Peter Eck
Address: W569 Duff Roblin Building, 190 Dysart Road, University of Manitoba, Winnipeg, MB R3T 2N2 Canada;
Phone: +1- 204 291 2917
E-mail: Peter.Eck@umanitoba.ca
© 2019 Sift Desk Journals. All Rights Reserved
VOLUME: 4 ISSUE: 5
Page No: 760-769
Peter Eck
Address: W569 Duff Roblin Building, 190 Dysart Road, University of Manitoba, Winnipeg, MB R3T 2N2 Canada;
Phone: +1- 204 291 2917
E-mail: Peter.Eck@umanitoba.ca
Yougui Chen1, Usha Thiyam-Hollander1,2, N. A. Michael Eskin1 & Peter Eck1*
1 Department of Human Nutritional Sciences, University of Manitoba, Winnipeg, Manitoba R3T 2N2, Canada
2 Richardson Centre for Functional Foods and Nutraceuticals
Aibing Wang(bingaiwang@hunau.edu.cn)
Lucia Morbidelli(lucia.morbidelli@unisi.it)
Wiem Meddeb(wimed2@yahoo.fr)
Márcio Rodrigues(marciorodrigues@fcsaude.ubi.pt)
Peter Eck , Phenolics from canola crude extracts protect cells from oxidative stress(2019)SDRP Journal of Food Science & Technology 4(5)
Background: By-products of canola oil production are currently discarded or low value commodities. This study investigates if these by-products are a significant source of extractable dietary phenols which could exhibit cellular antioxidants activity.
Methods: Endogenous phenolic compounds obtained from canola oil deodistillates and canola meal using different extraction techniques were identified and examined for their in vitro antioxidant activities in “test tube” and cellular assays.
Results: Sinapine was the predominant phenolic in the canola meal crude extract, while canolol was the only significant phenol in the accelerated solvent extract of canola meal. The deodistillate did not have canolol or sinapine present, but contained high molecular weight phenols of unknown identity. The “test tube” antioxidant assays indicated that canola meal crude extract and sinapic acid both exhibited stronger antioxidant potentials compared to the other extracts. A dose dependent cyto-protection effect was observed under oxidative challenge by H2O2, when cells were incubated with the canola meal accelerated solvent extract, deodistillate extract and sinapic acid.
Conclusions: This study demonstrates that canola by-products can be the sources of health promoting phenols, for possible formulation into value added nutraceuticals.
Key words: canolol, canola meal, canola oil deodistillates, cellular antioxidants
Canola (Brassica napus L.) is the third largest oilseed crop worldwide with the seeds containing about 42-43% oil [1]. The production of canola oil involves crushing the seeds, which separates the crude oil from the meal, followed by oil-refining using neutralization with alkali, bleaching with clay, and deodorizing via steam distillation [2]. Several by-products are fractionated and used in human nutraceuticals, such as canola meal proteins [3]. Due to its origin, the canola seed contains more dietary phenolic compounds than other oilseeds [4], almost all of which remain in the processing by-products [5]. We therefore hypothesize that canola by-products are a significant source of extractable dietary phenols.
The consumption of dietary phenols is associated with the prevention of some of the most detrimental chronic diseases, such as various cancers, inflammatory and cardiovascular diseases [6-9]. Although a variety of mechanisms have been reported for the preventative actions of phenolics, significant antioxidative activity has been attributed to dietary phenols [10, 11]. Specifically, some canola derived phenols have been shown to exhibit significant antioxidant activity in vitro [12-14] and in vivo [15, 16]. We therefore hypothesize that canola phenols extracted from processing by-products exhibit antioxidant activity and could protect cells from oxidative damage.
A novel method has been developed for extracting canola phenols from meal and deodorizer distillates. In addition we show significant antioxidant activity for the crude extracts in chemical assays and that they can protect cells from an oxidative challenge.
Materials
Canola meal and canola oil deodistillates were provided by Viterra Canola Processing Inc. (Winnipeg, MB, Canada). All chemicals were of analytical grade. Methanol, n-hexane, Folin–Ciocalteau phenol reagent, sodium carbonate, iron (III) chloride dihydrate, TPTZ (2,4,6- tripyridy-s-triazine, Sigma), D-(+)-Glucose, sodium chloride, potassium chloride, monopotassium phosphate, disodium hydrogen phosphate, magnesium sulfate heptahydrate, calcium chloride dehydrate, sodium dodecyl sulphate, dimethyformamide, glacial acetic acid, and thiazolyl blue tetrazolium bromide (MTT) were purchased from Sigma Aldrich, Canada. Sinapic acid was purchased from Sigma-Aldrich (St. Louis, MO, USA). Dulbecco's Modified Eagle Medium (DMEM), Phosphate Buffered Saline (PBS) and antibiotic/antimycotic solution were purchased from Thermal Fisher Scientific (Waltham, MA, USA).
Preparation of crude phenolic extracts from canola by-products
Canola meal was subjected to 2 extraction methods: ultrasonic extraction and accelerated solvent extraction. Canola oil deodistillate was extracted using liquid-liquid solvent extraction.
Ultrasonic extraction of canola meal: Canola meal was defatted using a Soxtec 2050 (Foss-Tecator, Foss North America, MN, USA). The defatted sample was extracted following the procedure described by Khattab et al. [5] to investigate residual phenolics. Defatted sample (1 g) was extracted with 9 mL 70% methanol with ultrasound treatment for 1 minute. The ultrasound treated methanolic mixture was centrifuged at 5000 x g for 10 min at refrigerated condition. The supernatant was collected and filtered using Whatman No.1 filter paper. Residue was extracted following the same procedure twice. The filtrates were pooled and made up to 30 mL.
Accelerated solvent extraction of canola meal: Canola meal was heated at 180 ºC in oven for 15 min (pre-treated) prior to Accelerated Solvent Extraction (ASE) (ASE 300, Dionex). Fifty grams of heated substrates thoroughly were mixed with Ottawa sand in a ratio of 1:1 (w/w) using a spatula. Two filter papers were placed at the bottom of each sample cells followed by completely filling it with canola seeds/ meal. Cell caps were hand tightened securely for both sides and were placed in ASE cell holder and extraction was carried out using n-hexane. The ASE extraction was carried under the following conditions: temperature: 190 ºC; static time: 5 minutes; static cycles, 2 cycles; flush volume: 60%; Purge time: 1 min; pressure: 1500 psi; heat time: 5 min. The hexane extract was re-extracted with 70% methanol
Extraction of Phenolics from canola oil deodistillate: The deodistillate sample was extracted with 50% aqueous methanol, as previously described [17]. Two grams of deodistillates were solubilized with 10 mL hexane (1:5 w/v), followed by extraction using 50% methanol (3 mL) three times. For production of the extract, the same procedure of extraction was maintained and 400 g of deodistillates was solubilized in 1600 ml of n-hexane for 15 minutes, followed by extraction with 400 ml of 50% methanol, five times. The pooled methanolic extracts were evaporated to dryness/ minimum volume in a rotary evaporator (at 50 oC until methanol evaporates, and at 70 oC for 30 minutes for the evaporation of water). A minimum quantity (10 mg) of dried DDL extract was dissolved in 5 ml 50% methanol for further quantification.
Quantification and identification of phenolic compounds by RP-HPLC-DAD
Quantification and identification of phenolic compounds in the extracts was established following a RP-HPLC-DAD analysis as described previously [5]. An 80 mm × 4.0 mm i.d., 4 µm, Synergi Fusion-RP column (Phenomenex, Torrance, Canada) was used for sinapic acid derivatives separation (UV detection at 270 nm and 330 nm). Standards of sinapine, sinapic acid, and canolol were used to authenticate the retention time and UV absorption spectra. The contents of all sinapic acid derivatives were expressed as sinapic acid equivalents (SAE) in μg/g of sample, wherein sinapine and sinapic acid was detected at 330nm, canolol was detected at 270 nm.
Preparation of phenolic solutions
All the crude canola phenolic extracts were concentrated using a rotary evaporator at 50 ± 5 ºC to a minimum volume and re-dissolved in 2 mL methanol prior HPLC quantification. After quantification of phenolic compounds in the canola extracts, they were diluted in (i) HEPES PO4 treatment buffer (glucose: 5mM, HEPES: 10 mM, NaCl: 147 mM, KCl: 5 mM, KH2PO4: 1.9 mM, Na2HPO4: 1.1 mM, MgSO4-7H2O: 0.3 mM; MgCl-6H2O: 1 mM, CaCl2-2H2O: 1.5 mM, pH: 7.4) for cell culture experiment or (ii) distilled water for Folin-Ciocalteau and DPPH assays to desired concentrations. Sinapic acid standard was dissolved in dimethyl sulfoxide (DMSO) before further dilution in treatment buffer or distilled water. Final treatment solutions contained <1% solvent, and there was no cytotoxicity to CHO and CaCo-2 cells at those concentrations.
Folin-Ciocalteau assay
Folin–Ciocalteu assay was carried out as described [18], with slight modifications to be performed in 96-well microplates. Aliquots (20 μL) of phenolic solutions at different concentrations were loaded on a 96-well microplate, followed by addition of 100 μL Folin–Ciocalteau phenol reagent and the microplate was mixed well. After 5 min, 7.5% sodium carbonate (80 μL) was added and the microplate was covered and kept at dark for 2 h. The absorbance was measured at 750 nm using a spectrophotometric microplate reader (BioTek, Winooski, USA). The results were quantitatively expressed as the slope coefficient of the lines representing the dependence of A750 on the concentration of the investigated phenolic solutions. The slope was obtained by linear regression analysis in a concentration range up to 2 mM.
Ferric ion reducing antioxidant power (FRAP) assay
The reducing capacity of phenolic solutions was determined by the FRAP assay [19] with modifications to be performed in 96-well microplates. Briefly, the FRAP solution was freshly prepared using 10 mL of 0.3 M acetate buffer (pH 3.6), 1 mL of 20 mM FeCl3 solution and 1 mL of 10 mM TPTZ (2,4,6- tripyridy-s-triazine) solution in 40 mM HCl and warmed at 37 ºC before using. Twenty-five μL of phenolic solutions at different concentrations and 175 μL FRAP solution were added on a 96-well microplate and incubated at room temperature for 10 min. The absorbance was measured at 595 nm with a spectrophotometric microplate reader (BioTek, Winooski, USA). The results were quantitatively expressed as the slope coefficient of the lines (not shown) representing the dependence of A595 on the concentration of the investigated phenolic solutions, where higher values of slope indicated higher FRAP antioxidant activity. The slope was obtained by linear regression analysis in a concentration range up to 1 mM.
2,2-diphenyl-1-picrylhydrazyl (DPPH) scavenging activity assay
The DPPH scavenging activity assay was carried out according to the method proposed by Brand-Williams et al. [20] with modifications to be performed in 96-well microplates. Briefly, 25 μL of phenolic solutions at different concentrations were added on a 96-well microplate. Then 175 μL freshly prepared DPPH ethanolic solution (0.1 mM) was added to the sample and the microplate was covered and incubated at room temperature for 10 min. The absorbance was measured at 516 with a spectrophotometric microplate reader (BioTek, Winooski, USA). The dose that inhibited free radicals by 50% (IC50) was calculated linear correlation from the average data.
Cell culture
Human epithelial colorectal adenocarcinoma (CaCo-2) and Chinese Hamster Ovary (CHO) cell lines were obtained from ATCC (cat no. HTB-37 and CCL-61) and cultured at 37 ºC in a humidified 5% CO2 incubator. The cells were maintained in Dulbecco's Modified Eagle Medium (DMEM) containing 10% Fetal Bovine Serum (FBS) and 1% antibiotic. Cells were grown in 25 T-flasks until they reached more than 80% confluence before splitting or seeding. For experiments, the cells were seeded on a 96-well plate in 100 μL of growth medium and incubated for 24 h at 37 °C.
Cellular protection against H2O2 induced oxidative stress
For the oxidative stress induction, hydrogen peroxide (H2O2) was prepared in the HEPES PO4 treatment buffer. Initially, cells in 96-well plate were treated with a range of concentrations of phenolic solutions and incubated at 37 ºC for 23 h, followed by addition of H2O2 (final concentration: 500 μM) for a further 1 h. The wells were washed with HEPES PO4 treatment buffer and the protection effect of the phenolic solutions on the oxidative induced cells was determined via cell viability using the colorimetric MTT assay. For MTT assay, 100 μL MTT solution (0.5 mg/mL) was added to the buffer washed well and the microplate incubated for 2 h. Further, 100 μL MTT solubilisation solution (16% sodium dodecyl sulphate, 4% dimethyformamide, 2% glacial acetic acid, pH: 4.7) was added to the well to dissolve formazan crystals. The plate was then shaken and absorbance read at 570 nm with a spectrophotometric microplate reader (BioTek, Winooski, USA). The final results were expressed as cell viability in percentage of the sham-treated positive control.
Statistical analyses
The results are expressed as mean ± SD of at least three independent experiments. Differences between different treatment groups and the control counterparts were determined using one-way ANOVA with least significant difference t-test by using SAS 9.4 software (SAS Institute Inc., USA). Statistical significance was accepted at p<0.05. Regression analysis was carried out using Microsoft Excel 2010.
Phenols are distinct in canola by-product extracts
The major identifiable phenols in the different canola processing by-products were sinapine and canolol (structures was shown in Figure 1), which are also major phenols of intact canola seeds or crude canola oils [13, 21]. Sinapine was the predominant component in the canola meal crude extract (CM) (Figure 2 A), while canolol was the sole significant phenol in the accelerated solvent extract from canola meal (Figure 2 B). The extract from the deodistillate did not contain canolol or sinapine, but higher molecular weight phenols of unknown identity (Figure 2 C).
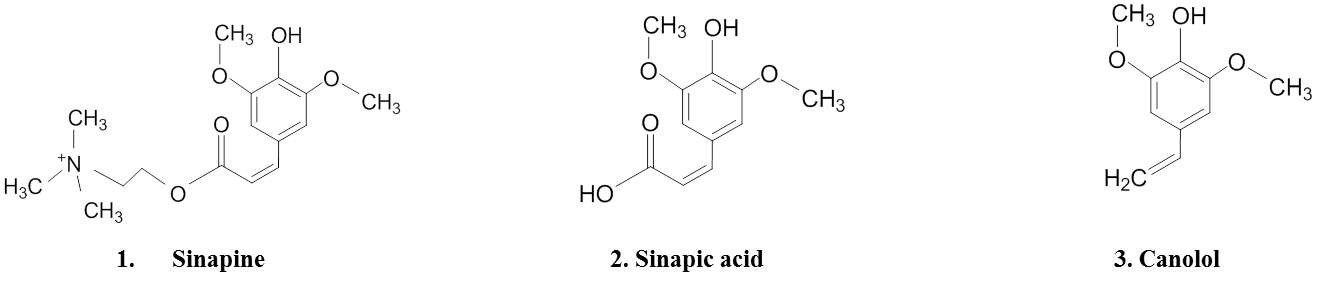
Figure 1. Chemical structures of canola principle phenolics: (1) sinapine, (2) sinapic acid and (3) canolol.
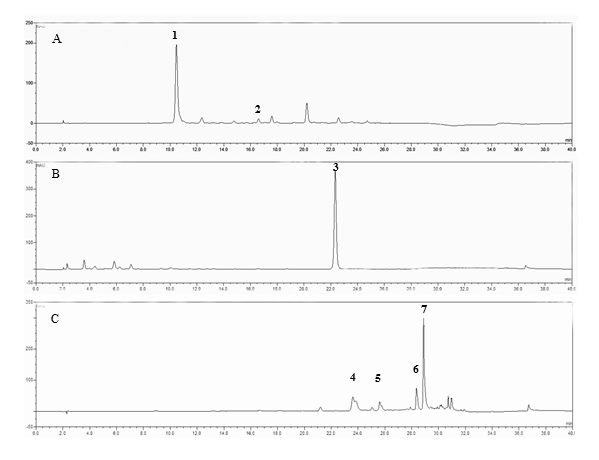
Figure 2. HPLC chromatogram of phenolic extracts of canola processing by-products. (A), Canola meal crude extract (CM) predominantly contains sinapine (peak 1) and some other phenols, such as sinapic acid (peak 2). (B), Canola meal extract obtained from Accelerated Solvent Extraction (ASE) exclusively contains canolol. (C) The deodistillates extract (DDL) contains higher molecular weight phenols of undetermined identity (peaks 4-7).
Phenolics from canola by-products are antioxidants in artificial chemical test systems.
Plants phenolics are increasingly recognized as antioxidants [22]. We therefore assessed the chemical antioxidant capacity using three chemical methods, Folin–Ciocalteu, FRAP, and DPPH assays, which determine the reducing capacity and scavenging activities to ferric ions and free radicals, respectively. The results for Folin–Ciocalteu assay revealed that crude canola meal extract exhibited the highest antioxidant capacity (slope coefficient: 1.12 ± 0.02 μM-1), followed by sinapic acid (0.50 ± 0.06 μM-1), deodistillates extract (0.38 ± 0.02 μM-1) and canola meal accelerated solvent extract (0.26 ± 0.01 μM-1) (Table 1). Similarly, FRAP values of deodistillate extract (slope coefficient: 1.40 ± 0.01 μM-1) and canola meal accelerated solvent extract (1.02 ± 0.04 μM-1) were also significantly lower than that of crude canola meal (2.64 ± 0.07 μM-1) and sinapic acid (2.42 ± 0.07 μM-1). The DPPH activity in this study was expressed as the concentration of extract required to inhibit 50% DPPH radicals (IC50). The results showed a similar patent with the other two assays, crude canola meal extract (IC50: 71.2 ± 0.9 μM) and sinapic acid (IC50: 78.1 ± 1.1 μM) exhibited stronger antioxidant activity than that of deodistillates extract (IC50: 561.6 ± 72.8 μM) and canola meal accelerated solvent extract (IC50: 461.9 ± 5.8 μM).
Table 1. Antioxidant capacity of canola phenolic extracts/solutions
|
|
Crude canola meal extract |
Canola meal accelerated extract |
Deodistillate extract |
Sinapic acid |
|
FC1 (mM-1) |
1.12±0.07a |
0.26±0.01d |
0.38±0.02c |
0.50±0.06b |
|
FRAP2 |
2.64±0.07a |
1.02±0.04d |
1.40±0.01c |
2.42±0.07b |
|
DPPH3 |
71.2±0.9a |
461.9±5.8b |
561.6±72.8c |
78.8±1.1a |
Data represents the mean ± standard deviation of at least three independent experiments. Values in each row share same letters are not significant different (p<0.05).
1 FC, Reducing capacity measured by Folin-Ciocalteu method; values are expressed as the slope coefficients calculated by linear regression (see material and methods), where higher antioxidant activity is expressed by an increase in the slope.
2 FRAP, ferric ion reducing antioxidant power, values are slope coefficients calculated by linear regression (see material and methods), where higher antioxidant activity is expressed by an increase in the slope.
3 DPPH, 2,2-diphenyl-1-picrylhydrazyl free radical scavenging activity, values are the concentration of phenolics needed to inhibit the DPPH● concentration by 50% (IC50).
Phenolics from canola by-products do not exhibit evident toxicity to CHO and CaCo-2 cells
The cytotoxicity of the phenolics from canola by-products was determined using MTT assay with cells were treated with different concentrations of phenolic solutions from 0 to1000 µM. The data indicated that none of the tested phenolic extracts has obvious cytotoxicity against normal CHO and CaCo-2 cells at the concentration below 500 µM (Figure 3). All the extracts showed slight or medium cytotoxicity when the concentrations reach 1000 µM. This indicated the concentrations used in the experiment were safe to the cells.
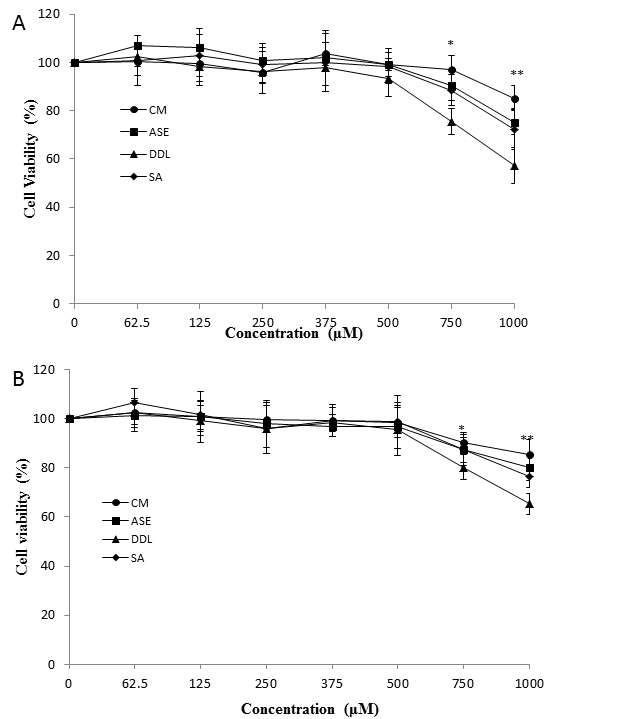
Figure 3. Effect of canola by-products extracts on cell viability under different concentrations on CHO (A) and CaCo-2 (B) cells using 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) assay. Asterisks indicates significant difference with control (* p<0.05, ** p<0.01). CM, Canola meal crude extract; ASE, Canola meal accelerated solvent extract; DDL, Deodistillates extract; SA, Sinapic acid.
Phenolics from canola by-products protect cells from oxidative damage
Hydrogen peroxide (H2O2) is one of the major contributors of oxidative stress and a number of studies have demonstrated that H2O2 induces oxidative stress by both acting as a reactive oxygen species (ROS) and a regulator of several redox enzymes [23, 24]. Therefore, we tested whether the antioxidant capacity determined in the chemical assays translated into a protective effect in enterocytes (CaCo-2) and fibroblastic cells (CHO) in culture challenged with H2O2.
Of all tested extracts, only the crude canola meal extract did not reduce cell death in both cell lines (Figures 4 A, 5 A). A dose dependent increase in cell viability was observed, however, when the canola meal accelerated solvent extract (Figures 4 B, 5 B) and the deodistillate extract (Figures 4 C, 5 C) were present during the oxidative challenge. Similarly, sinapic acid, a major phenolic in canola, protected the cells in a dose dependent manner (Figures 4 D, 5 D). We therefore conclude that the canola accelerated solvent extract, with its major component canolol, as well as the deodistillate extract (with some undetermined phenolic compounds), both exhibited an antioxidant effect in living cells.
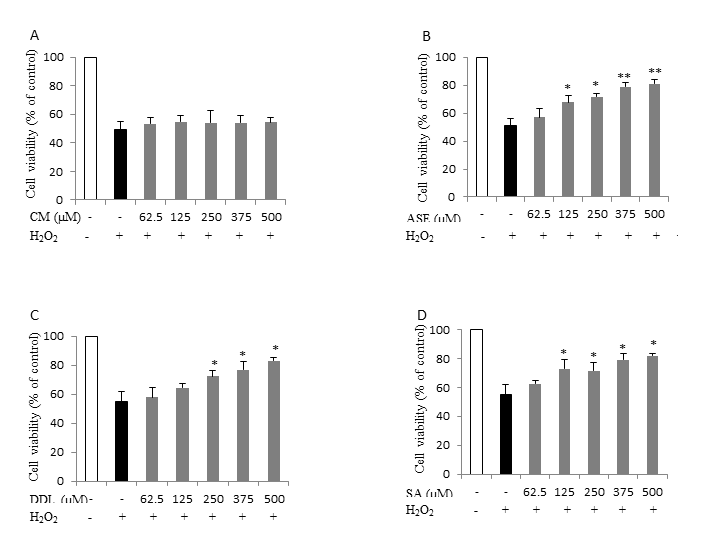
Figure 4. Effect of canola by-products extracts on CaCo-2 cell treated with H2O2-induced cytotoxicity. The data represents the mean ± standard deviation of at least three independent experiments. Asterisks indicates significant difference with control (* p<0.05, ** p<0.01). (A) CM, Canola meal crude extract. (B) ASE, Canola meal accelerated solvent extract. (C) DDL, Deodistillates extract. (D) SA, Sinapic acid.
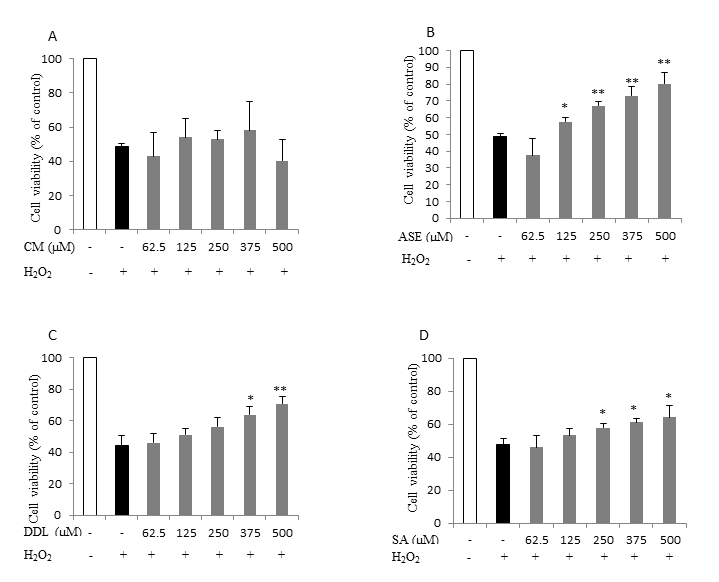
Figure 5. Effect of canola by-products extracts on CHO cell treated with H2O2-induced cytotoxicity. The data represents the mean ± standard deviation of at least three independent experiments. Asterisks indicates significant difference with H2O2 only treated cells (* p<0.05, ** p<0.01). (A) CM, Canola meal crude extract, (B) ASE, Canola meal accelerated solvent extract, (C) DDL, Deodistillates extract, (D) SA: Sinapic acid.
The presented data support both of our hypotheses: a) that canola by-products were a significant source of dietary phenols when extracted properly; and b) that the extracted phenols exhibited antioxidant activity and protected cells from oxidative damage.
The predominant phenols in whole canola seeds are sinapic acid derivatives including sinapine, sinapic acid, and sinapoyal glucose [13]. Canolol does not occur naturally in canola seeds, but is formed in crude canola oils [21]. In the canola meal crude extract sinapine was the predominant phenol, while in the accelerated solvent extract canolol was almost exclusively present. The presence of canolol in accelerated solvent extract was attributed to the high temperature and pressure which caused decarboxylation of the predominant phenol of canola meal (sinapine). Previous studies have demonstrated that thermal treatment of canola seeds and meals, such as roasting and microwave heating [25, 26] and supercritical extraction [27], can produce extracts or oils rich in canolol. Our result suggested that accelerated solvent extraction is a novel technique for production of canolol, and an alternative to previously published methods [5, 26, 27]. Moreover, some unknown phenols of slightly higher molecular weight were found in deodistillate extract.
The phenolic extracts of canola by-products showed significant antioxidant activities in the “test tube” systems. The Folin–Ciocalteu assay is the most commonly used method for measuring the total phenols of foods and nutraceuticals by determining the reducing capacity of the sample [28]. Other investigators suggested that the Folin–Ciocalteu assay should be seen as a measure of antioxidant capacity rather than a measure of total phenols [29]. The other antioxidant assays (FRAP and DPPH) are also based on electron transfer mechanism [30]. These assays don’t measure thiol antioxidants and are preferred by some researchers as methods for measuring antioxidant activity of foods [31]. The difference in antioxidant activities in “test tube” systems between the canola by-product samples in our study might be due to the different phenolic compounds in the extracts. The predominant phenolic compound in crude canola meal extract was sinapine which is a hydrophilic phenol. Several earlier studies have demonstrated that hydrophilic phenols can exhibit stronger antioxidant capacities than their lipophilic forms in the homogeneous polar media [32, 33]. Our results are in agreement with work by Terpinc’s et al. [34] who reported that decarboxylation products of hydroxycinnamic acids (e.g. canolol) exhibited poorer reducing capacities when measured by the Folin–Ciocalteu assay than the corresponding phenolic (e.g. sinapic acid). Sørensen et al. [35] also reported that sinapine and sinapic acid exhibit higher antioxidant activity than canolol as determined with FRAP and DPPH assays. However, both Terpinc’s et al. [34] and Sørensen et al. [35] suggested that canolol exhibited stronger antioxidant activity in emulsion systems.
Phenols can exhibit antioxidative effects in “test tube” systems relevant to food stability and increased shelf-life, however, this might not translate into protective effects in cells. Our data showed a clear dose dependent cyto-protection effect under oxidative challenge by H2O2, when the cells were incubated with the canola meal accelerated solvent extract (predominantly containing canolol) and the deodistillate extract (containing unknown high molecular weight phenols). An identical effect was achieved with sinapic acid, a known antioxidant found in canola seeds. The most potent cyto-protective extracts did not exhibit the most potent antioxidant activity when chemically assayed (Folin–Ciocaltau, FRAP, DPPH). We therefore propose that canola derived phenols could have an antioxidative effect in vivo, in general tissues or specifically in the intestinal tract. The antioxidant effect exhibited upon cells, which is important for health promoting properties, is different than the antioxidant activity in solution, which is more relevant for the protection of food (e.g. prolonged shelf-life).
The bioavailability and intracellular concentrations of canola derived phenols, such as canolol, sinapine and sinapic acid, are unknown. Phenolic bioavailability varies greatly although plasma levels are generally not exceeding low micromolar concentrations [36]. All extracts exhibited antioxidative protection from about 100 µM onwards in both cell lines tested, where CHO cells represent the parenchymal cells of organs and CaCo-2 cells represent the enterocyte. If phenols are bioavailable, the plasma concentration might reach 100 µM and so that the extracts could exhibit systemic antioxidant effects in vivo.
Even if the bioavailability is limited and the phenols not absorbed in the small intestine, they could reach concentrations exceeding 200 µM in the intestinal lumen. Our extracts protected the enterocytes (CaCo-2) from oxidative challenge at concentrations of 100 µM and higher, and enterocytes representing the outermost barrier towards the intestinal lumen are likely to experience these levels of phenols. This might be relevant in situations of oxidative challenge in the intestinal tract, such as evident in inflammatory bowel disease.
This study demonstrated that distinct phenolics can be obtained from different by-products of canola when subjected to different extraction techniques. Although the deodistillate and accelerated solvent extracts of canola showed lower antioxidant activity based on the F-C, FRAP and DPPH assays than crude canola meal extract and sinapic acid, they demonstrated significant higher antioxidant potentials in reducing H2O2 induced cytotoxicity in cells. This study has shown that canola by-products can be the sources of health promoting phenols, with potential as potent nutraceuticals. However, further studies are required to accurately assess the bioavailability of these phenolics.
Funding
The work as supported by Peter Eck’s Canada Research Chair and the Manitoba Agri-Food Research and Development Initiative (ARDI).
Author's contributions
Yougui Chen participated in the studies design, conducted the laboratory analyses and drafted the manuscript. Usha Thiyam-Hollander, Michael Eskin and Peter Eck designed the studies. Peter Eck edited the manuscript.
Abbreviations:
CaCo-2: Human colon adenocarcinoma cells; CHO: Chinese hamster ovary cell; DPPH: 2,2-diphenyl-1-picrylhydrazyl radical; FRAP: ferric reducing antioxidant power; IC50: Concentration to give 50% inhibition; ROS: reactive oxygen species
C. C. o. Canada, "Canola Meal Feed Industry Guide 4th Edition," ed, 2009.
E. Unger, "Processing," in Canola: chemistry, production, processing, and utilization vol. 1, N. Daun, E. M., and D. Hickling, Eds., 1 ed Urbana Academic Press and AOCS Press, 2011, pp. 163-188. PMid:21938494
View Article PubMed/NCBIS. I. Hashmi, P. N. Satwadhar, H. Deshpande, K. A. Syed, and B. Vibhute, "Rapeseed meal nutraceuticals," Journal of Oilseed Brassica, vol. 1, pp. 43-54, 2010 2010.
M. Naczk, R. Amarowicz, A. Sullivan, and F. Shahidi, "Current research developments on polyphenolics of rapeseed/canola: a review," Food Chemistry, vol. 62, pp. 489-502, Aug 1998. 00198-2
View ArticleR. Khattab, M. Eskin, M. Aliani, and U. Thiyam, "Determination of Sinapic Acid Derivatives in Canola Extracts Using High-Performance Liquid Chromatography," Journal of the American Oil Chemists Society, vol. 87, pp. 147-155, Feb 2010. PMid:20157351
View Article PubMed/NCBII. Rahman, S. K. Biswas, and P. A. Kirkham, "Regulation of inflammation and redox signaling by dietary polyphenols," Biochemical Pharmacology, vol. 72, pp. 1439-1452, Nov 30 2006. PMid:16920072
View Article PubMed/NCBIV. E. Steele, G. J. Kelloff, D. Balentine, C. W. Boone, R. Mehta, D. Bagheri, et al., "Comparative chemopreventive mechanisms of green tea, black tea and selected polyphenol extracts measured by in vitro bioassays," Carcinogenesis, vol.21, pp. 63-67, Jan 2000. PMid:10607735
View Article PubMed/NCBIG. D. Stoner and H. Mukhtar, "Polyphenols as Cancer Chemopreventive Agents," Journal of Cellular Biochemistry, pp. 169-180, 1995.
View ArticleJ. A. Vita, "Polyphenols and cardiovascular disease: effects on endothelial and platelet function," American Journal of Clinical Nutrition, vol. 81, pp. 292s-297s, Jan 2005. PMid:15640493
View Article PubMed/NCBIJ. A. N. Laranjinha, L. M. Almeida, and V. M. C. Madeira, "Reactivity of Dietary Phenolic-Acids with Peroxyl Radicals - Antioxidant Activity Upon Low-Density-Lipoprotein Peroxidation," Biochemical Pharmacology, vol. 48, pp. 487-494, Aug 3 1994. 90278-X
View ArticleM. Skerget, P. Kotnik, M. Hadolin, H. R. Hras, M. Simonic, and Z. Knez, "Phenols, proanthocyanidins, flavones and flavonols in some plant materials and their antioxidant activities," Food Chemistry, vol. 89, pp. 191-198, Feb 2005.
View ArticleA. Szydlowska-Czerniak, K. Trokowski, G. Karlovits, and E. Szlyk, "Determination of Antioxidant Capacity, Phenolic Acids, and Fatty Acid Composition of Rapeseed Varieties," Journal of Agricultural and Food Chemistry, vol. 58, pp. 7502-7509, Jul 14 2010. PMid:20545342
View Article PubMed/NCBIU. Thiyam, P. Claudia, U. Jan, and B. Alfred, "De-oiled rapeseed and a protein isolate: characterization of sinapic acid derivatives by HPLC-DAD and LC-MS," European Food Research and Technology, vol. 229, pp. 825-831, Sep 2009.
View ArticleS. Vuorela, A. S. Meyer, and M. Heinonen, "Impact of isolation method on the antioxidant activity of rapeseed meal phenolics," Journal of Agricultural and Food Chemistry, vol. 52, pp. 8202-8207, Dec 29 2004. PMid:15612818
View Article PubMed/NCBIH. Cao, I. Hininger-Favier, M. A. Kelly, R. Benaraba, H. D. Dawson, S. Coves, et al., "Green tea polyphenol extract regulates the expression of genes involved in glucose uptake and insulin signaling in rats fed a high fructose diet," J Agric Food Chem, vol. 55, pp. 6372-8, Jul 25 2007. PMid:17616136
View Article PubMed/NCBID. H. Kim, B. H. Yoon, W. Y. Jung, J. M. Kim, S. J. Park, D. H. Park, et al., "Sinapic acid attenuates kainic acid-induced hippocampal neuronal damage in mice," Neuropharmacology, vol. 59, pp. 20-30, Jul-Aug 2010. PMid:20363233
View Article PubMed/NCBIB. Harbaum-Piayda, K. Oehlke, F. D. Sonnichsen, P. Zacchi, R. Eggers, and K. Schwarz, "New polyphenolic compounds in commercial deodistillate and rapeseed oils," Food Chemistry, vol. 123, pp. 607-615, Dec 1 2010.
View ArticleW. Sumaryono, P. Proksch, V. Wray, L. Witte, and T. Hartmann, "Qualitative and Quantitative-Analysis of the Phenolic Constituents from Orthosiphon-Aristatus," Planta Medica, vol. 57, pp. 176-180, Apr 1991. PMid:17226146
View Article PubMed/NCBII. F. F. Benzie and J. J. Strain, "The ferric reducing ability of plasma (FRAP) as a measure of 'antioxidant power': The FRAP assay," Analytical Biochemistry, vol. 239, pp. 70-76, 1996. PMid:8660627
View Article PubMed/NCBIW. Brand-Williams, M. E. Cuvelier, and C. Berset, "Use of a free radical method to evaluate antioxidant activity," LWT - Food Science and Technology, vol. 28, pp. 25-30, 1995. 80008-5
View ArticleH. Kuwahara, A. Kanazawa, D. Wakamatu, S. Morimura, K. Kida, T. Akaike, et al., "Antioxidative and antimutagenic activities of 4-vinyl-2,6-dimethoxyphenol (Canolol) isolated from canola oil," Journal of Agricultural and Food Chemistry, vol. 52, pp. 4380-4387, Jul 14 2004. + PMid:15237940
View Article PubMed/NCBIJ. Dai and R. J. Mumper, "Plant Phenolics: Extraction, Analysis and Their Antioxidant and Anticancer Properties," Molecules, vol. 15, pp. 7313-7352, Oct 2010. PMid:20966876
View Article PubMed/NCBIC. H. Coyle and K. N. Kader, "Mechanisms of H2O2-induced oxidative stress in endothelial cells exposed to physiologic shear stress," ASAIO J, vol. 53, pp. 17-22, Jan-Feb 2007. PMid:17237644
View Article PubMed/NCBIT. K. Prasad, M. D. Anderson, B. A. Martin, and C. R. Stewart, "Evidence for Chilling-Induced Oxidative Stress in Maize Seedlings and a Regulatory Role for Hydrogen-Peroxide," Plant Cell, vol. 6, pp. 65-74, Jan 1994. PMid:12244221
View Article PubMed/NCBIR. Y. Khattab, M. N. A. Eskin, and U. Thiyam-Hollander, "Production of Canolol from Canola Meal Phenolics via Hydrolysis and Microwave-Induced Decarboxylation," Journal of the American Oil Chemists Society, vol. 91, pp. 89-97, Jan 2014.
View ArticleA. Spielmeyer, A. Wagner, and G. Jahreis, "Influence of thermal treatment of rapeseed on the canolol content," Food Chemistry, vol. 112, pp. 944-948, Feb 15 2009.
View ArticleF. Pudel, V. Habicht, T. Piofczyk, B. Matthäus, K.-W. Quirin, and A. Cawelius, "Fluidized bed treatment of rapeseed meal and cake as possibility for the production of canolol," OCL, vol. 21, p. D103, 2014.
View ArticleT. Katsube, H. Tabata, Y. Ohta, Y. Yamasaki, E. Anuurad, K. Shiwaku, et al., "Screening for antioxidant activity in edible plant products: Comparison of low-density lipoprotein oxidation assay, DPPH radical scavenging assay, and Folin-Ciocalteu assay," Journal of Agricultural and Food Chemistry, vol. 52, pp. 2391-2396, Apr 21 2004. PMid:15080652
View Article PubMed/NCBIJ. D. Everette, Q. M. Bryant, A. M. Green, Y. A. Abbey, G. W. Wangila, and R. B. Walker, "Thorough Study of Reactivity of Various Compound Classes toward the Folin-Ciocalteu Reagent," Journal of Agricultural and Food Chemistry, vol. 58, pp. 8139-8144, Jul 28 2010. PMid:20583841
View Article PubMed/NCBIR. L. Prior, X. L. Wu, and K. Schaich, "Standardized methods for the determination of antioxidant capacity and phenolics in foods and dietary supplements," Journal of Agricultural and Food Chemistry, vol. 53, pp. 4290-4302, May 18 2005. PMid:15884874
View Article PubMed/NCBIA. C. Tan, I. Konczak, I. Ramzan, and D. M. Y. Sze, "Antioxidant and cytoprotective activities of native Australian fruit polyphenols," Food Research International, vol. 44, pp. 2034-2040, Aug 2011.
View ArticleM. J. Jordan, V. Lax, M. C. Rota, S. Loran, and J. A. Sotomayor, "Relevance of Carnosic Acid, Carnosol, and Rosmarinic Acid Concentrations in the in Vitro Antioxidant and Antimicrobial Activities of Rosmarinus officinalis (L.) Methanolic Extracts," Journal of Agricultural and Food Chemistry, vol. 60, pp. 9603-9608, Sep 26 2012. PMid:22957812
View Article PubMed/NCBIL. Perez-Fons, M. T. Garzon, and V. Micol, "Relationship between the Antioxidant Capacity and Effect of Rosemary (Rosmarinus officinalis L.) Polyphenols on Membrane Phospholipid Order," Journal of Agricultural and Food Chemistry, vol. 58, pp. 161-171, Jan 13 2010. PMid:19924866
View Article PubMed/NCBIP. Terpinc, T. Polak, N. Segatin, A. Hanzlowsky, N. P. Ulrih, and H. Abramovic, "Antioxidant properties of 4-vinyl derivatives of hydroxycinnamic acids," Food Chemistry, vol. 128, pp. 62-69, Sep 1 2011. PMid:25214330
View Article PubMed/NCBIA. D. M. Sorensen, J. Friel, J. K. Winkler-Moser, C. Jacobsen, D. Huidrom, N. Reddy, et al., "Impact of endogenous canola phenolics on the oxidative stability of oil-in-water emulsions," European Journal of Lipid Science and Technology, vol. 115, pp. 501-512, May 2013.
View ArticleA. Scalbert and G. Williamson, "Dietary intake and bioavailability of polyphenols," Journal of Nutrition, vol. 130, pp. 2073s-2085s, Aug 2000. PMid:10917926
View Article PubMed/NCBI