH was adjusted to 12. Then, 84 µl of phenol solution (7% phenol containing 34 mg of sodium nitroprusside) was added. The mixture was incubated at 37°C for 15 min. Difference in absorbance between 630 and 670 nm was measured.
Vitamins A and E: Vitamins A and E were both extracted and measured using an isocratic HPLC with two detectors – UV and fluorescence detection, based on [30] with some modifications. Briefly, 1 g of homogenized leaf was mixed with 5 ml of hexane for 25 s, then centrifuged at 4,577g RCF (4,700 rpm) for 20 min at 4°C. Next, 3 ml of supernatant was evaporated under a gentle stream of N2 at 45ºC. The residue was dissolved in 200 µl of methanol and 20 µl was injected onto an isocratic HPLC (Aligant-3170, Malgrave, Victoria, Australia).
High Performance Liquid Chromatography: The vitamin A and E analyses were performed on Agilent-1100 series HPLC (Victoria, Australia), equipped with a quaternary pump LC-1311 A, a degasser model 1322 A, and auto sampler G 1329 A (all Agilent, Victoria, Australia). The mobile phase was 96% methanol; flow rate =1 ml/min; Columns: A pre-column RP (15 × 3.2 mm; Applied Bio-systems, Foster City, CA, USA), particle size = 7 µm connected to an analytical column (RP, Bio-systems) (220 × 4.6 mm; particle size = 5 µm). The column was maintained at 35ºC then connected to an Agilent UV diode array detector monitoring at 325 nm and a fluorescence detector (model 1321 A), operated at 292ex nm and 330em nm. The whole system was linked to a PC computer operated by Agilent (Chemistation model LC 3 D) and to an ALM thermostat (model G13308).
Vitamin C: Following [31 ], 0.25 g of sunflower powder was homogenized with 2 ml of 10% trichloroacetic acid, and centrifuged at 4577 g RCF for 10 min. Next, 0.5 ml of supernatant was mixed with 1 ml of Folin-Ciocalteu reagent (diluted 10-fold with distilled water) and 0.5 ml of 20% sodium carbonate. The solution was incubated at room temperature for 10 min and absorbance measured at 765 nm.
2.5. Statistical analysis
Data are presented as the mean ± SE (n =4). Data were analysed using one-way analysis of variance (ANOVA). The Minitab v17 statistical program software was used to compare the differences among treatment groups. Significance level was set at p < 0.05. Explanatory variables were nominal concentration of chemicals (Ag+ and AgNPs) in mg.kg-1 soil, and concentration of parameters in mg kg-1 or U/g homogenized leaf for antioxidant enzyme activity.
3.1. Ag in sunflower plants
Sunflower plants exposed to AgNPs / Ag+ in a pot experiment with amended soil had fallen down by time of harvest (Figure 1) because of weak stems, probably caused by a deficiency of calcium (data shown in table 5). Silver from AgNPs or Ag+ accumulated in root hair > root > leaf > stem > flower > (Table 2). The amount of Ag in sunflower leaves from exposure to Ag+ in soil was 3-fold more than that accumulated on exposure to AgNPs and 6-fold more than the control. The seeds planted in soil amended with AgNPs showed a marked decrease in root size with only few roots having few root hairs (~50% less root hair), whereas the grown in soil mixed with AgNO3 showed the smallest root size and ~75% less root hairs were evident compared to the control (Figure 2).

Figure 1. Sunflower plants just before harvest (53 days after sowing). Erect plants are the controls and the others are grown in soil amended with silver as AgNPs or Ag+ (as AgNO3).

Figure 2. Sunflower root growth following exposure to 150 mg/kg of Ag as AgNPs (centre) and Ag+ (as AgNO3; right) for 53 days, compared with the control plants (left).
Table 2. Silver (Ag) concentration (mg.kg-1; mean ± SE) in sunflower plants exposed to 150 mg/kg Ag in soil as AgNPs or Ag+.
|
Plant part |
Control |
AgNPs |
Ag+ |
|
Root |
1.51 ± 0.45 |
17.83 ± 6.69 |
15.61 ± 1.96 |
|
Root hairs |
2.65 ± 2 |
156 ± 9.69 |
182 ± 36.24 |
|
Stem (S1) |
1.95 ± 1.09 |
3.75 ± 3.19 |
2.76 ± 1.16 |
|
Stem (S2) |
1.32 ± 0.63 |
2.99 ± 2.67 |
1.3 ± 0.4 |
|
Stem (S3) |
0.32 ± 0.04 |
0.76 ± 0.24 |
1.29 ± 0.6 |
|
Stem (S4) |
0.26 ± 0.01 |
0.48 ± 0.54 |
1.27 ± 0.1 |
|
|
3.85 ± 1.77 |
7.98 ±6.64 |
6.62 ± 2.26 |
|
Leaf |
1.86 ± 0.24 |
2.75 ± 0.96 |
9.63 ± 4.94 |
|
Flower |
0.31 ± 0.02 |
0.53 ± 0.14 |
1.09 ± 0.5 |
|
Seed base |
0.42 ± 0.09 |
1.04 ± 0.59 |
0.85 ± 0.33 |
|
Seeds |
0.32 ± 0.12 |
0.77 ± 0.21 |
1.04 ± 0.52 |
Exposure of sunflower plants to AgNPs/Ag+ in soil decreased the photosynthesis process resulting in generation of ROS, which damage chloroplast leading to growth retardation or death of the plants. In addition, a reduction in root size and lack of root hairs in the Ag-exposed plants was associated with a decrease in size of nearly all parts of the plant. This was more obvious in plants grown in soil amended with Ag+ (as AgNO3) than AgNPs, which is consistent with the literature reports [32, 33]. Krizkova et al. [29] report a decrease of fresh weight in sunflower plants exposed to Ag+, and that this loss increased with time and exposure dose. These authors surmised that this weight loss was connected with an increase in metabolic activity due to very limited supply of the inorganic and organic compounds needed for plant growth. Krizkova et al. [29] reported that Ag changes water transport, which alters the anatomical structure of the root. The effects of Ag on sunflower roots they report include a decline in rhizodermal cells, which necrotized and were replaced by exodermal cells. Mazumdar [34] reports that when Ag particles enter root cells, they damage both the cell walls and vacuoles, which may contribute to increased entry of Ag into the plant including the root. Root damage could also be due to large Ag particles penetrating through small pores in the cell walls. Metal accumulation in plants is dependent on metal speciation, and Ag accumulates more in the root than other plant parts, followed by leaves and stem [33]. It is assumed that Ag will also accumulate in the seeds and if so, a food safety risk. Other factors that contribute to Ag accumulation in the root are solubility of Ag compounds, plant species and the culture medium [33].
The plants cultivated in soil amended with AgNPs or Ag+ were not strong enough and collapsed (Figure 1) probably due to a deficiency of Ca as calcium pectate [35, 36]. When plants become deficient in Ca, the new tissues that are formed at root and shoot tips exhibit distorted growth due to improper cell formation. This may be the reason that the plants exposed to AgNPs/Ag+ curved at the upper parts of the plant resulting in tilting of the plant. The reduction of Ca concentration in the plants exposed to the two Ag forms (data shown on table 5) could also be attributed to a decrease in the expression of Ca channel proteins, which can lead to a reduction in Ca uptake. [37].
3.2. Antioxidant enzymes, LPO, oxidase, peroxidases
Results are presented as the mean ± SE of four replicates and significance was set at P < 0.05.
Compared to the control plants, leaves of sunflower plants grown in soil containing 150 mg/kg AgNPs or Ag+ (as AgNO3) respectively showed significantly increased catalase enzyme activity (> 8- and 12-fold), SOD activity (> 2- and 3-fold), and GPx enzyme activity (> 2- and 4-fold). GST enzyme activity in sunflower leaves exposed to Ag+ was significantly increased c. 2-fold compared to the control and plants exposed to AgNPs (Table 3).
In sunflower leaves of plants exposed to AgNPs and Ag+ there were significant increases in MDA concentrations (0.3- and 0.7-fold more), ascorbate oxidase activity (2- and 4-fold more), pyrogallol peroxidase (6- and 11-fold more), and guaiacol peroxidase activity (~ 0.5 and 1.3-fold more) respectively compared to the control (Table 3).
Table 3. Analysis of antioxidant enzyme activity and other leaf parameters (U/g wet wt) in sunflower plants exposed to 150 mg/kg Ag as AgNPs or Ag+ (as AgNO3) in amended soil, at harvest 53 days after sowing in pots.
|
|
Control |
AgNPs |
Ag+ |
|
Catalase |
0.24 ± 0.06 a |
4.24 ± 0.46 b |
6 ± 0.1c |
|
SOD |
10 ± 1.5 a |
21.11 ± 4 b |
32 ± 6 c |
|
GST |
259.38 ± 0.29 a |
267 ± 23.08 a |
535 ± 147 b |
|
GPx |
0.00032 ± 0.00005 a |
0.0013 ± 0.0004 b |
0.0022 ± 0.0002 c |
|
MDA conc. |
46 ± 1.63 a |
60 ± 1.24 b |
82 ± 1.12 c |
|
Ascorbate oxidase |
8 ± 0.62 a |
15 ± 0.36 b |
18 ± 0.52 c |
|
Pyrogallol peroxidase |
17 ± 2 a |
191 ± 31 b |
300 ± 56 c |
|
Guaiacol peroxidase |
1.6 ± 0.41 a |
2.39 ± 0.11 a |
3.84 ± 0.26 b |
Means ± SE of four replicates. Means with different letters in a row are significantly different (P < 0.05).
Reactive oxygen species are generated during the normal metabolism of eukaryotic cells [38] and may cause oxidative damage to macromolecules such as lipids, proteins, and DNA [39]. We analysed four antioxidant enzymes (SOD, CAT, GST, GPx) and LPO to evaluate the response to ROS in leaves of sunflowers exposed to 150 mg/kg AgNPs or Ag+ in soil.
In general, metal ions cause oxidative stress in sunflowers and generate ROS toxic species such as free radicals O•−2, OH• and non-radical like H2O2 and 1O2 that can cause degradation of plant proteins in addition to a reduction in number of chloroplasts in leaf tissue. Thus, ROS can affect a range of enzymes and activate the enzymatic antioxidant system, which responds to abiotic and biotic stressors [40, 41]. The antioxidant enzymes studied were CAT (catalyse dismutation of H2O2 to water and oxygen), SOD (frontline defence enzyme against ROS), GST (detoxification enzyme) and GPx (prevents damage from H2O2) [41].
Sunflower leaves exposed to the two Ag forms (AgNPs and Ag+) exhibited elevated CAT, SOD, GST and GPx activation, which is in agreement with [42], which found these enzymes were markedly elevated in sunflower leaf exposed to Ag+ more than AgNPs. Similar findings on increased CAT and GPx activities have been reported on exposure of Bacopa monnieri to 10 and 100 mg kg-1 Ag+, much higher than from exposure to the same concentration of AgNPs [11]. AgNPs sized 20 nm at a concentration of 10 mg L-1 significantly induced oxidative stress with an increase in ROS in a higher aquatic plant Spirodela polyrhiza. In S. polyrhiza exposed to 5 and 10 mg L-1 concentrations of 6-nm and 20-nm AgNPs particle sizes, SOD, CAT and guaiacol peroxidase were significantly increased [43], which also agrees with our results. Jiang et al. [43] reported that exposure of sunflowers to 6-nm AgNPs showed increased activity of these enzymes compared to 20-nm AgNPs particles, showing that the higher activity of the small sized particles was probably due to the relatively larger surface area.
Increased anthocyanins and chloroplast, which play a major role in photosynthesis, were increased in Arabidopsis thaliana exposed to AgNPs [44]. In addition, the plant tissues accumulated H2O2 and also exhibited an increase in nucleic acids and proteins, which could be detected by staining plant tissues with 3,3-diaminobenzidine. The plant leaf under stress exhibited a deep brown colour, compared with the control group. Excessive accumulation of ROS and subsequent LPO is identified by MDA measurement. Li et al. [44] reported that SOD activity in A. thaliana on exposure to AgNPs (0.1, 0.5, 1 mg L-1) significantly declined by 18.2% compared to the control, which is contrary to our findings. In contrast, CAT and POX in the AgNPs-treated group were induced by 2.1- and 1.1-fold respectively compared to the controls, which is in agreement with our findings. The reason for this could be the different plant types and AgNPs particle sizes. (30-nm in my study, and from 9 to 10 nm in [44]). Exposure of tobacco plants to 25-, 50-, 75-, 100- or 500-µM doses of AgNPs and Ag+ in milli Q water in an Erlenmeyer flask resulted in toxicity by Ag+ > AgNPs [45], which is consistent with our observations in sunflowers grown in TSL soil, except for CAT activity which declined on exposure to AgNPs but increased on exposure to Ag+.
Tomato plants exposed to 0, 25, 50, 75, and 100 mg L-1 of 20-nm AgNPs resulted in a linear dose-response increase in all amino acids and also the activity of antioxidant enzymes CAT, SOD, POX in shoot and root at 50 mg/L Ag but total soluble protein decreased [46], all of which agrees with our results. However, at 100 mg L-1 AgNPs, SOD activity declined probably due to unspecified enzyme degradation [47] or due to binding of Ag to the active centre of the SOD enzyme [48], the activity of which was higher in the shoot than roots. The SOD and CAT activity increased in castor oil seedlings exposed to AgNPs/Ag+ [49], which is in agreement with our data. Heavy metals such as Cu and Zn also induced CAT, SOD, POX in castor beans Ricinus communis [49].
The effect of AgNPs on sunflower leaf GST activity was minimal but Ag+ increased GST compared to the control. GST is a detoxification enzyme involved in the metabolism of many chemicals including pesticides and heavy metals. It catalyses the conjugation of electrophilic molecules with reduced glutathione (GSH) to more water soluble and less toxic metabolites [50]. The GSH conjugation is the first step of the mercapturic acid pathway, which is an important detoxification process [51]. GST is induced and able to detoxify chlorophenol in plants grown in polluted soil [52] including on exposure to high concentrations of Cd [53].
A significant increase in GPx was observed in sunflower leaf in plants exposed to AgNPs and Ag+ compared to the controls. The GPx activity in plants depends on physiological and genetic status, time interval, type and concentration of exposure to pollutants including heavy metals and is a response to oxidative stress [54]. GPx catalyses the reduction of H2O2 via GSH to protect plant cells from oxidative stress [38] not only caused by heavy metals but also by salinity and drought conditions [55].
Nickle (Ni) is a heavy metal that cause oxidative stress in Amaranthus paniculatus with a resultant increase in LPO, GPx, SOD and CAT [56]. However, the authors reported that antioxidant enzymes including GPx in roots was progressively inhibited by increased Ni concentration in the growth solution but that a marked increase in GPx and SOD activity occurred in leaves. There are several publications [12, 57-60] on oxidative stress in plants with resultant increase in ROS, LPO and GPx. The exposure of sunflowers to AgNPs and Ag+ resulted in an increase not only in GPx but also other POX such as pyrogallol peroxidase and guaiacol peroxidase.
Exposure of plants to external stress induces LPO. Sunflower plants exposed to AgNPs and Ag+ resulted in a significant increase in MDA in the leaf with the effects due to Ag+ more than AgNPs. In A. thaliana exposed to AgNPs and Ag+, tissues accumulated ROS and resulted in LPO in the leaf, which was quantified by MDA [44]. A similar effect was observed in tomato plants, with a concentration-dependent increase in MDA on exposure to Ag [61]. Exposure of the aquatic plant Spirodela polyrhiza to 5 mg L-1 AgNPs in 10% Hoagland solution resulted in a significant increase in LPO compared to the control [62]. MDA was also significantly increased in leaves of the tobacco plant exposed to 500 µM AgNPs and to 100 µM Ag+ [45]. Thus, the changes in MDA observed in the leaves when the tobacco plant is exposed to AgNPs/Ag+ are similar to the ones observed in our study with sunflower leaves. The main effects of heavy metal ions on plants include an increase in LPO and lipoxygenase activity, and an associated decrease in chlorophyll and GSH [7]. Ascorbate oxidase is also an antioxidant defence enzyme, which can resist ROS. It plays a major role in detoxifying H2O2 in plant cells. The ascorbate oxidase catalyses the conversion of H2O2 to water by acting as a specific electron donor [63]. An increase in the POX enzyme on exposure to AgNPs and Ag+ has been reported [64]. The POX used in our study were pyrogallol peroxidase and guaiacol peroxidase
Exposure of Bacopa monnieri to AgNPs produced more pyrogallol peroxidase than in plants exposed to Ag+ [11] along with an increase in the TPC and a decrease in protein and carbohydrate. In our study, the increase in pyrogallol peroxidase on exposure of the sunflower plant to Ag+ was significantly higher than by AgNPs, which is similar to the finding in the tobacco plants exposed to 50, 75, 100 and 500 µM [45] but different to that reported by [11] in B. monnieri. Guaiacol peroxidase activity on exposure of sunflower to the two Ag forms showed significant increase in the plants exposed to Ag+ only and not AgNPs compared to the control, which is in agreement with the observations of [61] who exposed the tomato plant to AgNPs (6 nm particle size) and Ag+ and observed a significant increase in guaiacol peroxidase activity and also the findings in the aquatic plant Spirodela polyrhiza exposed to AgNPs [62].
3.3. Total phenolic compounds and Total soluble carbohydrates
The TPC concentration was significantly increased in the leaves of sunflower plants exposed to Ag+ > AgNPs > control. A similar observation has been recorded on exposure of B. monnieri to heavy metals [11] and also that phenolic derivatives can act as a metal stress chelator, as an antioxidant to reduce ROS and LPO. These authors reported a reduction in protein and carbohydrate concentrations also [11].
Total phenolic compounds increased by 0.3- and 1-fold, whereas total soluble carbohydrates decreased by 20% and 35%, respectively compared to the control.
Total soluble carbohydrate concentration in sunflower leaves significantly declined following exposure of plants to 150 mg kg-1 of AgNPs and Ag+ in soil for 53 days and this was most marked in the plants exposed to Ag+. Similar results have been reported in B. monnieri exposed to 10, 100 ug/kg of AgNPs and 10, 100 mg kg-1 of Ag+ with a slow decline during the first 10 days of cultivation and a sharp decrease in TSC thereafter [11], which agreed with our findings. Similarly, Salama [10] reported that the exposure of corn (Zea mays) and common bean (Phaseolus vulgaris) to doses up 60 mg kg-1 of AgNPs caused an increase in TSC, but at higher AgNPs concentrations, such as 80 mg kg-1, the TSC concentration decreased significantly. Application of colloidal AgNPs (1 mg/ml) as a foliar spray dose of 50 ml/L increased TSC and also improved the appearance of sunflower leaves [65]
3.4. Vitamins A, E and C
Vitamin A concentration in the leaves of sunflower plants exposed to AgNPs and Ag+ were increased by 1- and 0.3-fold, and vitamin E increased by 6.8- and 2.8-fold, respectively compared to the control (Figure 4). Vitamin C concentrations in leaves exposed to AgNPs and Ag+ increased by 25% (P < 0.05) and 21% respectively compared to the control.
Exposure of sunflower plant to 150 mg kg-1 AgNPs or Ag+ resulted in a significant increase in vitamin A and E concentrations, with AgNPs > Ag+ > control. The exposure of sunflower plants to AgNPs and Ag+ stimulates the production of ascorbate oxidase due to ROS production, which results in the production of L-ascorbic acid. Vitamins A and E (alpha-tocopherol) act as a defence in addition to ascorbate oxidase, which oxidises L-ascorbic acid to dehydroascorbic acid, and therefore the amount of dehydroascorbic acid is increased, and the total ascorbic acid is decreased [66]. A significant increase in the antioxidant vitamins A and E in the bean plant (Phaseolus vulgarus) occurred following exposure to heavy metals Pb (1.5, 2, 2.5 mM), Cu (0.1, 0.2, 0.3 mM), Cd (0.05, 0.06 and 0.08 mM) and Hg (0.02, 0.04 and 0.06) for 10 days [67], which is in agreement with our findings.
3.5. Chlorophyll and total carotenoids
Sunflower leaf chlorophyll A in plants grown in soil containing 150 mg kg-1 Ag as AgNPs and Ag+ (as AgNO3) decreased by 30% and 40% respectively. Chlorophyll B was not significantly affected. Total carotenoid concentration in the sunflower leaves exposed to Ag+ decreased by ~ 50% compared to the control (Figure 3).
Leaf pigments include chlorophyll A and B and total carotenoids, which are essential for photosynthesis and hence plant growth [68]. Silver nanoparticles are taken up by plants into intracellular spaces and transported inside the plant through the plasmodesmata of root cells, then pass through shoots and accumulate in leaves causing inhibition of chlorophyll. The ratio between chlorophyll A and chlorophyll B indicates the response to light and shade conditions and is used as a sensitive biomarker of pollution and environmental stress [69]. Carotenoids are located in chloroplast and with chlorophyll are intricately involved in the photosynthetic process. In addition, they can protect chlorophyll and the thylakoid membrane (a sheet like membrane-bound structure in chloroplast that plays a role in light-dependent photosynthesis) from peroxidative damage [25]. Carotenoids dissolve in non-polar and polar solvents such as diethyl ether and methanol. Hence, we selected methanol for extraction of chlorophyll and total carotenoids in a one-step extraction with good recovery; besides methanol is easier to handle. Mazumdar [34] reported that using 1000 µg/ ml AgNPs in Hoagland’s nutrient solution caused a significant inhibition in both chlorophyll types and total carotenoids in Vigna radiata and Bactris campestris, which is in agreement with our findings There are several reports that AgNPs inhibit plant growth by inhibition of chlorophyll formation and total soluble protein [70, 71] . However, Qian [12] showed that AgNPs inhibited the growth of A. thaliana by disruption of the thylakoid membrane structure and thereby decreased the chlorophyll concentration. In our study, we did not investigate the impact of AgNPs on thylakoid membrane structure, but we agree that chlorophyll concentration is reduced on exposure to AgNPs. In contrast, Qian et al. [12] suggested that Ag+ as AgNPs did not profoundly affect chloroplast structure but could alter the transcription of antioxidants and aquaporin genes, indicating that AgNPs can change the balance between oxidant and antioxidant activity thereby affecting the homoeostasis of the A. thaliana plant. The decline in chlorophyll content in plants exposed to heavy metals has been reported in several studies, including a decrease in the ratio of chlorophyll A to chlorophyll B on exposure to Cu and Hg stress with chlorophyll B responding more to Cu, while a decrease in this ratio was observed in plants exposed to other heavy metals such as Cd and Pb [54].
Carotenoids are a partner pigment in photosynthesis. Carotenoids also act as an antioxidant non-enzymatic pigment that defends chlorophyll and cell membranes against ROS produced on exposure to different heavy metals and environmental stress [72-74]. A role of this pigment is to reduce chlorophyll concentration to protect against chloroplast damage [74]. Decline in carotenoid content indicates heavy metal toxicity, while its enhancement reflects detoxification of ROS species [75, 76]. Our results that chlorophyll and total carotenoids are inhibited in sunflower leaves on exposure of the sunflower plant to AgNPs and Ag+ in soil are confirmed by [7].
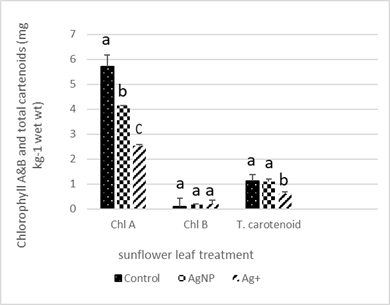
Figure 3. Chlorophyll A and B, and total carotenoid concentrations, in leaves of sunflower plants grown in soil containing 150 mg/kg Ag as AgNPs and Ag+ (as AgNO3). Results are presented as mean ± SE of four replicates. Means with different letters on the bars are significantly different (P < 0.05).
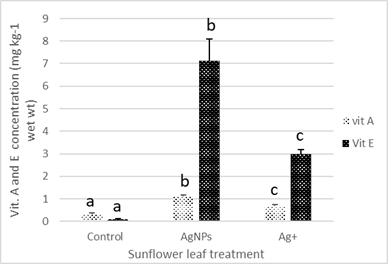
Figure 4. Sunflower leaf vitamin A and E in plants grown in soil containing 150 mg Ag/kg as AgNPs and Ag+ (as AgNO3). Results are presented as mean ± SE of four replicates. Means with different letters on the bars are significantly different (P < 0.05).
3.6. Urease concentration
Urease levels increased significantly with the Ag+ treatment (1-fold more) but were not significantly different from the control with AgNPs treatment (~0.4 fold more) (Table 4).
Urease is a crucial plant enzyme that catalyses the hydrolysis of urea to form ammonia and carbon dioxide [77]. In our study, there was a significant increase in urease activity in sunflower leaves with the increase being most marked in plants exposed to Ag+ more than AgNPs more than control.
Table 4. Analysis of other leaf parameters in sunflower plants exposed to 150 mg/kg Ag as AgNPs or Ag+ (as AgNO3) in amended soil, at harvest 53 days after sowing in pots.
|
|
Control |
AgNPs |
Ag+ |
|
Total protein (mg/ kg wet wt). |
20.18 ± 0.77 a |
16.74 ± 0.95 b |
14.14 ± 0.97 c |
|
Total phenolic compounds (mg equivalent to gallic acid wet wt). |
0.151± 0.01 a |
0.203 ± 0.002 b |
0.294 ± 0.02 c |
|
Total soluble carbohydrates (g/ kg wet wt). |
168 ± 9.65 a |
142 ± 8.8 b |
109 ± 12.6 |
|
Urease (mmol/ g wet wt) |
3.03 ± 0.3 a |
4.27 ± 0.79 b |
6 ± 1.21 c |
|
Vitamin C (mg/kg wet wt) |
117.16 ± 9 a |
166.46 ± 16.8 b |
147. 91 ± 2.77 c |
Means ± SE of four replicates. Means with different letters in a row are significantly different (P < 0.05).
3.7. Total protein
The total protein concentrations in sunflower leaves exposed to AgNPs and Ag+ (as AgNO3) were ~ 12.5% and 25% lower respectively compared to the control. Proteins are large biomolecules consisting of a series of amino acids. In plants, protein has five main functions: plant growth, role in hormones and enzymes, immune function, and in energy production. Exposure of sunflower plants to AgNPs and Ag+ resulted in a reduction of protein in sunflower leaf with Ag+> AgNPs> control. Exposure of sunflower plants to 0 (control), 0.1, 0.5- and 1-mM Ag+ cultivated in an environmental test chamber (MLR-350 H, Sanyo, Japan) showed that protein content in the shoot > root indicative of transport of proteins from roots to shoots [29]. A reduced protein concentration in both the shoot more than root exposed to heavy metals has been reported [41]. A similar decline of up to 70% protein has been observed in the tomato plant leaf exposed to 50 µM Cd in soil compared to the controls and this was accompanied by an accumulation of ammonium ions [61]. In the aquatic plant Spirodela polyrhiza, the effect of 6-nm AgNPs of 5 mg/L increased total soluble protein content [43]. The increase in total soluble protein was much higher when the plant was exposed to 10 mg L-1 20-nm sized AgNPs [43]. This is in contrast to our finding of a decline in sunflower leaf protein in plants exposed to 150 mg/kg Ag and this was most marked in the plants exposed to Ag+ more than AgNPs. This could be due to the smaller particles size and the lower Ag dose used by [43]. Heavy metals such as Pb, Cd, Mn, Co, Cr, and Co also cause significant inhibition of total soluble protein and plant growth in Zea mays [78], which also agreed with our results. On exposure of Bacopa monnieri to AgNPs, the leaf protein concentration declined over time, which agreed with our study [11]. These authors also added that the decrease in protein concentration in B. monnieri was accompanied by an increase in TPC on exposure to heavy metals. The reduction in total amount of soluble protein when exposed to heavy metals could be due to an increase in protease activity [79] or because of the diverse structural alteration and thereby denaturing the protein [80].
Table 5: Concentration of Ca in sunflower exposed to 150mg.kg-1 AgNPs/Ag+
|
Element |
Ca |
|
Part 1: Root |
|
|
Control |
5129 ± 422 a |
|
AgNPs |
4077 ± 273 ab |
|
Ag+ |
3135± 110 b |
|
Part 2: Stem (S-1) |
|
|
Control |
6974 ± 568 a |
|
AgNPs |
5789 ± 174 a |
|
Ag+ |
5555 ± 301 a |
|
Part 3: Stem (S-2) |
|
|
Control |
10684 ± 177 a |
|
AgNPs |
57761 ± 291 b |
|
Ag+ |
7502 ± 188 c |
|
Part 3: Stem (S-3) |
|
|
Control |
9369 ± 716 a |
|
AgNPs |
7777 ± 374 ab |
|
Ag+ |
6041 ± 365 b |
|
Part 3: Stem (S-4) |
|
|
Control |
10860 ± 117 a |
|
AgNPs |
9953 ± 722 b |
|
Ag+ |
7260 ± 330 c |
|
Part 4: Leaves |
|
|
Control |
40939 ± 830 a |
|
AgNPs |
28815 ± 2001 b |
|
Ag+ |
27044 ± 1692 b |
|
Part 5: Flower |
|
|
Control |
11080 ± 830 a |
|
AgNPs |
8903 ± 117 b |
|
Ag+ |
4815 ± 262 c |
|
Part 6: Seed |
|
|
Control |
15926 ± 1003 a |
|
AgNPs |
10274 ± 969 ab |
|
Ag+ |
53041 ± 1256 b |
Exposure of sunflower plants to AgNPs and Ag+ (as AgNO3) caused oxidative stress similar to that caused by other heavy metals such as Cd, Pb, Hg, and Ni. The stress of exposure to such heavy metals can lead to ROS, LPO and elevated defence enzymes (CAT, SOD, GST, and GPx) activities in addition to increased activity of POX (pyrogallol peroxidase and guaiacol peroxidase), and antioxidant vitamins (Vitamin A, E, and C) and TPC. The increase in TPC was accompanied by a decrease in total soluble protein. Silver accumulated in the sunflower plant with the concentration in the root more than leaf more than stem. Silver nanoparticles can alter the transcription of antioxidants and oxidants, causing an imbalance in homoeostasis of the plant. The major impacts of AgNPs and Ag+ on the sunflower plant are the reduction of total protein, inhibition of chlorophyll synthesis through damage to chloroplast and TSC in the leaf, reduction in root size, loss of root hair and inhibition of stem length, all of which together can lead to a reduction in growth and yield of the crops. The ratio of chlorophyll A to chlorophyll B could be used as a sensitive biomarker of exposure to pollution and environmental stress. Overall, the responses of the plant parameters measured in this study were most marked in the sunflower plants exposed to Ag+ > AgNPs.
The security risk of sunflower seeds after exposure to AgNPs and Ag+ increased and authorities must control the discharge of the waste of the factory used AgNPs in their products and the waste of municipality which contains AgNPs before use this waste as fertilizer.
Supplementary data 7.1
Calculation of antioxidant enzymes activity in earthworm
(µM min–1 mg protein–1) and sunflower (U/g)
W = Weight of earthworm (g)
P = Total amount of protein in the earthworm extract
U = Unit of enzyme activity
Vs = Total volume of the earthworm extract, in litres
Ve = Volume of earthworm extract used for enzyme measurement, in litres
Vt = Total mixture of the reaction mixture
∆A/min = Absorbance variation per minute calculated on the linear part of the curve
A = f(time) when measuring enzyme activity
ɛ = Extinction coefficient at specific wavelength for the enzyme (in M/cm) of the compound released by the reaction, that allows calculation of the amount of substrate hydrolysed by the enzyme according to the Beer-Lambert Law:
A = ɛ. 1. C. Where A is the absorbance at specific wavelength, 1 is the path length (cm), and C is the concentration of the product formed which corresponds to the concentration of substrate hydrolysed, in M/l.
Calculation in cuvette using spectrophotometry:
The path length 1 = 1 cm; (∆A/min) / ɛ= C (Mol L–1 min–1); C x Vt x 106 = µMol/min in the reaction mixture
(C x Vt x 106) x (Vs/Ve) = total amount of µMol/min in the earthworm extract corresponding to the total amount of units (U)
[(C x Vt x 106) x (Vs/Ve)]/ P= Specific activity in U/mg.
Supplementary data 2:
Calculation of chlorophyll A & B and total carotenoid in sunflower exposed to 150 mg.kg-1 AgNPs/Ag+
For chlorophyll A: Absorbance at 665.2 nm × 16.72 – (Absorbance at 652.4 nm × 9.16)
For chlorophyll B: Absorbance at 652.4 nm × 34.09 – (Absorbance at 665.2 nm × 15.28)
For total carotenoids: Absorbance at 470 nm × 1000 – [(1.63 × CA) – (104.96 × CB) / 221.
This work was supported by Lincoln University and Landcare Research, Lincoln, New Zealand. The technical assistance of Mr. Martin Webley is gratefully acknowledged.
Dakal, T.C., et al., Mechanistic basis of antimicrobial actions of silver nanoparticles. Frontiers in Microbiology. 2016.7:1831.
Graf, C., et al., A general method to coat colloidal padticles with silica. Langmuir, 2003;19(17):6693–7003.
View ArticleGatto, M A., Nassem, S., Arafat, M. Y., Mahmoud, D. A., Qasim, k., Zubai, S. Physiochemical properties of nanomaterials implications in associated toxic manifestation. BioMed Research, 2014;2: 1 – 8.
Anjum, N.A., et al., Silver nanoparticles in soil–plant systems. Journal of Nanoparticle Research. 2013.15(9):1896.
Sairam, R., D. Singh, and G. Srivastava, Changes in activities of antioxidant enzymes in sunflower leaves of different ages. Biologia Plantarum. 2003.47(1):61.
Stoikou, V., et al., Μetal uptake by sunflower (Helianthus annuus) irrigated with water polluted with chromium and nickel. Foods. 2017;6(7):51. PMid:28714908
View Article PubMed/NCBIGallego, S.M., M.P. Benavídes, and M.L. Tomaro, Effect of heavy metal ion excess on sunflower leaves: evidence for involvement of oxidative stress. Plant Science, 1996. 121(2):151–9. 04528-1
View ArticleLeela, A. and M. Vivekanandan, Tapping the unexploited plant resources for the synthesis of silver nanoparticles. African Journal of Biotechnology, 2008.7(17).
Sewalem, N., S. Elfeky, and F. El-Shintinawy, Phytoremediation of lead and cadmium contaminated soils using sunflower plants. Journal of Stress Physiology & Biochemistry, 2014.10(1):123–34.
Salama, H.M.H., Effects of silver nanoparticles in some crop plants, common bean (Phaseolus vulgaris L.) and corn (Zea mays L.). International Research Journal of Biotechnology, 2012.3(10):190–7.
Krishnaraj, C., et al., Effect of biologically synthesized silver nanoparticles on Bacopa monnieri (Linn.) Wettst. plant growth metabolism. Process Biochemistry, 2012.47(4): 651–8.
Qian, H., et al., Comparison of the toxicity of silver nanoparticles and silver ions on the growth of terrestrial plant model Arabidopsis thaliana. Journal of Environmental Sciences, 2013.25(9):1947–56.
Cabrera-de la Fuente, M., et al., Effect of application of silver nitrate on antioxidant status in watermelon plants. Pakistanian Journal of Botany, 2014.46(5):1843–6.
Carey, L.M., On allotropic forms of silver. American Journal of Science, 1889.37:476–91.
Simmler, M., et al., Lignite reduces the solubility and plant uptake of cadmium in pasturelands. Environmental Science & Technology, 2013.47(9):4497–504.
Al Mamun, S., et al., Municipal composts reduce the transfer of Cd from soil to vegetables. Environmental Pollution, 2016.213:8–15.
Havir, E.A. and N.A. McHale, Biochemical and developmental charcterization of multiple forms of catalase in tobacco leaves. Plant Physiology and Biochemistry, 1987. 84:450–5.
van Rossum, M., M. Alberda, and L.H.W. van der Plas, Role of oxidative damage in tulip bulb scale micropropagation. Plant Science, 1997.130(2):207–16.
Habig, W.H., M. Pabst, and J. WE, Glutathione-S-Transferase: The first enzymatic step in mercapturic acid formation. Journal of Biological Chemistry, 1974.249(22):7130–9.
Rotruck, J.A., et al., Selenium biochemical role in citrus mitochondria to arsenate. Nature, 1973.206:319–20.
Buege, J.A. and S.D. Aust, Microsomal lipid peroxidation. Methods in Enzymology, 1978.52:302–5.
Vines, H.M. and M.F. Oberbacher, Response of oxidation and phosphorylation in citrus mitochondria to arsenate. Nature, 1965.206:319–20.
Addy, S.K. and R.N. Goodman, Polyphenol oxidase and peroxidase in apple leaves inoculated with a avirulent strain for Erviniaamylovora. Indian Phytopathology, 1972. 25:575–9.
Urbanek, H., E. Kuzniak-Gebarowska, and H. Herka, Elicitation of defense responses in bean leaves by Botrytis cinerea polygalacturonase. Acta Physiologia Plantarum, 1991.43:43–50.
Sumanta, N., et al., Spectrophotometric analysis of chlorophylls and carotenoids from commonly grown fern species by using various extracting solvents. Research Journal of Chemical Sciences, 2014.4(9):63–9.
Dubios, M., et al., Colorimetric method for determination of sugars and related substances. Analytical Chemistry, 1956.28:350–6.
Ainsworth, E.A. and K.M. Gilles, Estimation of total phenolic content and other oxidation substrates in plant tissues using Folin-Ciocalteu. Nature Protocol, 2007.2:875–7.
Bradford, M.M., A rapid and sensitive method for quantitation of microgram quantities of protein utilizing the principal of protein-dye binding. Analytical Biochemistry, 1976. 72:248–54. 90527-3
View ArticleKrizkova, S., et al., Multi-instrumental analysis of tissues of sunflower plants treated with silver (I) ions- plants as bioindicators of environmental pollution. Sensors (Basel), 2008.8:1445–463.
Chotyakul, N., et al., Simultaneous HPLC quatification of vitamins A and E content in raw, pasteurized and UHT cow's milk and their changes during storage. European Food Research and Technology, 2014.238(4):535–47.
Jagota, K.S. and H.M. Dani, A new colorimetric technique for the estimation of vitamin c using Folin phenol reagent. Analytical Biochemistry, 1982.127:178–82.
Vishwakarma, K., et al., Differential phytotoxic impact of plant mediated silver nanoparticles (AgNPs) and silver nitrate (AgNO3) on Brassica sp. Frontiers in Plant Science, 2017.8:1501.
Yang, J., et al., Alteration of crop yield and quality of wheat upon exposure to silver nanoparticles in a life cycle study. Journal of Agricultural and Food Chemistry, 2018.66(11):2589–97.
Mazumdar, H., The impact of silver nano particles on plant biomass and chlorophyll content. International Journal of Engineering and Science, 2014.4(7):12–20.
Boyer, J.S., Enzyme-less growth in Chara and terrestrial plants. Frontiers in Plant Science, 2016.7:866.
Buechel, T. Role of calcium in plant culture. Promix 2018.
Maathuis, F.J.M., Physiological functions of mineral macronutrients. Plant Biology, 2009.12:250–8.
Zhang,L., Wu, M,Teng, Y, Jia, S, Yu, D., Wei, T., Chen, C, and Song, W. Overexpression of the Glutathione Peroxidase 5 (RcGPX5) Gene From Rhodiola crenulata Increases Drought Tolerance in Salvia miltiorrhiza, Front Plant Sci. 2018; 9:1950. PMid:30687353 PMCid:PMC6333746
View Article PubMed/NCBIGill, S.S. and N. Tuteja, Reactive oxygen species and antioxidant machinery in abiotic stress tolerance in crop plants. Plant Physiology and Biochemistry, 2010.48(12):909–30.
Karuppanapandian, T., et al., Reactive oxygen species in plants: their generation, signal transduction, and scavenging mechanisms. Australian Journal of Crop Science, 2011.5(6):709.
Das, K. and A. Roychoudhury, Reactive oxygen species (ROS) and response of antioxidants as ROS-scavengers during environmental stress in plants. Frontiers in Environmental Science, 2014.2:53.
Gracia, J.S., et al., Metal contamination effects on sunflower (Helianthus annuus L.) growth and protein expression in leaves during development. Journal of Agricultural and Food Chemistry, 2006.54(22):8623–8630.
Jiang, H.S., et al., Physiological analysis of silver nanoparticles and AgNO3 toxicity to Spirodela polyrhiza. Environmental Toxicology and Chemistry, 2012.31(8):1880–6.
Li, X., et al., The interctive effects of diclofop-methyl and silver nanoparticles on Arabidopsis thaliana: Growth, photosynthesis and antioxidant system. Environmental Pollution, 2018.232:212–9.
Cvjetko, P. and M. Zovko. Phytotoxic effects of silver nanoparticles in tobacco plants. Environmental Science and Pollution Research, 2017. PMid:29222658
PubMed/NCBIMehrian, S.K., R. Heidare, and F. Rahmani, Effect of silver nanoparticles on free amino acids content and antioxidant defense system of tomato plants. Indian Journal of Plant Physiology, 2015.20(3):257–63.
Filek, M., et al., The protective role of selenium in rape seedlings subjected to cadmium stress. Plant Physiology 2008.165:833–44.
Stroinski, A. and M. Kozlowska, Cadmium induced oxidative stress in potato tuber. Acta Societatis Botanicorum Poloniae, 1997.66:189–95.
Yasur, J. and P.U. Rani, Environmental effects of nanosilver impact on castor seed germination, seedling growth, and plant physiology. Environmental Science and Pollution Research, 2013.20:8636–48.
Vanhaelen, N., et al., Hoverfly glutathione S-transferaseand effect of Brassicaceae secondary metabolite. Pesticide Biochemistry and Physiology, 2001.71(3):170–7.
Ramesy, E.E., Dilda, Pierie. Glutathion S- transferase comjugates as prodrug to target drug resistance tumer. Frontier in Pharmacology, 2014, 5: 181.
Schroder, P., L. Lyubenova, and C. Huber, Do heavy metals and metalloids influence the detoxification of organic xenobiotics in plants? Environmental Science and Pollution Research, 2009.16:795.
Komives, T., A.A.A. Aioub, and G. Gullner, Effect of chemicals stress caused by heavy metals on the glutathione-S-transferse enzyme activity in maize (Zea mays L.). Acta Phytopathologicaet Entamologica Hungarica, 1995.30(3–4):215–20.
Hassan, Z., et al., Biochemical amd molecular response of oilseed crops to heavy metal stress: Yield and adaptations under Environmental stress. Oilseed Crops. Pakistan: Research Gate 2017. 12:236 – 246.
Rahimizadeh, M., et al., The effect of micronutrients on antioxidant enzymes metabolism in sunflower (Helianthus annuus L.). Helia, 2007.30(47):167–74.
Pietrini, F., et al., Evaluation of nickel tolerance in Amaranthus panicultus L by measuring photosynthesis, oxidative status, antooxidative response and metal-binding molecule content. Environmental Science and Pollution Research, 2015.22(1):482–94.
Bailly, C., et al. Antoxidant system in sunflower seeds as affected by priming. Seed Science Research, 2000.10(2):35–42.
Habibi, D., et al., Antoxidative enzymes in sunflower subjected to drought stress. 4th International Crop Science Congress, Australia, 2004. PMCid:PMC523047
Jiang, Y. and H. H, Drought and heat stress injury to two cool-season turf grasses in relation to antioxidant metabolism and lipid peroxidation. Crop Science, 2001.41:436–42.
Malan, C., M.M. Greyling, and J. Gressel, Correlation between Cu.Zn suoeroxide dismutase and xenobiotic stress tolerance in maize inbreds. Plant Science, 1990.69:157–66.
Karami, S., R. Heidare, and F. Rahmani, Effect of silver nanoparticles on free amino acids content and antioxidant defense system of tomato plants. Indian Journal of Plant Physiology, 2015.20(3):257–63.
Jiang, H.S., et al., Silver nanoparticles induced accumulation of reactive oxygen species and alteration of antioxidant system in the aquatic plant Spirodela polyrhiza. Environmental Toxicology and Chemistry, 2014.33(6):1398–405.
Nicholas Smirnof., The function and metabolism of ascorbic acid in plants. Annals of Botany, 1996. 78:661- 669.
View ArticleCvjetko, P., Zovko, M., Peharec, P., Renata, S., Tkalec, M, Domijan, A M. Phytotoxic effects of silver nanoparticles in tobacco plants, Environmental Science and Pollution Research, 2017. 25(6): 5590- 5602 PMid:29222658
View Article PubMed/NCBIYassen, A.A.M., et al., Effect of foliar spray of nanosilver silver and organic fertilizer (Agastar) and salicyclic acid on some morphological characteristics and carbohydrate content in (Helianthus annuus L.). Journal of Agriculture and Ecology Research, 2016.9(3):1–7.
Shimada, Y. and S. Ko, Ascorbic acid and ascorbate oxidase in vegetables. Chugokugakuen Journal, 2008.7:7–10.
Zengin, F.K. and O. Munzuroglu, Effects of some heavy metal on content of chlorophyll, proline and some antioxidant chemicals in bean (Phaseolus vulgaris L.) seedlings. Acta Biologica Cracoviensia, 2005.47(2):157–64.
Costache, M.A., Campeanu, and G. Neata, Studies concerning the extraction of chlorophyll and total carotenoids from vegetables. Romanian Biotechnology Letters, 2012.17(5):7702–8.
Porra, R.J., Recent advances and re-assessments in chlorophyll extraction and assay procedures for terrestrial, aquatic, and marine organisms, including recalcitrant algae, . Scheer H (ed) Chlorophyll, 1991: p. 31–57.
Haghighi Pak, Z., Karimi, N., Abbaspous,H. Effect of silver nanoparticles exposure on growth, physiology and biochemical parameters of Dracocephalum moldavica. Iranian Journal of Plant Physiology 2017. 7(4): 21732183.
Farghaly, F A., Nafady, N A,Green synthesis of silver nanoparticles using leaf extract of Rasmarinus officinalis and it is effect on tomato and wheat plants. Journal of Agriculture Science (2015), 7(11): 277- 287.
Hou, W., et al., Effect of copper and cadmium on heavy metal polluted waterbody restorationtion by duckweed (Lemma minor). Plant Physiology and Biochemistry, 2007.45:62–69.
Kenneth, E., Pallett, K., Young, A., 2000. Antioxidants in higher plants.
McElroy, J.S. and D.A. Kopsell, Physiological role of carotenoids and other antioxidants in plants and application to turfgrass stress management. New Zealand Journal of Crop and Horticultural Science, 2009.37(4):327–33.
ul Hassan, Z., Ali, S., Rehan, A., Farahat, A. Biochemical and molecular responses of oil seed crops to heavy metal stress. 2017, In Book Oil Seed Crops: 236- 248.
Tewari, R., et al., Modulation of oxidative stress responsive enzymes by excess cobalt. Plant Science, 2002.162:381–8.
Hubalek, J., et al., Spectrometric and voltammetric analysis of urease-nickel. Sensors 2007.7(7):1238–55.
Abdul, G., Toxic effects of heavy metals on plant growth and metal accumulation in maiz (Zea mays L.). Iranian Journal of Toxicology, 2010.4:325–34.
Palma, J., L. Sandalio, and F. Corpas, Plant protease, protein degradation and oxidative stress: role of peroxisomers. Plant Physiology and Biochemistry, 2002.40:521–30.
Tamás, MJ., 1,* Sharma, SK., 2 Ibstedt, S.,1 Jacobson, T., 1 and Christen,2P. Heavy Metals and Metalloids As a Cause for Protein Misfolding and Aggregation. Biomolecules. 2014.; 4(1): 252–267. PMid:24970215
View Article PubMed/NCBI