Charlie Li 1
1 Work was previously performed at Department of Food Science and Technology, Oregon State University, Corvallis, OR
Affiliated with: Department of Environmental Toxicology, University of California-Davis, CA 95616
© 2019 Sift Desk Journals. All Rights Reserved
VOLUME: 3 ISSUE: 6
Page No: 527-533
Charlie Li 1
1 Work was previously performed at Department of Food Science and Technology, Oregon State University, Corvallis, OR
Affiliated with: Department of Environmental Toxicology, University of California-Davis, CA 95616
Dequan Zhang(dqzhang0118@126.com)
Yu-Pei Chen(ypchen666@mail2000.com.tw)
Charlie Li, The Effect of Natural Antioxidant Extracted from Shrimp Shell on Oxidative and Hydrolytic Rancidity of Sable Fish Mince(2018)SDRP Journal of Food Science & Technology 3(6)
Antioxidant from shrimp waste was extracted and purified by silica gel glass column chromatography. Antioxidant activity was measured for crude and purified fractions. Both crude extract and purified fractions were applied into sablefish mince and stored at refrigerator and freezer temperatures for various times. Crude extract, and 0.20% and 0.50% (w/w) purified antioxidant from shrimp waste significantly lowered TBA and peroxide values of sablefish minces stored at 40C for 8 days and frozen for 120 days, respectively. Results indicated that crude extract and 0.20% and 0.50% purified antioxidant effectively inhibited lipid oxidation. After 120 days of storage, 0.20% antioxidant had significantly higher free fatty acid content than those of other treatments, while 0.10% and 0.50% antioxidant had significantly higher free fatty acid production compared to other samples after 4 days storage at 40C.
Keywords: Natural Antioxidant, Sable fish mince, lipid oxidation
Abbreviation:
2-thiobarbituric acid (TBA)
Peroxide value (PV)
Free Fatty Acid (FFA)
Lipid oxidation causes various off-flavor, off-odor, texture, and nutrient damage in muscle food. Seafood is especially susceptible to the deterioration process due to the presence of poly-unsaturated fatty acids. Fish containing high amount of fat content, such as, sablefish, and mackerel, are extremely susceptible to autoxidation and rapid deterioration resulting in unstable food products.
Sablefish, or black cod (Anoplopoma fimbria) is distributed in the North Pacific Ocean from northern Mexico northward to the Bering Sea, and along the Asian Coast from Kamchatka southward to the northeastern Coast of Japan. As sablefish are particularly well suited for salting and smoke curing, they are actively fished for local markets by both Canada and the United States.
Oxidative rancidity in seafood could be inhibited by several methods [1]. The most common method is to lower the temperature as in chilled and frozen storage. Another method is to eliminate oxygen by vacuum packing and/or modified atmosphere storage. A third method uses the addition of synthetic and natural antioxidants to retard lipid oxidation. Although synthetic commercial antioxidants such as butylated hydroxyanisole (BHA), butylated hydroxytoluene (BHT), propyl gallate (PG), tertiary-butylhydroquinone (TBHQ), etc. have been widely used to inhibit lipid oxidation of food products, there has been some concern over the safety of synthetic antioxidants [2-3]. There are some other disadvantages; such as, cost and/or the fear of an adverse reaction by the consumer in utilizing synthetic additives. Several authors have reported that various antioxidants were used to inhibit lipid oxidation in fish systems [4-10]. Moreover, some natural products such as polyphenols, dried spices, and fresh spices were used to inhibit oxidative rancidity in ground fish (Scomberomorus commersoni) (11-14). Hydrolytic rancidity is caused by fat hydrolysis and free fatty acids are released.
The objective was to test whether the antioxidant from shrimp waste could inhibit the lipid oxidation in sablefish mince.
Materials
Samples of shrimp waste including peel and head parts (Pandalus jordani), and fresh sablefish (Anoplopoma fimbria) were obtained from a local seafood company and transported in ice to the Oregon State University Seafood Laboratory in Astoria, Oregon. Shrimp waste samples were vacuum packed, blast frozen, and stored at -200C.
Preparation of Antioxidant Crude Extract
One kg sample of shrimp waste was extracted with 2,000 mL of 95% ethanol solution. The mixture was blended until homogeneous, using a Waring blender. The slurry was filtered through a funnel using Whatman No. 1 filter paper. Antioxidant activity of the crude extract was then evaluated in a β-carotene-linoleic emulsion as described by Marco (1968). The ability of antioxidant to prevent the oxidative destruction of the β-carotene and linoleic acid emulsion is expressed as the decrease in absorbance at 470 nm. The solvent was evaporated to dryness by rotary evaporation. The dried powder was prepared for applying into sablefish mince.
Silica Gel Glass Column Chromatography
A 2,000 mL crude ethanol extract was concentrated to about 20 mL by rotary evaporator (Buchi, Switzerland) and subjected to column chromatography. A glass column of a diameter of 2.5 cm and length of 58.4 cm was packed with silica gel 60-200 mesh in hexane slurries. The fractions were first eluted with 150 ml 100% hexane, and then 20% acetone in hexane (A/H), 40% (A/H), 60% (A/H), 80% (A/H), 100% acetone, 20% methanol in acetone (M/A), 40% (M/A), 60% (M/A), 80% (M/A), and finally 100% methanol. The flow rate of the eluents was kept at 2.5-3.0 ml/min. Antioxidant activity of fractions was measured in the β-carotene-linoleic emulsion system. The most active fractions that were eluted with methanol in acetone (80%) were repeatedly collected and evaporated to dryness.
Preparation of Sablefish Mince
Fresh sablefish were cleaned, and gutted. After filleting, the fillets were ground using a Hobart Model N50 grinder. Approximately 26% mince was obtained from raw fish. The fat content of the fish was analyzed according to modified Folch extraction method [16]. Samples of 0.50% (w/w) crude extract and 0.10%, 0.20%, and 0.50% (w/w) purified antioxidant from shrimp waste were added to the mince on a total weight basis. A commercial antioxidant, 0.02% Tenox 6 (a mixture of BHA, BHT, PG, and citric acid), was applied as a percentage of fat content of the sablefish mince. The control group contained no antioxidant. Each mixture was homogenized with a Kitchen Aid mixer. Samples of each treatment, 60 g of mince, were put into individual plastic bags. The samples were stored in the blast freezer at -200C for 120 days. 2-Thiobarbituric acid value (TBA), peroxide value (PV), and free fatty acid (FFA) were analyzed at 0, 30, 60, 90, and 120 days. Another set of samples was stored at refrigerated temperature (40C) for 8 days and sampled at 0, 4, and 8 days for TBA, PV and FFA analyses.
TBA Analysis
TBA was determined as described by Yu and Sinnhuber [17] with some modification. About 200 mg of minced samples were mixed with 3 drops of antioxidant mixture containing TBHQ in propylene glycol and Tenox 6, 3 ml of TBA solution, and 17 ml of trichloroacetic acid (TCA)-HCL reagent. Samples were flushed with nitrogen and tightly closed with a screw cap, placed in a boiling water bath for 30 min and cooled in tap water. 5 ml of chloroform was added and about 15 ml of the colored solution was transferred into a 50 ml conical centrifuge tube. The tubes were then centrifuged for 10 minutes at 3000 rpm. A portion of the clear, colored, aqueous solution was transferred to a cuvette for absorbance measurement at 535 nm using Beckman spectrophotometer DU 640 (Beckman, Fullerton, CA).
Determination of Peroxide Value
The fat from fish mince was extracted utilizing the procedure of Bligh and Dyer [16], and PV of extracted fat were measured as described in AOAC (28.022, 28.023, 50.037, and 50.038), 1975. PV was calculated as PV (milliequiv. Peroxide/kg sample) = S x N x 1000/g sample where S=ml Na2S2O3 and N=normality Na2S2O3 solution.
Determination of Free Fatty Acid
Free fatty acid (FFA) was determined according to the procedure of Ke and Woyewada [18]. Aqueous sodium hydroxide was added until an end-point was reached as indicated by a color change of meta-cresol purple. The FFA was represented as µmol g-1 of tissue sample.
Statistical Analysis
Analysis was performed with two replications. Data was subjected to one factor analysis of variance (ANOVA) to compare treatments. Least Significant Difference (LSD) was further applied to determine differences between treatments if F-test was significant.
The antioxidant activity of crude extract and purified fractions is shown in Fig 1. Crude extract and the fraction 10 eluted with methanol in acetone (80%) showed a strong antioxidant activity while fractions 3 and 9, eluted with 40% acetone in hexane and 60% methanol in acetone, respectively, also had some antioxidant activity (Fig 1). Polar compound(s) would have longer retention times than nonpolar compound(s) under these conditions. It was previously reported that antioxidants from ginger and Eucalyputs leaves were fractionated by silica gel column chromatography [19-20]. These antioxidants were eluted by nonpolar solvents such as hexane; hexane and ethyl acetate (9:1 v/v), etc. The data shown here from silica gel column chromatography was consistent with our previous results [15], which indicated that the antioxidant possesses polar characteristics.
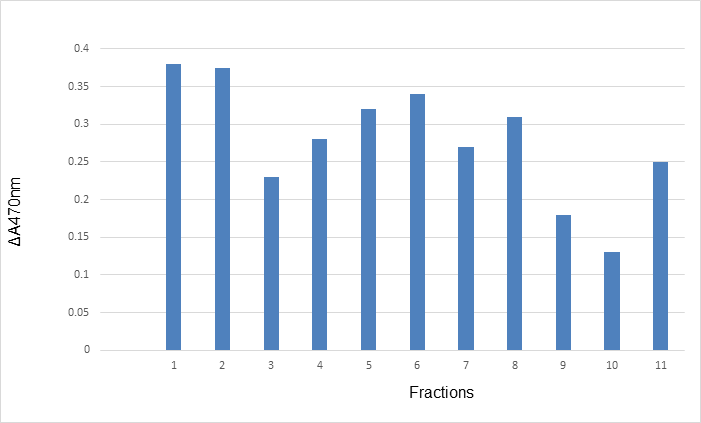
Fig. 1. Antioxidant activity of crude extract and purified fractions eluted from silica gel glass column chromatography.
Effect of Antioxidants at Refrigerator Storage
The results from fat analysis showed that the sablefish mince contained about 7.60% total fat content according to the Bligh and Dyer method [16]. Based on the fat content, the sablefish belonged to fatty fish species (3-10% lipids), such as, yellowtail, halibut and rosefish [21], but contains less total fat compared to mince from block frozen Gulf menhaden and IQF Gulf menhaden [9].
The TBA values for sablefish mince samples with treated and untreated antioxidants for 8 days of storage are shown in Fig. 2. After 4 days of storage, the samples treated with crude extract, and 0.20% and 0.50% antioxidant from shrimp waste had significantly lower TBA values than the control sample containing no antioxidant (P<0.01); while 0.10% antioxidant had higher TBA values. Differences in TBA values between crude extract, 0.20%, and 0.50% antioxidant were not significant during storage tests. At the 8 days of storage, crude extract, 0.2% and 0.5% antioxidants from shrimp shell waste had about 85%, 82%, and 84% TBA value compared with the negative control.
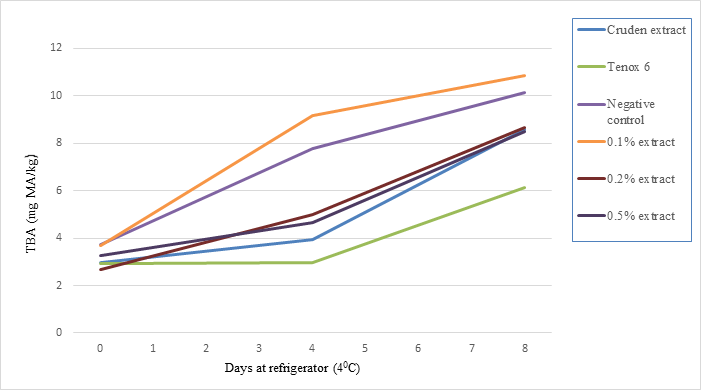
Fig. 2. The effect of antioxidant treatments on TBA values of sablefish mince samples stored at refrigerator temperature (40C) for 8 days. Crude extract, 0.5%, or 0.10%, 0.20% and 0.50% purified antioxidant was added to mince samples.
The change in peroxide value of the mince samples with and without antioxidant treatments stored at 40C is shown in Fig. 3. Crude extract, and 0.20% and 0.50% purified antioxidant, had significantly lower PV than the control after 8 days of storage (P<0.01). At the 8 days of storage, crude extract, 0.2% and 0.5% antioxidants from shrimp shell waste had about 72%, 70%, and 70% PV compared with the negative control while 0.10% treatment was not as effective in decreasing PV. Statistical analysis indicated that there was no significant difference between the effects of crude extract, and 0.20%, and 0.50% purified antioxidant on PV. The results from PV were consistent with those of TBA values, in that antioxidant at 0.10% concentration was not sufficient to inhibit lipid oxidation; while crude extract, 0.20%, and 0.50% antioxidant lowered lipid oxidation in the sablefish mince to some degrees. The results suggested that the system might become saturated with antioxidant at level at 0.2% since no further decrease in lipid oxidation was observed.
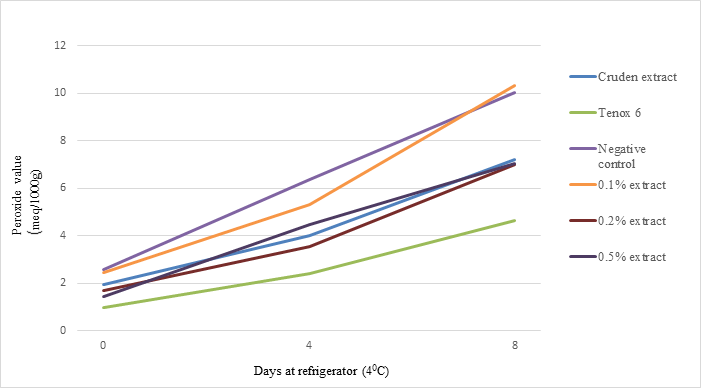
Fig. 3. Effect of crude extract and purified antioxidant treatments on PV of sablefish mince samples stored at refrigerator temperature (40C) for 8 days.
Effect of antioxidant on free fatty acid values was evaluated. Samples treated with 0.1% and 0.5% antioxidants increased more rapidly in FFA during 8 days of storage at 40C than controls (Fig. 4). After 8 days, the samples treated with 0.50% antioxidant had given values 9.57±0.62 µmol/g tissue as compared to control, which had 7.96±0.25 µmol/g tissue; but 0.10%, 0.20% purified antioxidant and crude extract did not affect the free fatty acid significantly. At day 8, Crude extract, 0.2% and 0.5% antioxidants from shrimp shell waste had about 95%, 97%, and 120% FFA values compared with the negative control.The results suggested that antioxidant at higher concentration from shrimp waste may increase free fatty acid production during refrigerated storage.
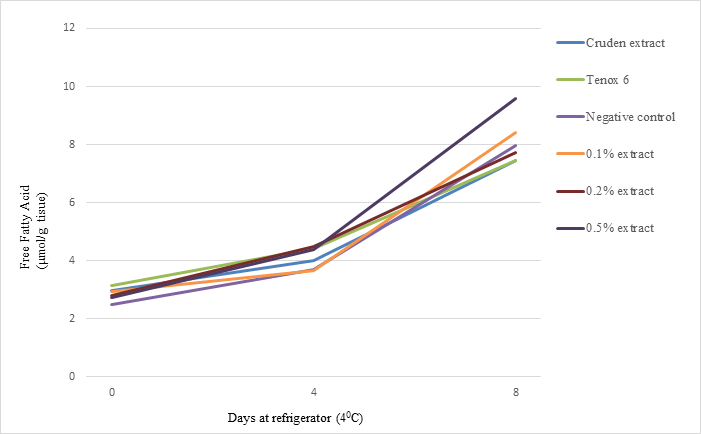
Fig. 4. Effects of crude extract and purified antioxidant treatments on FFA of sablefish mince samples at refrigerator temperature (40C) for 8 days.
Research has been conducted to compare the effectiveness of varying levels of natural antioxidants at refrigerated temperature. It was reported that α-tocopherol at a level of 200 ppm was effective in preventing lipid oxidation of the ground fish at 40C, while a lower concentration (30 ppm) had no effect [13]. We have found that the same concentration of antioxidant from shrimp waste (0.20%) had an effect on lipid oxidation. Furthermore, it was reported that polyphenols had a concentration-dependent effect on inhibition of lipid oxidation in which the concentration at 0.005% level was more effective than at 0.001% in fresh cooked ground fish [14]. Decker and Crum [22] reported that carnosine at concentrations of 0.50%, 1.0%, and 1.5% significantly reduced the formation of TBA values by 33-43 % in cooked, unsalted ground pork. Compared to the effect of carnosine, natural antioxidant from shrimp waste appears to inhibit lipid oxidation at lower concentrations. However, the effectiveness of antioxidants on lipid oxidation might vary with different products.
Effect of Antioxidants during Frozen Storage
TBA values of all samples increased after 1 month of storage as shown in Fig. 5. Samples treated with crude extract, 0.20% and 0.50% antioxidant, and Tenox 6 had significantly lower TBA values than the control group after 2 months of frozen storage (P<0.05). There were no differences (P>0.05) in TBA values between samples with either 0.20%, 0.50% levels, or crude extract treatments. At the end of 4 months of storage, crude extract, 0.2% and 0.5% antioxidants from shrimp shell waste had about 80%, 83%, and 72% TBA value compared with the negative control. The effectiveness of crude extract and purified antioxidants were less than that of Tenox 6 on inhibiting oxidation.
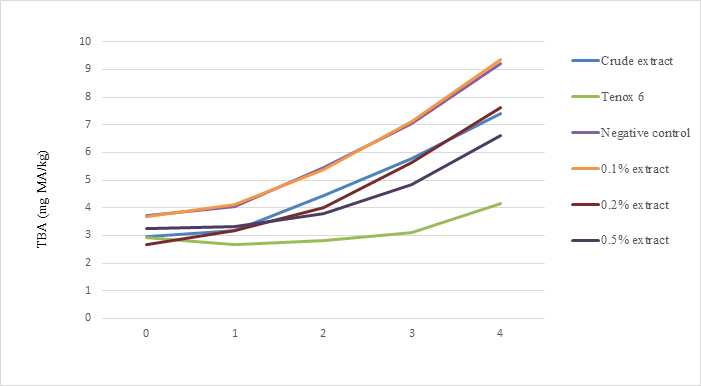
Fig. 5. Effect of crude extract and purified antioxidant treatments on TBA values of sablefish mince samples in frozen storage (-200C) for 4 months.
Peroxide values (PV) of sablefish mince samples are shown in Fig 6. After 4 months of storage, 0.20% and 0.50% antioxidant had significantly lower PV compared to controls. Antioxidant at 0.2% and 0.5% levels was nearly as effective as Tenox 6, while crude extract and 0.1% antioxidant were less effectiveness at 120 days. At the end of 4 months of storage, crude extract, 0.2% and 0.5% antioxidants from shrimp shell waste had about 88%, 57%, and 63% PV compared with the negative control.
The effective of several antioxidants on protecting oxidative rancidity in mackerel mince was reported [9]. They reported that ascorbic acid and erythorbic acid were very strong antioxidants with only a very little change in PV during frozen storage. They also found that Tenox 20 (0.08% TBHQ) had an effective in decreasing PV, but did not completely control lipid oxidation, which is similar to the result of the antioxidant from shrimp waste. Several natural antioxidants have been used to inhibit lipid oxidation in food. Boyd et al. [5] reported that rosemary extract was effective in decreasing TBA values in cooked sea trout. While rosemary extracts alone or in combination with tocopherols, had little effect on PV. Antioxidant activity of rosemary oleoresin was comparable to a commercial blend of BHA/BHT/citric acid in suppressing lipid autoxidation in turkey sausage [23]. Other spices, such as, cloves, cinnamon, black pepper, thyme, etc., also showed antioxidative activity to inhibit lipid oxidation in a variety of food systems [24-25].
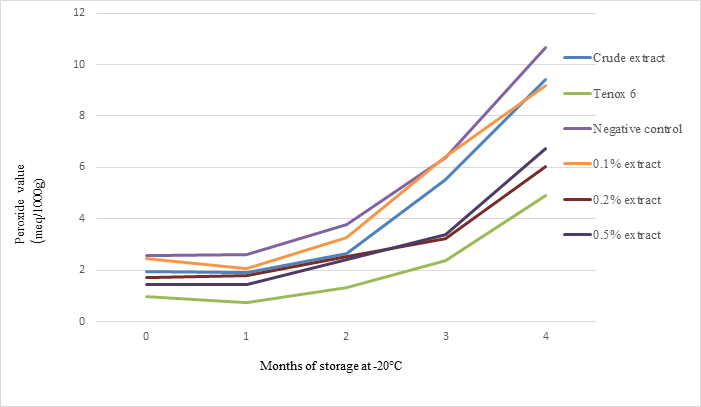
Fig. 6. Effect of crude extract and purified antioxidant treatments on PV of sablefish mince samples in frozen storage (-200C) for 4 months.
Our results showed that free fatty acid values of all samples including controls increased with prolonged frozen storage times up to 4 months (Fig. 7). After 4 months of storage, 0.20% antioxidant had a significantly higher free fatty acid compared to other treatments. These results differed from the results in refrigerated storage, in which 0.50% antioxidant increased FFA production. Crude extract, 0.2% and 0.5% antioxidants from shrimp shell waste had about 100%, 117%, and 106% FFA values compared with the negative control.
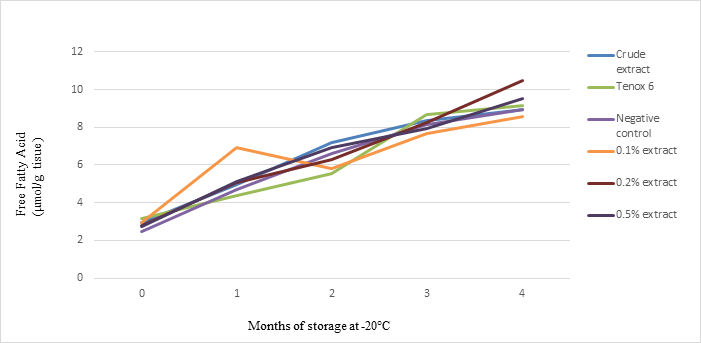
Fig. 7. Effect of crude extract and purified antioxidant treatments on free fatty acid (FFA) of sablefish mince samples in frozen storage (-200C) for 4 months.
It has been suggested that TBHQ possibly stimulated the activity of lipase, which released more free fatty acid in mullet muscle during frozen storage [26]. Boyd et al. [5] also reported that cooked fish flakes treated with antioxidants such as TBHQ and rosemary extract produced higher FFA values compared with the untreated control. Hwang and Regenstein (1988) reported that Tenox 20 (0.08% TBHQ) treatment at -70C did not inhibit lipid hydrolysis on menhaden mince. Previous studies conducted by [27] and Shewfelt et al. [28] have shown that increased in FFA did not correlate to increased lipid oxidation.
Our previous studies indicated that the antioxidant isolated from shrimp waste is an ortho-substituted phenolic compound which could play a role in providing hydrogen or electrons to a free radical. The polar characteristic of the natural antioxidant from shrimp waste makes it easy to incorporate into food products. The natural antioxidant derived from seafood source, which possesses similar flavor and low cost, has potential use for prolonging the shelf life of various seafood products by inhibiting lipid oxidation. Another potential application is that the antioxidant compound would be useful in stabilizing red color in the skin of number of rockfish species and help to attract a higher price in more discerning markets.
Hamilton, RJ. 1983. The Chemistry of Rancidity in Foods. Ch. 1. In Rancidity in Foods, J.C. Allen and R.J. Hamilton (Ed.), p.1. Applied Science Publishers, New York.
Chen C, Pearson AM, Gray JI, Bennink MR (1986) Antioxidant inhibition of mutagenicity in fried meat processing. Euro Meeting of Meat Res Wor. 1: 211-213.
Imaida K, Fukishima S, Shirai T, Ohtani M, Nakamish K, Ito M (1983). Promoting activities of butylated hydroxyanisole and butylated hydroxytoluene on 2-day urinary bladder carcinogenesis and inhibition of glutamyl transpeptidase positive foci development in the liver of rats. Carcinogen. 4: 895-899.
View ArticleLee S, Faustman C, Djordjevic D, Faraji H, Decker EA (2006). Effect of antioxidants on stabilization of meat products fortified with n-3 fatty acids. Meat Sci. 72:18-24. PMid:22061369
View Article PubMed/NCBIBoyd LC, Green DP, Giesbrecht, FB, King MF (1993) Inhibition of oxidative rancidity in frozen cooked fish flakes by tert-butylhydroquinone and rosemary. J. Sci. Food Agric. 61: 87-93.
View ArticleDragoev SG (2008) Inhibition of lipid peroxidation of frozen mackerel by pre-storage antioxidant superficial treatment. Bulgarian J. Agric Sci. 14: 283-289.
Jiang J, Xiong Y (2016). Natural antioxidants as food and feed additives to promote health benefits and quality of meat products: A review. Meat Sci. 120: 107-117. PMid:27091079
View Article PubMed/NCBISecci G, Parisi G (2016) From farm to fork: lipid oxidation in fish products: A review. Italian J. Ani Sci. 15:124-136.
View ArticleEymard S, Jacobsen C, Baron CP (2010) Assessment of Washing with Antioxidant on the Oxidative Stability of Fatty Fish Mince during Processing and Storage. J. Agric & Food Chem. 58:6182–6189. PMid:20423096
View Article PubMed/NCBIWade S, Fang X (1992) The synergistic antioxidant effect of rosemary extract and α-tocopherol in sardine oil model system and frozen-crushed fish meat. J. Food Proc. Pres. 16: 263-274.
View ArticleBAEK, N. 2012. Http://vtechworks.lib.vt.edu/bitstream/handle/10919/19214/Baek_N_T_2013.pdf?sequence=1
Halldorsdottir SM, Kristinsson, HG, Sveinsdottir, H, Thorkelsson G, Hamaguchi PY (2013) The effect of natural antioxidants on haemoglobin-mediated lipid oxidation during enzymatic hydrolysis of cod protein. Food Chem. 141: 914–919. PMid:23790867
View Article PubMed/NCBIRupasinghe HP, Erkan N, Yasmin A (2010) Antioxidant protection of eicosapentaenoic acid and fish oil oxidation by polyphenolic-enriched apple skin extract. J. Agric. Food Chem. 58:1233-9. PMid:20000574
View Article PubMed/NCBITaghvaei M, Jafari SM (2015) Application and stability of natural antioxidants in edible oils in order to substitute synthetic Additives. J. Food Sci. 52: 1272–1282.
View ArticleLi, S.J. (Aug. 2, 1994)
View ArticleBligh EG, Dyer WJ (1959) A rapid method of total lipid extraction and purification. Can. J. Biochem Physiol. 37: 911-917. PMid:13671378
View Article PubMed/NCBIYu TC, Sinnhuber, RO (1967) An improved 2-thiobarbituric acid (TBA) procedure for the measurement of autoxidation in fish oil. J. Am Oil. Chem Soc. 44: 256-258. PMid:6068658
View Article PubMed/NCBIKe PJ, Woyewoda, AD (1978) A titrimetric method for determination of free fatty acids in tissues and lipids with ternary solvents and m-cresol purple indicator. Anal Chem Acta., 99: 387-391. 83585-6
View ArticleOsaka T, Nakimi, M (1981) A novel type of antioxidant isolated from leaf wax of Eucalptus leaves. Agric. Biol. Chem. 45: 735-739.
Kikuzaki H, Nakatani N (1993) Antioxidant effects of some ginger constituents. J. Food Sci., 58: 1407-1410.
View ArticleSHENOUDA SY(1980). Theories of protein denaturation during frozen storage of fish flesh. Ch. 26 in Advances in Food Research, C.O. Chichester (Ed.), p. 275-311. Academic Press., New York, N.Y. 60320-1
View ArticleDecker EA, Crum AD (1993) Antioxidant activity of carnosine in cooked ground pork. Meat Sci. 34: 245-253. 90031-C
View ArticleBarbut S, Josephson, DB, Maurer AJ (1985) Antioxidant properties of rosemary oleoresin in turkey sausage. J. Food Sci. 50:1356-135.
View ArticleAl-Jalay B, Blank G, McConnell B, Al-Khayat, M (1987) Antioxidant activity of selected spices used in fermented meat sausage. J. Food Protect. 50: 25-27.
View ArticleChipault JR, Mizuno GR, Hawkins JM, Lundberg WO (1952) The antioxidant properties of natural spices. Food Res.17:46-55.
View ArticleDeng JC (1978) Effect of iced storage on free fatty acid production and lipid oxidation in mullet muscle. J. Food Sci., 43, 337-340.
View ArticleMai J, Kinsella, JE (1979) Changes in lipid composition of cooked minced carp (Cyprinus carpio) during frozen storage. J. Food Sci. 44: 1619-1626.
View ArticleShewfelt RL, McDonald RE, Hultin HO (1981) Effect of phospholipid hydrolysis on lipid oxidation in flunder muscle microsomes. J. Food Sci., 46: 1297-1301.
View Article