CORRESPONDENCE AUTHOR: Dr John Monro
Address: The New Zealand Institute for Plant & Food Research Limited, Private Bag 11 600, Palmerston North 4442,New Zealand.
E-mail: john.monro@plantandfood.co.nz
© 2019 Sift Desk Journals. All Rights Reserved
VOLUME: 1 ISSUE: 2
Page No: 60-67
CORRESPONDENCE AUTHOR: Dr John Monro
Address: The New Zealand Institute for Plant & Food Research Limited, Private Bag 11 600, Palmerston North 4442,New Zealand.
E-mail: john.monro@plantandfood.co.nz
Suman Mishra1, John Monro1*, Keith Armstrong2, Lidia Motoi2, Kevin Sutton2
1 The New Zealand Institute for Plant & Food Research Limited, Private Bag 11 600, Palmerston North, New Zealand.
2 The New Zealand Institute for Plant & Food Research Limited, Private Bag 4704, Christchurch New Zealand.
Mustapha Diaf(diafmustapha@gmail.com)
Suman Mishra, Digestive analysis of processing effects on the glycaemic impact of oats in vitro(2016)SDRP Journal of Food Science & Technology 1(2)
Forty six lines of oats from a program of breeding for low glycaemic impact were assessed by in vitro digestive analysis that allowed for theoretical effects of homeostasis on blood glucose concentrations. The oats were steamed and rolled (“rolled oats”; RO), and a portion made into porridge (PO) before in vitro digestive analysis. Carbohydrate digestibility was much greater and more rapid for PO than RO after 20 min digestion (RO, 16.9 g/100 g; PO, 42.5 g/100g on a dry oats basis) but similar after 120 min digestion (43 g versus 49.8 g), reflecting a high proportion of slowly digested starch in RO. Taking into account theoretical blood glucose disposal, RO was equivalent in glycaemic effect to 10.8 g of glucose and the PO to 17.5 g of glucose per 40 g serving (10.8 versus 17.5 g glucose equivalents). Based on published GI values, in vitro glycaemic analysis allowing for homeostasis provided realistic relative estimates of effects of processing on glycaemic properties of cereal foods.
Development of cereal cultivars and products to reduce the effect of diets on blood glucose is becoming an increasingly important driver of food research (Becker, 2012). The task of transferring the attributes of cereal cultivars, such as low rates of starch digestion, through to beneficial outcomes such as reduced glycaemic responses in consumers is, however, multilevel.
As well as the intrinsic carbohydrate digestibility and yield of glucose during digestion of a selected cereal kernel (Englyst, Englyst, Hudson, Cole, & Cummings, 1999), there is a need to consider the effects of further processing on digestion (Bjorck, Granfeldt, Liljeberg, Tovar, & Asp, 1994). In addition, foods in which cereals are one of many ingredients may contain substantial quantities of sugars with lower glycaemic potency than glucose, such as sucrose (60% that of glucose) and fructose (22% that of glucose). Such sugars, nonetheless, contribute to the glycaemic response (Livesey, 2003) so should be counted in the glycaemic impact by converting them to grams of glucose equivalents with respect to glycaemic impact (GGE).
Beyond digestion, accuracy in predicting the glycaemic response is improved by allowing for the dose-dependent, homeostatic, apparent blood glucose disposal (GD) reaction to blood glucose loading (Monro, Mishra, &Venn, 2010), because the postprandial glycaemic response results from a net excess of blood glucose loading over glucose disposal.
In vitro estimation of the glycaemic impact of foods commonly involves measuring the content of rapidly (RDS) and slowly digested (SDS) starch ((Englyst, Englyst, Hudson, Cole, & Cummings, 1999). But while rapidly digestible starch (RDS), rapidly available carbohydrate (RAC), and derivatives such as the relative area under the digestion curve (giving an hydrolysis index (HI) (Goni, Garcia-Alonso, & Saura-Calixto, 1997) do give a prediction of glycaemic effect (Brighenti, Pellegrini, Casiraghi, & Testolin, 1995; Monro & Mishra, 2010), such methods do not allow for the fact that the body disposes of absorbed glucose in a dose-dependent manner. That is, the higher the glucose loading, the greater the rate of glucose disposal (Monro, Mishra, &Venn, 2010) so the smaller the increment in glycaemic response.
Bearing in mind the above factors to consider when linking the digestibility of a cereal cultivar to its possible place in a healthy diet, in vitro digestive analysis of glycaemic impact was conducted using a selection of 28 oat lines. One aim was to determine the relative responses of different oat lines already preselected for low glycaemic impact, to further processing into porridge, so as to identify lines with potential as low glycaemic impact porridge oats. A second aim was to demonstrate the utility of glucose disposal baselines in analysis of glycaemic potency based on in vitro digestion.
2.1. Samples
Twenty-eight oat lines were obtained from the Plant & Food Research cereal breeding programme. They were selections relatively low in rapidly digested starch (RDS) from a larger population characterized with respect to starch digestibility (Mishra, Monro, Armstrong, Motoi & McLachlan, 2009). The oats were firstly “rolled” in a laboratory process that simulated commercial production of rolled oats, involving conditioning (partial hydration), steaming and crushing of the oat kernels, as previously described (Mishra, Monro, Armstrong, Motoi & McLachlan, 2009). The rolled oat (RO) samples were also cooked in water (100 °C, 10 min with stirring) to produce a cooked porridge (PO).
2.2. Digestive analysis in vitro
An in vitro digestive analysis previously shown to predict human relative glycaemic responses to cereal products was used to characterize the oats in terms of the mass of glycaemic carbohydrate to which they subject the body, and the predicted relative blood glucose response to the carbohydrate. The digestion procedure involved a 30 min pepsin digestion, followed by pancreatic digestion during which aliquots were removed and made to 80% in ethanol at specified times, for measurement of released sugars and dextrins as glucose equivalents (GE). GE was measured as reducing sugar, against a glucose reference, in an aliquot of the 80% ethanol-soluble fraction after a secondary digestion with amyloglucosidase and invertase to convert dextrins and sucrose to monosaccharide (Monro, Mishra, Venn, 2010).
The analysis usually involves converting the GE values to glycaemic glucose equivalent (GGE) values by adjusting for the relative glycaemic potency of the sugars contributing to the GE value, but in the present case nearly all of the GE was derived from starch, so GGE was assumed to equal GE. Digestible carbohydrate fractions were measured as GE in the aliquots removed during digestion. These fractions were, by convention, defined as rapidly digested starch (RDS; 0-20 min pancreatic digestion), slowly digested starch (SDS; 20-120 min digestion), and inaccessible digestible carbohydrate (IDC; digested between 120 and 180 min after homogenizing at 120 min and adding 0.1 ml additional amyloglucosidase (Megazyme, E-AMG).
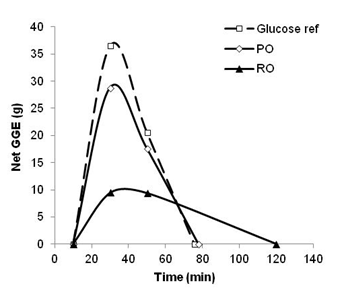
The theoretical relative glycaemic effect of the GGE loading into the digestion medium was determined as glucose equivalents with respect to glycaemic response (GEGR) by diminishing the GGE loading at each time point on the digestion curves by the estimated GGE dose-dependent glucose disposal (GD) response at the time (Monro, Mishra, Venn, 2010), which represents the homeostatic response of the body to blood glucose loading, to provide netGGE values. Simulated blood glucose response curves of netGGE versus time could then be plotted. By comparing the area under the netGGE curves (AUC) with the AUC for glucose references, in vitro relative glycaemic impact (RGI) values for consumed quantities of the oats were determined.
While RGI gives a predictive estimate of relative glycaemic response to a whole food, glycaemic index (GI) is a percentage estimate of the relative glycaemic effect of the available carbohydrate component only. In vitro glycaemic index (GI) values were obtained from the values for RGI per 100 g oats by dividing by the proportion of carbohydrate in the oats (0.52-0.53 g available carbohydrate per g oats).
2.3 Data analysis
Steps in analysis (Fig. 1) of the sugar release profiles for the RO and PO samples allowed determination of the following properties:
(Table 1).
Statistical analyses were conducted in a Microsoft Excel spreadsheet (Microsoft Corp., Redmond, WA, USA).
There was little difference between the 28 oat lines tested and the effects of processing were far greater than any genetic differences in digestibility in the population studied (Table 1). The low inter-line variability observed in of the 28 oat lines screened for glycaemic potential was possibly because they had already been selected from a larger population on the basis of having a low RDS:TS ratio. Also, the parent population had been developed for agronomic and yield characteristics, before the current emphasis on selection for health properties had arisen. Given that the diversion of carbon into structural cell wall polysaccharides is associated with a reduced proportion being allocated by the plant to yields of edible grain, it is possible that the potential for low glycaemic impact had been reduced in the parent population as a by-product of breeding for yield. Incorporation of new genetic material from wild types may be required to reintroduce low glycaemic traits.
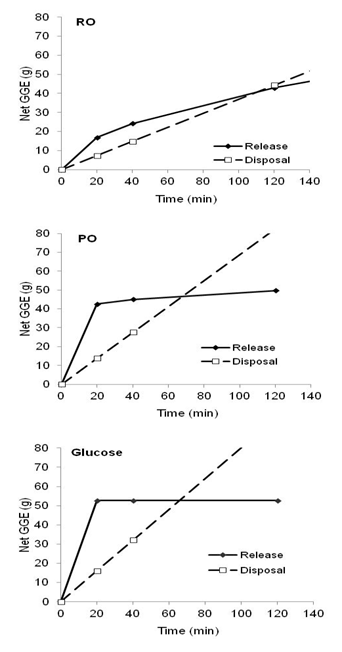
In view of the uniformity of the oat lines the population means (Table 1) were used in further analysis of the results to determine the effects of processing RO into PO. The profiles of digested carbohydrate release by pancreatin were very different for the RO and PO samples (Fig. 2).
The digestion of PO was rapid and approached a plateau by about 20 min while the digestion of RO was more gradual and continuous. Homogenising the sample at 120 min did not greatly add to the sugar already released from the PO sample whereas carbohydrate digested from the RO sample increased a further 20%.
There has recently been a large international effort into development of static and dynamic in vitro assays that will more accurately mimic gastrointestinal conditions than assays such as the one used in the present study (Minekus, Alminger, Alvito, Ballance, Bohn, Bourlieu, et al., 2014). Despite the complexity of processes affecting digestion (Gidley, 2013), the present study has shown that within certain types of food category, such as high starch cereals, in which susceptibility of starch to digestion is a major factor determining available carbohydrate release and glycaemic impact, a simple in vitro assay can be useful. A great advantage over more complicated assays is that it allows a large number of replicates or a large number of samples to be simultaneously analysed, with gains in precision, reproducibility and economy, particularly compared with complicated procedures that allow digestion of only one sample at a time. In the present study 46 samples in both the raw and cooked state were analysed for susceptibility to digestion in a relatively short time.
Recent work on the digestion kinetics of starch has shown that during the 120 min digestion of starch two populations of starch molecule, that differ in their susceptibility to amylase digestion, are hydrolysed(Butterworth, Warren, Grassby, Patel, & Ellis, 2012). It has been suggested, therefore that it is not strictly correct to label starch digested at 20 min as rapidly digestible starch. In the present study, RDS refers not to rapidly digestible, but to rapidly digested starch, and acknowledges that it is derived from the products of both rapidly digestible and slowly digestible starch that have appeared by 20 min digestion. Since glycaemic response depends on the absorption of glucose from the gut, the concentration of starch digestion products is important, but their source at any one time is not important to the current holistic analysis of oats as a whole.
The potentially digestible carbohydrate fractions in the RO and PO, defined by their rate of release during pancreatic digestion, reflected their digestion profiles (Table 1). Of the total available carbohydrate (RDS, SDS), RDS was a much lower proportion in RO than PO (RO, 32%; PO, 81%), and SDS was a much higher proportion in RO than in PO (RO, 49%, PO 14%). The potentially digestible but inaccessible carbohydrate (IDS) made up 19% of the RO carbohydrate as opposed to only 5% remaining in PO after digestion. Total starch estimated by summation of RDS, SDS, and IDC as a percent of sample was almost the same for RO and PO (RO, 48.1 ± 3.0%; PO, 47.5 ± 2.6%).
Expressed on a GGE per 40 g serving basis, GGE release at 20 min from RO was the glycaemic equivalent to 6.8 g glucose, compared with 17 g per serving from PO. GGE release was much less rapid for the RO than the PO samples (Table 2).
Expressing the AUCs of the oats relative to the AUC for glucose allowed the theoretical effect of a serving of oats to be expressed in terms of glucose equivalents with respect to glycaemic response (GEGR). A 40 g serving of uncooked rolled oats was thus estimated to be equivalent to 10.8 g glucose in its effect while 40 g of the rolled oats made into porridge was equivalent to 17.5 g glucose in effect (Table 3).
Expressing the results, not on an equal food weight (eg per serving) basis, but on an approximately 50 g carbohydrate basis, allowed estimates of glycaemic index (GI) to be calculated. Using a standard curve of the glucose dose versus AUC, in vitro GI values were obtained by expressing the AUC for 100 g oats (uncooked 52.9 g carbohydrate: cooked, 52.3 g carbohydrate; mean 52.6 g) as a percentage of the AUC for glucose equal in weight to the carbohydrate (monosaccharide equivalents) of the oats (52.6 g). The GI thus calculated for RO was 51% and that for PO was 83%. These in vitro estimates were close values from a large number of clinical determinations; 55 ± 2 for rolled oats, and 79 ± 3 for instant oat porridge (Brand-Miller & Holt, 2004). Our results confirm the validity of the present in vitro analysis.
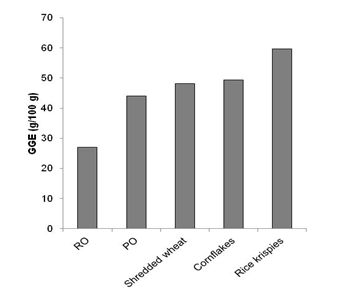
The GI values, being percentages based on the relative response per equal weight of carbohydrate, can be regarded as GEGR per 100 g carbohydrate. To use GI values accurately in choosing foods and in dietary management involving other carbohydrate sources, it is necessary to allow for the carbohydrate content of the food portion consumed, which is less straightforward for consumers than using a GEGR value that directly represents the relative effect of the food portion. In contrast, determination of GEGR per 40 g serving for RO and PO allows the results to be expressed in easily understood terms. Also, expressing the glycaemic potency of RO and PO using GEGR (g) as a virtual food component allows the foods to be directly compared with other carbohydrate foods. The comparison may be made in terms of customarily consumed quantities (GEGR; g/serving) and per 100 g food (Relative GlycaemicPotency; GEGR g/100 g) in the same way as nutrients in foods are shown in nutrition information panels. The relative glycaemic potency (GEGR; g/100 g) of RO and PO compared with other breakfast cereals on a dry basis shows that rolled oats has a relatively low glycaemic impact while porridge was not substantially lower than the other processed breakfast cereals (Fig. 5).
The large increase in digestibility and glycaemic impact of the rolled oats upon making them into porridge undoubtedly reflects the fact that while the process of preparing rolled oats involves heat and crushing, hydration and therefore starch gelatinization is incomplete (Lentle & Janssen, 2011). Furthermore, rolling oats incompletely disrupts kernel structure and gives a product less digestible and less glycaemic than more finely milled oats (Granfeldt, Eliasson, & Bjorck, 2000). Porridge-making appeared to extend the processes of gelatinization and structural disintegration to the point where access to starch allowed the PO digestion curve to reach a plateau at about 20 min, with almost no further digestion after 120 min. In contrast, 20 % of the RO carbohydrate digested between 120 and 180 min upon homogenizing to remove structure, after a more gradual digestion than in the PO samples during the preceding 120 min.
The differences between RO and PO in rate and extent of carbohydrate digestion suggest that oats consumed as rolled oats may be healthier than if consumed as porridge for several reasons:
The postprandial blood glucose spike would be less acute for RO than PO, lowering insulin demand implicated in development of Type2 diabetes, and glycation implicated in damage associated with the complications of diabetes.
The sustained carbohydrate release from RO may lead to more prolonged satiety than provided by PO, which may be helpful in managing obesity.
The greater proportion of carbohydrate measured as IDC in RO compared with PO will contribute to colonic loading of resistant starch, with multiple benefits derived from hind gut fermentation.
The greater loss of potentially digestible carbohydrate to the colon in RO compared with PO would result in a lower metabolizable energy intake per gram of oats, and combined with a reduced insulin response, may assist in weight control.
The relative glycaemic impact of foods for which carbohydrate availability is sustained by slow digestive release can be determined from the area under the curve of net GGE versus time, which is based on the difference between cumulative release of GGE and cumulative theoretical glucose disposal, and appears to give a realistic estimate of relative glycaemic effect.
JM devised the glycaemic analysis, analysed the results and wrote the paper. SM carried out the in vitro digestions. LM prepared the rolled oats. KA provided the oat lines. KS managed the research program in which the study was located.
Funding was partly provided by the New Zealand Foundation for Research, Science and Technology (Contract C02X0401)

|
|
Carbohydrate fraction (g/100 g sample) |
|||||||||
|
|
RDS4 |
SDS |
IDS |
TS |
||||||
|
RO |
|
|
|
|
||||||
|
Mean1 |
15.3 |
23.7 |
9.0 |
48.1 |
||||||
|
SD2 |
3.4 |
2.0 |
2.2 |
3.0 |
||||||
|
SEM |
0.51 |
0.29 |
0.33 |
0.44 |
||||||
|
Range |
10.2-20.5 |
18.5-26.7 |
5.0-15.6 |
41.7-56.4 |
||||||
|
Analytical SD3 |
0.22 |
0.75 |
0.65 |
1.62 |
||||||
|
PO |
|
|
|
|
||||||
|
Mean1 |
38.7 |
6.6 |
2.3 |
47.5 |
||||||
|
SD2 |
2.9 |
1.9 |
0.9 |
2.6 |
||||||
|
SEM |
0.43 |
0.29 |
0.14 |
0.39 |
||||||
|
Range |
33.1-43.2 |
2.5-13.0 |
0.8-5.0 |
42.0-53.2 |
||||||
|
Analytical SD3 |
0.6 |
0.53 |
0.34 |
|
Carbohydrate fraction (g/100 g sample) |
|||||
|
|
RDS4 |
SDS |
IDS |
TS |
||||||
|
RO |
|
|
|
|
||||||
|
Mean1 |
15.3 |
23.7 |
9.0 |
48.1 |
||||||
|
SD2 |
3.4 |
2.0 |
2.2 |
3.0 |
||||||
|
SEM |
0.51 |
0.29 |
0.33 |
0.44 |
||||||
|
Range |
10.2-20.5 |
18.5-26.7 |
5.0-15.6 |
41.7-56.4 |
||||||
|
Analytical SD3 |
0.22 |
0.75 |
0.65 |
1.62 |
||||||
|
PO |
|
|
|
|
||||||
|
Mean1 |
38.7 |
6.6 |
2.3 |
47.5 |
||||||
|
SD2 |
2.9 |
1.9 |
0.9 |
2.6 |
||||||
|
SEM |
0.43 |
0.29 |
0.14 |
0.39 |
||||||
|
Range |
33.1-43.2 |
2.5-13.0 |
0.8-5.0 |
42.0-53.2 |
||||||
|
Analytical SD3 |
0.6 |
0.53 |
0.34 |
1.47 |
||||||
|
Relative glycaemic impact1 |
RO |
PO |
|
|
|
|
|
GGE/100 g food (g) at 20 min |
16.9 |
42.5 |
|
GGE/100 g food (g) at 120 min |
43 |
49.8 |
|
GGE/40 g serving (g) at 20 min |
6.8 |
17 |
|
GGE/40 g serving (g) at 120 min |
17.2 |
19.9 |
|
|
RO |
PO |
Glucose2 |
|
GD rate (g•min-1) |
0.37 |
0.69 |
0.80 |
|
AUC (g•min-1) |
615 |
996 |
1198 |
|
“Available” CHO (g/100 g)1 |
52.9 |
52.3 |
100 |
|
GEGR/100 g food (g) |
27 |
44 |
100 |
|
GEGR/40 g serving (g) |
10.8 |
17.5 |
40 |
|
GI (%; GEGR/100 g carbohydrate)3 |
51.3 |
83.2 |
100 |